Graft-versus-host disease on:
[Wikipedia]
[Google]
[Amazon]
Graft-versus-host disease (GvHD) is a syndrome, characterized by inflammation in different organs. GvHD is commonly associated with bone marrow transplants and stem cell transplants.
White blood cells of the donor's immune system which remain within the donated tissue (the graft) recognize the recipient (the host) as foreign (non-self). The white blood cells present within the transplanted tissue then attack the recipient's body's cells, which leads to GvHD. This should not be confused with a
 In the clinical setting, graft-versus-host disease is divided into
In the clinical setting, graft-versus-host disease is divided into
 Three criteria, known as the Billingham criteria, must be met in order for GvHD to occur.
* An immuno-competent graft is administered, with viable and functional immune cells.
* The recipient is immunologically different from the donor – histo-incompatible.
* The recipient is immunocompromised and therefore cannot destroy or inactivate the transplanted cells. In particular, it involves an inability of the recipient's
Three criteria, known as the Billingham criteria, must be met in order for GvHD to occur.
* An immuno-competent graft is administered, with viable and functional immune cells.
* The recipient is immunologically different from the donor – histo-incompatible.
* The recipient is immunocompromised and therefore cannot destroy or inactivate the transplanted cells. In particular, it involves an inability of the recipient's
transplant rejection
Transplant rejection occurs when transplanted tissue is rejected by the recipient's immune system, which destroys the transplanted tissue. Transplant rejection can be lessened by determining the molecular similitude between donor and recipient ...
, which occurs when the immune system of the transplant recipient rejects the transplanted tissue; GvHD occurs when the donor's immune system's white blood cells reject the recipient. The underlying principle ( alloimmunity) is the same, but the details and course may differ. GvHD can also occur after a blood transfusion
Blood transfusion is the process of transferring blood products into a person's circulation intravenously. Transfusions are used for various medical conditions to replace lost components of the blood. Early transfusions used whole blood, but mo ...
if the blood products used have not been irradiated or treated with an approved pathogen reduction system.
Types
 In the clinical setting, graft-versus-host disease is divided into
In the clinical setting, graft-versus-host disease is divided into acute
Acute may refer to:
Science and technology
* Acute angle
** Acute triangle
** Acute, a leaf shape in the glossary of leaf morphology
* Acute (medicine), a disease that it is of short duration and of recent onset.
** Acute toxicity, the adverse ef ...
and chronic forms, and scored or graded on the basis of the tissue affected and the severity of the reaction.
In the classical sense, acute graft-versus-host disease is characterized by selective damage to the liver
The liver is a major organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the synthesis of proteins and biochemicals necessary for digestion and growth. In humans, it i ...
, skin
Skin is the layer of usually soft, flexible outer tissue covering the body of a vertebrate animal, with three main functions: protection, regulation, and sensation.
Other animal coverings, such as the arthropod exoskeleton, have different ...
(rash), mucosa
A mucous membrane or mucosa is a membrane that lines various cavities in the body of an organism and covers the surface of internal organs. It consists of one or more layers of epithelial cells overlying a layer of loose connective tissue. It i ...
, and the gastrointestinal tract
The gastrointestinal tract (GI tract, digestive tract, alimentary canal) is the tract or passageway of the digestive system that leads from the mouth to the anus. The GI tract contains all the major organs of the digestive system, in humans and ...
. Newer research indicates that other graft-versus-host disease target organs include the immune system (the hematopoietic system, e.g., the bone marrow
Bone marrow is a semi-solid biological tissue, tissue found within the Spongy bone, spongy (also known as cancellous) portions of bones. In birds and mammals, bone marrow is the primary site of new blood cell production (or haematopoiesis). It i ...
and the thymus
The thymus is a specialized primary lymphoid organ of the immune system. Within the thymus, thymus cell lymphocytes or '' T cells'' mature. T cells are critical to the adaptive immune system, where the body adapts to specific foreign invaders ...
) itself, and the lung
The lungs are the primary organs of the respiratory system in humans and most other animals, including some snails and a small number of fish. In mammals and most other vertebrates, two lungs are located near the backbone on either side of ...
s in the form of immune-mediated pneumonitis. Biomarkers can be used to identify specific causes of GvHD, such as elafin in the skin. Chronic graft-versus-host disease also attacks the above organs, but over its long-term course can also cause damage to the connective tissue
Connective tissue is one of the four primary types of animal tissue, along with epithelial tissue, muscle tissue, and nervous tissue. It develops from the mesenchyme derived from the mesoderm the middle embryonic germ layer. Connective tiss ...
and exocrine glands.
Mucosal damage to the vagina
In mammals, the vagina is the elastic, muscular part of the female genital tract. In humans, it extends from the vestibule to the cervix. The outer vaginal opening is normally partly covered by a thin layer of mucosal tissue called the hymen ...
can result in severe pain
Pain is a distressing feeling often caused by intense or damaging stimuli. The International Association for the Study of Pain defines pain as "an unpleasant sensory and emotional experience associated with, or resembling that associated with, ...
and scarring, and appears in both acute and chronic GvHD. This can result in an inability to have sexual intercourse
Sexual intercourse (or coitus or copulation) is a sexual activity typically involving the insertion and thrusting of the penis into the vagina for sexual pleasure or reproduction.Sexual intercourse most commonly means penile–vaginal pene ...
.
Acute
The ''acute'' or ''fulminant'' form of the disease (aGvHD) is normally observed within the first 10 to 100 days post-transplant, and is a major challenge to transplants owing to associated morbidity and mortality. About one-third to one-half of allogeneic transplant recipients will develop acute GvHD. It's less common in younger patients and in those with closerhuman leukocyte antigens
The human leukocyte antigen (HLA) system or complex is a complex of genes on chromosome 6 in humans which encode cell-surface proteins responsible for the regulation of the immune system. The HLA system is also known as the human version of th ...
(HLA) matches between donor and the patient.
The first signs are usually a rash, burning, and redness of the skin on the palms and soles. This can spread over the entire body. Other symptoms can include Nausea, vomiting, stomach cramps, diarrhea (watery and sometimes bloody), loss of appetite, jaundice, abdominal pain, and weight loss.
Acute GvHD of the GI tract
The gastrointestinal tract (GI tract, digestive tract, alimentary canal) is the tract or passageway of the digestive system that leads from the mouth to the anus. The GI tract contains all the major organs of the digestive system, in humans an ...
can result in severe intestinal inflammation, sloughing of the mucosal membrane, severe diarrhea, abdominal pain, nausea, and vomiting. This is typically diagnosed via intestinal biopsy. Liver GvHD is measured by the bilirubin level in acute patients. Skin GvHD results in a diffuse red maculopapular rash, sometimes in a lacy pattern.
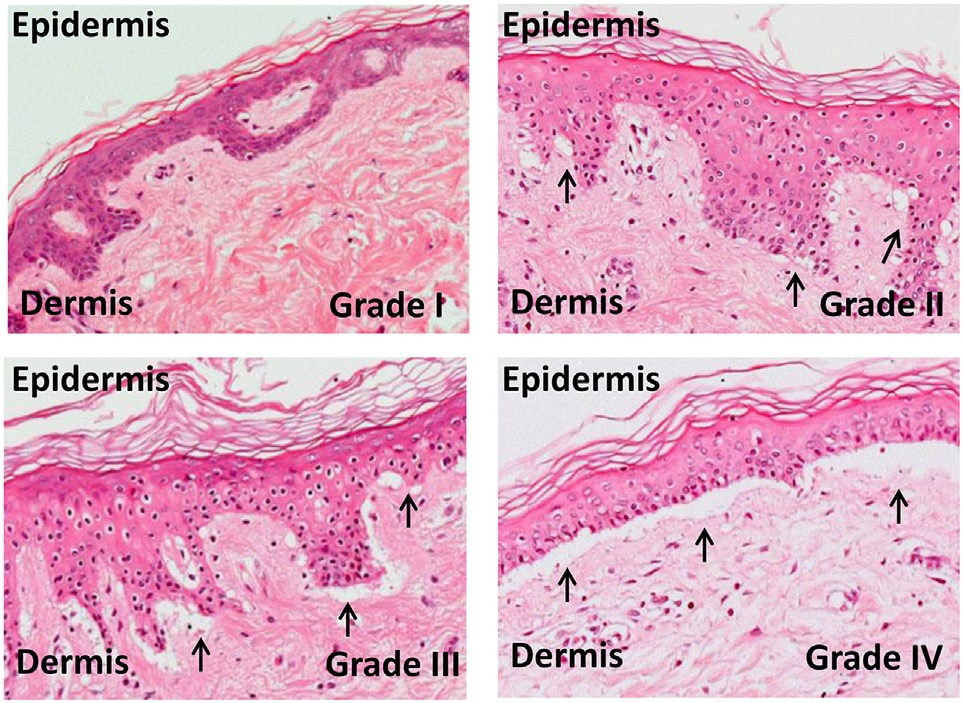
Acute GvHD is staged as follows: overall grade (skin-liver-gut) with each organ staged individually from a low of 1 to a high of 4. Patients with grade IV GvHD usually have a poor prognosis. If the GvHD is severe and requires intense immunosuppression involving steroids and additional agents to get under control, the patient may develop severe infections as a result of the immunosuppression and may die of infection. However, a 2016 study found that the prognosis for patients with grade IV GvHD has improved in recent years.
Chronic
The ''chronic'' form of graft-versus-host disease (cGvHD) normally begins 90 to 600 days post-transplant. The appearance of moderate to severe cases of cGVHD adversely influences long-term survival. The first symptom of cGvHD is commonly a rash on the palms of the hands or the soles of the feet, and the rash can spread and is usually itchy and dry. In severe cases, the skin may blister and peel, like a bad sunburn. A fever may also develop. Other symptoms of chronic GVHD can include: * Decreased appetite * Diarrhea * Abdominal (belly) cramps * Weight loss * Yellowing of the skin and eyes (jaundice) * Enlarged liver * Bloated abdomen (belly) * Pain in the upper right part of the abdomen (belly) * Increased levels of liver enzymes in the blood (seen on blood tests) * Skin that feels tight * Dry, burning eyes * Dryness or painful sores in the mouth * Burning sensations when eating acidic foods * Bacterial infections * Blockages in the smaller airways of the lungs In the oral cavity, chronic graft-versus-host disease manifests as lichen planus with a higher risk of malignant transformation to oralsquamous cell carcinoma
Squamous-cell carcinomas (SCCs), also known as epidermoid carcinomas, comprise a number of different types of cancer that begin in squamous cells. These cells form on the surface of the skin, on the lining of hollow organs in the body, and on the ...
in comparison to the classical oral lichen planus. Oral cancer associated with graft-versus-host disease may have more aggressive behavior with poorer prognosis, when compared to oral cancer in non-hematopoietic stem cell transplantation patients.
Causes
 Three criteria, known as the Billingham criteria, must be met in order for GvHD to occur.
* An immuno-competent graft is administered, with viable and functional immune cells.
* The recipient is immunologically different from the donor – histo-incompatible.
* The recipient is immunocompromised and therefore cannot destroy or inactivate the transplanted cells. In particular, it involves an inability of the recipient's
Three criteria, known as the Billingham criteria, must be met in order for GvHD to occur.
* An immuno-competent graft is administered, with viable and functional immune cells.
* The recipient is immunologically different from the donor – histo-incompatible.
* The recipient is immunocompromised and therefore cannot destroy or inactivate the transplanted cells. In particular, it involves an inability of the recipient's cell-mediated immunity
Cell-mediated immunity or cellular immunity is an immune response that does not involve antibodies. Rather, cell-mediated immunity is the activation of phagocytes, antigen-specific cytotoxic T-lymphocytes, and the release of various cytokines ...
to destroy or inactivate viable lymphocyte
A lymphocyte is a type of white blood cell (leukocyte) in the immune system of most vertebrates. Lymphocytes include natural killer cells (which function in cell-mediated, cytotoxic innate immunity), T cells (for cell-mediated, cytotoxic a ...
s from the donor.
After bone marrow transplantation, T cell
A T cell is a type of lymphocyte. T cells are one of the important white blood cells of the immune system and play a central role in the adaptive immune response. T cells can be distinguished from other lymphocytes by the presence of a T-cell r ...
s present in the graft, either as contaminants or intentionally introduced into the host, attack the tissues of the transplant recipient after perceiving host tissues as antigenically foreign. The T cells produce an excess of cytokine
Cytokines are a broad and loose category of small proteins (~5–25 kDa) important in cell signaling. Cytokines are peptides and cannot cross the lipid bilayer of cells to enter the cytoplasm. Cytokines have been shown to be involved in au ...
s, including TNF-α and interferon-gamma
Interferon gamma (IFN-γ) is a dimerized soluble cytokine that is the only member of the type II class of interferons. The existence of this interferon, which early in its history was known as immune interferon, was described by E. F. Wheelock ...
(IFNγ). A wide range of host antigens
In immunology, an antigen (Ag) is a molecule or molecular structure or any foreign particulate matter or a pollen grain that can bind to a specific antibody or T-cell receptor. The presence of antigens in the body may trigger an immune response. ...
can initiate graft-versus-host disease, among them the human leukocyte antigens
The human leukocyte antigen (HLA) system or complex is a complex of genes on chromosome 6 in humans which encode cell-surface proteins responsible for the regulation of the immune system. The HLA system is also known as the human version of th ...
(HLA). However, graft-versus-host disease can occur even when HLA-identical siblings are the donors. HLA-identical siblings or HLA-identical unrelated donors often have genetically different protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, res ...
s (called minor histocompatibility antigens) that can be presented by major histocompatibility complex
The major histocompatibility complex (MHC) is a large locus on vertebrate DNA containing a set of closely linked polymorphic genes that code for cell surface proteins essential for the adaptive immune system. These cell surface proteins are cal ...
(MHC) molecules to the donor's T-cells, which see these antigens as foreign and so mount an immune response.
Antigens most responsible for graft loss are HLA-DR
HLA-DR is an MHC class II cell surface receptor encoded by the human leukocyte antigen complex on chromosome 6 region 6p21.31. The complex of HLA-DR (Human Leukocyte Antigen – DR isotype) and peptide, generally between 9 and 30 amino acids in ...
(first six months), HLA-B
HLA-B (major histocompatibility complex, class I, B) is a human gene that provides instructions for making a protein that plays a critical role in the immune system. HLA-B is part of a family of genes called the human leukocyte antigen (HLA) c ...
(first two years), and HLA-A
HLA-A is a group of human leukocyte antigens (HLA) that are encoded by the HLA-A locus, which is located at human chromosome 6p21.3. HLA is a major histocompatibility complex (MHC) antigen specific to humans. HLA-A is one of three major types of ...
(long-term survival).
While donor T-cells are undesirable as effector cells of graft-versus-host disease, they are valuable for engraftment by preventing the recipient's residual immune system
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splinte ...
from rejecting the bone marrow graft ( host-versus-graft). In addition, as bone marrow transplantation is frequently used to treat cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
, mainly leukemia
Leukemia ( also spelled leukaemia and pronounced ) is a group of blood cancers that usually begin in the bone marrow and result in high numbers of abnormal blood cells. These blood cells are not fully developed and are called ''blasts'' or ...
s, donor T-cells have proven to have a valuable graft-versus-tumor
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
effect. A great deal of current research on allogeneic bone marrow transplantation involves attempts to separate the undesirable graft-vs-host disease aspects of T-cell physiology from the desirable graft-versus-tumor effect.
Transfusion-associated GvHD
This type of GvHD is associated with transfusion of un-irradiated blood to immunocompromised recipients. It can also occur in situations in which the blood donor ishomozygous
Zygosity (the noun, zygote, is from the Greek "yoked," from "yoke") () is the degree to which both copies of a chromosome or gene have the same genetic sequence. In other words, it is the degree of similarity of the alleles in an organism.
Mo ...
and the recipient is heterozygous
Zygosity (the noun, zygote, is from the Greek "yoked," from "yoke") () is the degree to which both copies of a chromosome or gene have the same genetic sequence. In other words, it is the degree of similarity of the alleles in an organism.
...
for an HLA haplotype
A haplotype ( haploid genotype) is a group of alleles in an organism that are inherited together from a single parent.
Many organisms contain genetic material ( DNA) which is inherited from two parents. Normally these organisms have their DNA o ...
. It is associated with higher mortality (80–90%) due to involvement of bone marrow lymphoid tissue, however the clinical manifestations are similar to GVHD resulting from bone marrow transplantation. Transfusion-associated GvHD is rare in modern medicine. It is almost entirely preventable by controlled irradiation of blood products to inactivate the white blood cells (including lymphocytes) within.
Thymus transplantation
Thymus transplantation may be said to be able to cause a special type of GvHD because the recipient's thymocytes would use the donor thymus cells as models when going through the negative selection to recognize self-antigens, and could therefore still mistake own structures in the rest of the body for being non-self. This is a rather indirect GvHD because it is not directly cells in the graft itself that causes it but cells in the graft that make the recipient's T cells act like donor T cells. It can be seen as a multiple-organ autoimmunity in xenotransplantation experiments of the thymus between different species. Autoimmune disease is a frequent complication after human allogeneic thymus transplantation, found in 42% of subjects over 1 year post transplantation. However, this is partially explained by the fact that the indication itself, that is, complete DiGeorge syndrome, increases the risk of autoimmune disease.Thymoma-associated multiorgan autoimmunity (TAMA)
A GvHD-like disease calledthymoma-associated multiorgan autoimmunity
Thymoma-associated multiorgan autoimmunity (TAMA) is a severe often fatal disease that presents in some patients with thymoma. It has also been referred to in the medical literature as "thymoma-associated graft-versus-host-like disease".
Presen ...
(TAMA) can occur in patients with thymoma. In these patients rather than a donor being a source of pathogenic T cells, the patient's own malignant thymus produces self-directed T cells. This is because the malignant thymus is incapable of appropriately educating developing thymocytes to eliminate self-reactive T cells. The result is a disease virtually indistinguishable from GvHD.
Mechanism
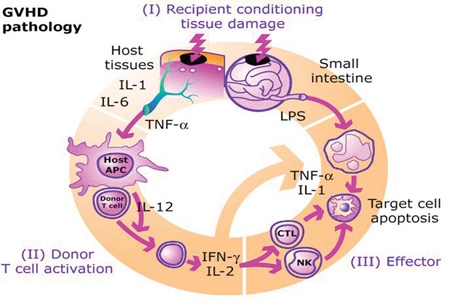
The pathophysiology of GvHD includes three phases: # The afferent phase: activation of APC ( antigen presenting cells) # The efferent phase: activation, proliferation, differentiation and migration of effector cells # The effector phase: target tissue destruction Activation of APC occurs in the first stage of GvHD. Prior to haematopoietic stem cell transplantation, radiation or chemotherapy results in damage and activation of host tissues, especially intestinal mucosa. This allows the microbial products to enter and stimulate pro-inflammatory cytokines such as IL-1 and TNF-α. These proinflammatory cytokines increase the expression of MHC and adhesion molecules on APCs, thereby increasing the ability of APC to present antigen. The second phase is characterized by the activation of effector cells. Activation of donor T-cells further enhances the expression of MHC and adhesion molecules, chemokines and the expansion of CD8 + and CD4 + T-cells and guest B-cells. In the final phase, these effector cells migrate to target organs and mediate tissue damage, resulting in multiorgan failure.Prevention
* DNA-based tissue typing allows for more precise HLA matching between donors and transplant patients, which has been proven to reduce the incidence and severity of GvHD and to increase long-term survival. * The T-cells of umbilical cord blood (UCB) have an inherent immunological immaturity, and the use of UCB stem cells in unrelated donor transplants has a reduced incidence and severity of GvHD. *Methotrexate
Methotrexate (MTX), formerly known as amethopterin, is a chemotherapy agent and immune-system suppressant. It is used to treat cancer, autoimmune diseases, and ectopic pregnancies. Types of cancers it is used for include breast cancer, leuke ...
, cyclosporin and tacrolimus are common drugs used for GvHD prophylaxis. Further research is necessary to evaluate whether mesenchymal stromal cells can also be used for the prophylaxis.
* Graft-versus-host disease can largely be avoided by performing a T-cell-depleted bone marrow transplant. However, these types of transplants come at a cost of diminished graft-versus-tumor effect, greater risk of engraftment failure, or cancer relapse, and general immunodeficiency
Immunodeficiency, also known as immunocompromisation, is a state in which the immune system's ability to fight infectious diseases and cancer is compromised or entirely absent. Most cases are acquired ("secondary") due to extrinsic factors that a ...
, resulting in a patient more susceptible to viral, bacterial
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were amon ...
, and fungal infection
An infection is the invasion of tissues by pathogens, their multiplication, and the reaction of host tissues to the infectious agent and the toxins they produce. An infectious disease, also known as a transmissible disease or communicable di ...
. In a multi-center study, disease-free survival at 3 years was not different between T cell-depleted and T cell-replete transplants.
Treatment
Intravenously administered glucocorticoids, such as prednisone, are the standard of care in acute GvHD and chronic GVHD. The use of these glucocorticoids is designed to suppress the T-cell-mediated immune onslaught on the host tissues; however, in high doses, this immune-suppression raises the risk of infections and cancer relapse. Therefore, it is desirable to taper off the post-transplant high-level steroid doses to lower levels, at which point the appearance of mild GVHD may be welcome, especially in HLA mis-matched patients, as it is typically associated with a graft-versus-tumor effect.. Cyclosporine and tacrolimus are calcineurin inhibitors. The substances are structurally different but have the same mechanism of action. Cyclosporine binds to the cytosolic protein Peptidyl-prolyl cis-trans isomerase A (known as cyclophilin), while tacrolimus binds to the cytosolic protein Peptidyl-prolyl cis-trans isomerase FKBP12. These complexes inhibit calcineurin, block dephosphorylation of the transcription factor NFAT of activated T-cells and its translocation into the nucleus. Standard prophylaxis involves the use of cyclosporine for six months with methotrexate. Cyclosporin levels should be maintained above 200 ng/ml. Other substances that have been studied for GvHD treatment include, for example: sirolimus,pentostatin
Pentostatin (or deoxycoformycin, trade name Nipent, manufactured by SuperGen) is an anticancer chemotherapeutic drug.
Mechanism
It is classified as a purine analog, which is a type of antimetabolite.
It mimics the nucleoside adenosine and thus i ...
, etanercept, and alemtuzumab.
In August 2017 the US FDA approved ibrutinib to treat chronic GvHD after failure of one or more other systemic treatments.
Clinical research
There are a large number of clinical trials either ongoing or recently completed in the investigation of graft-versus-host disease treatment and prevention. On May 17, 2012, Osiris Therapeutics announced that Canadian health regulators approvedProchymal
Prochymal is a stem cell therapy made by Osiris Therapeutics. It is the first stem cell therapy approved by Canada. It is also the first therapy approved by Canada for acute graft-vs-host disease (GvHD). Also known as remestemcel-L, Prochymal was ...
, its drug for acute graft-versus-host disease in children who have failed to respond to steroid treatment. Prochymal is the first stem cell drug to be approved for a systemic disease.
In January 2016, Mesoblast
Mesoblast Limited is an Australian-based regenerative medicine company. It seeks to provide treatments for inflammatory ailments, cardiovascular disease and back pain. The company is led by Silviu Itescu, who founded the company in 2004.
Mese ...
released results of a phase 2 clinical trial on 241 children with acute Graft-versus-host disease, that was not responsive to steroids. The trial was of a mesenchymal stem cell therapy known as remestemcel-L or MSC-100-IV. Survival rate was 82% (vs 39% of controls) for those who showed some improvement after 1 month, and in the long term 72% (vs 18% of controls) for those that showed little effect after 1 month.
HIV elimination
Graft-versus-host disease has been implicated in eliminating several cases of HIV, including The Berlin Patient and 6 others in Spain.See also
* Graft-versus-tumor effect * Immunosuppression *Transplant rejection
Transplant rejection occurs when transplanted tissue is rejected by the recipient's immune system, which destroys the transplanted tissue. Transplant rejection can be lessened by determining the molecular similitude between donor and recipient ...
References
Further reading
* Ferrara JLM, Deeg HJ, Burakoff SJ. ''Graft-Vs.-Host Disease: Immunology, Pathophysiology, and Treatment.'' Marcel Dekker, 1990 * Polsdorfer, JR ''Gale Encyclopedia of Medicine: Graft-vs.-host disease''External links
{{DEFAULTSORT:Graft-Versus-Host Disease Immune system disorders Transplantation medicine Noninfectious immunodeficiency-related cutaneous conditions Complications of surgical and medical care