Electromagnetic waves on:
[Wikipedia]
[Google]
[Amazon]
 In
In

 Maxwell's equations established that some charges and currents ("sources") produce a local type of electromagnetic field near them that does ''not'' have the behaviour of EMR. Currents directly produce a magnetic field, but it is of a
Maxwell's equations established that some charges and currents ("sources") produce a local type of electromagnetic field near them that does ''not'' have the behaviour of EMR. Currents directly produce a magnetic field, but it is of a

 In homogeneous, isotropic media, electromagnetic radiation is a
In homogeneous, isotropic media, electromagnetic radiation is a
 In 1862–64
In 1862–64
The Growth of Physical Science
Cambridge University Press Wilhelm Röntgen discovered and named

 EM radiation (the designation 'radiation' excludes static electric and magnetic and near fields) is classified by wavelength into
EM radiation (the designation 'radiation' excludes static electric and magnetic and near fields) is classified by wavelength into
 Most UV and X-rays are blocked by absorption first from molecular
Most UV and X-rays are blocked by absorption first from molecular
The Feynman Lectures on Physics Vol. I Ch. 28: Electromagnetic Radiation
*
''Electromagnetic Waves from Maxwell's Equations''
o
Project PHYSNET
"Electromagnetic radiation"
in the ''
physics
Physics is the natural science that studies matter, its fundamental constituents, its motion and behavior through space and time, and the related entities of energy and force. "Physical science is that department of knowledge which r ...
, electromagnetic radiation (EMR) consists of wave
In physics, mathematics, and related fields, a wave is a propagating dynamic disturbance (change from equilibrium) of one or more quantities. Waves can be periodic, in which case those quantities oscillate repeatedly about an equilibrium (re ...
s of the electromagnetic (EM) field, which propagate through space
Space is the boundless three-dimensional extent in which objects and events have relative position and direction. In classical physics, physical space is often conceived in three linear dimensions, although modern physicists usually cons ...
and carry momentum and electromagnetic radiant energy
Radiant may refer to:
Computers, software, and video games
* Radiant (software), a content management system
* GtkRadiant, a level editor created by id Software for their games
* Radiant AI, a technology developed by Bethesda Softworks for '' ...
. It includes radio waves, microwave
Microwave is a form of electromagnetic radiation with wavelengths ranging from about one meter to one millimeter corresponding to frequencies between 300 MHz and 300 GHz respectively. Different sources define different frequency ra ...
s, infrared
Infrared (IR), sometimes called infrared light, is electromagnetic radiation (EMR) with wavelengths longer than those of visible light. It is therefore invisible to the human eye. IR is generally understood to encompass wavelengths from around ...
, (visible) light, ultraviolet
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation ...
, X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
s, and gamma ray
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
s. All of these waves form part of the electromagnetic spectrum
The electromagnetic spectrum is the range of frequencies (the spectrum) of electromagnetic radiation and their respective wavelengths and photon energies.
The electromagnetic spectrum covers electromagnetic waves with frequencies ranging fro ...
.
Classically, electromagnetic radiation consists of electromagnetic waves, which are synchronized oscillation
Oscillation is the repetitive or periodic variation, typically in time, of some measure about a central value (often a point of equilibrium) or between two or more different states. Familiar examples of oscillation include a swinging pendulum ...
s of electric
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described by ...
and magnetic fields. Depending on the frequency of oscillation, different wavelengths of electromagnetic spectrum are produced. In a vacuum, electromagnetic waves travel at the speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
, commonly denoted ''c''. In homogeneous, isotropic media, the oscillations of the two fields are perpendicular to each other and perpendicular to the direction of energy and wave propagation, forming a transverse wave
In physics, a transverse wave is a wave whose oscillations are perpendicular to the direction of the wave's advance. This is in contrast to a longitudinal wave which travels in the direction of its oscillations. Water waves are an example of t ...
. The position of an electromagnetic wave within the electromagnetic spectrum
The electromagnetic spectrum is the range of frequencies (the spectrum) of electromagnetic radiation and their respective wavelengths and photon energies.
The electromagnetic spectrum covers electromagnetic waves with frequencies ranging fro ...
can be characterized by either its frequency
Frequency is the number of occurrences of a repeating event per unit of time. It is also occasionally referred to as ''temporal frequency'' for clarity, and is distinct from ''angular frequency''. Frequency is measured in hertz (Hz) which is eq ...
of oscillation or its wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, t ...
. Electromagnetic waves of different frequency are called by different names since they have different sources and effects on matter. In order of increasing frequency and decreasing wavelength these are: radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays and gamma rays.
Electromagnetic waves are emitted by electrically charged particle
In physics, a charged particle is a particle with an electric charge. It may be an ion, such as a molecule or atom with a surplus or deficit of electrons relative to protons. It can also be an electron or a proton, or another elementary pa ...
s undergoing acceleration, and these waves can subsequently interact with other charged particles, exerting force on them. EM waves carry energy, momentum and angular momentum
In physics, angular momentum (rarely, moment of momentum or rotational momentum) is the rotational analog of linear momentum. It is an important physical quantity because it is a conserved quantity—the total angular momentum of a closed syst ...
away from their source particle and can impart those quantities to matter
In classical physics and general chemistry, matter is any substance that has mass and takes up space by having volume. All everyday objects that can be touched are ultimately composed of atoms, which are made up of interacting subatomic part ...
with which they interact. Electromagnetic radiation is associated with those EM waves that are free to propagate themselves ("radiate") without the continuing influence of the moving charges that produced them, because they have achieved sufficient distance from those charges. Thus, EMR is sometimes referred to as the far field
The near field and far field are regions of the electromagnetic (EM) field around an object, such as a transmitting antenna, or the result of radiation scattering off an object. Non-radiative ''near-field'' behaviors dominate close to the ante ...
. In this language, the near field refers to EM fields near the charges and current that directly produced them, specifically electromagnetic induction and electrostatic induction
Electrostatic induction, also known as "electrostatic influence" or simply "influence" in Europe and Latin America, is a redistribution of electric charge in an object that is caused by the influence of nearby charges. In the presence of a charg ...
phenomena.
In quantum mechanics
Quantum mechanics is a fundamental theory in physics that provides a description of the physical properties of nature at the scale of atoms and subatomic particles. It is the foundation of all quantum physics including quantum chemistr ...
, an alternate way of viewing EMR is that it consists of photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
s, uncharged elementary particle
In particle physics, an elementary particle or fundamental particle is a subatomic particle that is not composed of other particles. Particles currently thought to be elementary include electrons, the fundamental fermions ( quarks, leptons, a ...
s with zero rest mass
The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. More precisely, i ...
which are the quanta of the electromagnetic field, responsible for all electromagnetic interactions. Quantum electrodynamics
In particle physics, quantum electrodynamics (QED) is the relativistic quantum field theory of electrodynamics. In essence, it describes how light and matter interact and is the first theory where full agreement between quantum mechanics and spec ...
is the theory of how EMR interacts with matter on an atomic level. Quantum effects provide additional sources of EMR, such as the transition of electrons to lower energy level
A quantum mechanical system or particle that is bound—that is, confined spatially—can only take on certain discrete values of energy, called energy levels. This contrasts with classical particles, which can have any amount of energy. The t ...
s in an atom and black-body radiation. The energy of an individual photon is quantized and is greater for photons of higher frequency. This relationship is given by Planck's equation ''E'' = ''hf'', where ''E'' is the energy per photon, ''f'' is the frequency of the photon, and ''h'' is Planck's constant. A single gamma ray photon, for example, might carry ~100,000 times the energy of a single photon of visible light.
The effects of EMR upon chemical compounds and biological organisms depend both upon the radiation's power
Power most often refers to:
* Power (physics), meaning "rate of doing work"
** Engine power, the power put out by an engine
** Electric power
* Power (social and political), the ability to influence people or events
** Abusive power
Power may a ...
and its frequency. EMR of visible or lower frequencies (i.e., visible light, infrared, microwaves, and radio waves) is called ''non-ionizing radiation
Non-ionizing (or non-ionising) radiation refers to any type of electromagnetic radiation that does not carry enough energy per quantum (photon energy) to ionize atoms or molecules—that is, to completely remove an electron from an atom or mole ...
'', because its photons do not individually have enough energy to ionize
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule ...
atoms or molecules, or break chemical bond
A chemical bond is a lasting attraction between atoms or ions that enables the formation of molecules and crystals. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds, or through the sharing of ...
s. The effects of these radiations on chemical systems and living tissue are caused primarily by heating effects from the combined energy transfer of many photons. In contrast, high frequency ultraviolet, X-rays and gamma rays are called '' ionizing radiation'', since individual photons of such high frequency have enough energy to ionize
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule ...
molecules or break chemical bond
A chemical bond is a lasting attraction between atoms or ions that enables the formation of molecules and crystals. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds, or through the sharing of ...
s. These radiations have the ability to cause chemical reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the pos ...
s and damage living cells beyond that resulting from simple heating, and can be a health hazard.
Physics
Theory
Maxwell's equations
James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish mathematician and scientist responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism and li ...
derived a wave form of the electric and magnetic equations, thus uncovering the wave-like nature of electric
Electricity is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described by ...
and magnetic fields
A magnetic field is a vector field that describes the magnetic influence on moving electric charges, electric currents, and magnetic materials. A moving charge in a magnetic field experiences a force perpendicular to its own velocity and to ...
and their symmetry. Because the speed of EM waves predicted by the wave equation coincided with the measured speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
, Maxwell concluded that light itself is an EM wave. Maxwell's equations were confirmed by Heinrich Hertz
Heinrich Rudolf Hertz ( ; ; 22 February 1857 – 1 January 1894) was a German physicist who first conclusively proved the existence of the electromagnetic waves predicted by James Clerk Maxwell's equations of electromagnetism. The unit ...
through experiments with radio waves.
Maxwell realized that since a lot of physics is symmetrical and mathematically artistic in a way, that there must also be a symmetry between electricity and magnetism. He realized that light is a combination of electricity and magnetism and thus that the two must be tied together. According to Maxwell's equations
Maxwell's equations, or Maxwell–Heaviside equations, are a set of coupled partial differential equations that, together with the Lorentz force law, form the foundation of classical electromagnetism, classical optics, and electric circuits.
...
, a spatially varying electric field is always associated with a magnetic field that changes over time. Likewise, a spatially varying magnetic field is associated with specific changes over time in the electric field. In an electromagnetic wave, the changes in the electric field are always accompanied by a wave in the magnetic field in one direction, and vice versa. This relationship between the two occurs without either type of field causing the other; rather, they occur together in the same way that time and space changes occur together and are interlinked in special relativity
In physics, the special theory of relativity, or special relativity for short, is a scientific theory regarding the relationship between space and time. In Albert Einstein's original treatment, the theory is based on two postulates:
# The laws ...
. In fact, magnetic fields can be viewed as electric fields in another frame of reference, and electric fields can be viewed as magnetic fields in another frame of reference, but they have equal significance as physics is the same in all frames of reference, so the close relationship between space and time changes here is more than an analogy. Together, these fields form a propagating electromagnetic wave, which moves out into space and need never again interact with the source. The distant EM field formed in this way by the acceleration of a charge carries energy with it that "radiates" away through space, hence the term.
Near and far fields
magnetic dipole
In electromagnetism, a magnetic dipole is the limit of either a closed loop of electric current or a pair of poles as the size of the source is reduced to zero while keeping the magnetic moment constant. It is a magnetic analogue of the electric ...
type that dies out with distance from the current. In a similar manner, moving charges pushed apart in a conductor by a changing electrical potential (such as in an antenna) produce an electric dipole
The electric dipole moment is a measure of the separation of positive and negative electrical charges within a system, that is, a measure of the system's overall polarity. The SI unit for electric dipole moment is the coulomb- meter (C⋅m). T ...
type electrical field, but this also declines with distance. These fields make up the near-field near the EMR source. Neither of these behaviours are responsible for EM radiation. Instead, they cause electromagnetic field behaviour that only efficiently transfers power to a receiver very close to the source, such as the magnetic induction inside a transformer
A transformer is a passive component that transfers electrical energy from one electrical circuit to another circuit, or multiple circuits. A varying current in any coil of the transformer produces a varying magnetic flux in the transformer' ...
, or the feedback behaviour that happens close to the coil of a metal detector
A metal detector is an instrument that detects the nearby presence of metal. Metal detectors are useful for finding metal objects on the surface, underground, and under water. The unit itself, consist of a control box, and an adjustable shaft, ...
. Typically, near-fields have a powerful effect on their own sources, causing an increased "load" (decreased electrical reactance
In electrical circuits, reactance is the opposition presented to alternating current by inductance or capacitance. Greater reactance gives smaller current for the same applied voltage. Reactance is similar to resistance in this respect, but does ...
) in the source or transmitter, whenever energy is withdrawn from the EM field by a receiver. Otherwise, these fields do not "propagate" freely out into space, carrying their energy away without distance-limit, but rather oscillate, returning their energy to the transmitter if it is not received by a receiver.
By contrast, the EM far-field is composed of ''radiation'' that is free of the transmitter in the sense that (unlike the case in an electrical transformer) the transmitter requires the same power to send these changes in the fields out, whether the signal is immediately picked up or not. This distant part of the electromagnetic field ''is'' "electromagnetic radiation" (also called the far-field
The near field and far field are regions of the electromagnetic (EM) field around an object, such as a transmitting antenna, or the result of radiation scattering off an object. Non-radiative ''near-field'' behaviors dominate close to the ante ...
). The far-fields propagate (radiate) without allowing the transmitter to affect them. This causes them to be independent in the sense that their existence and their energy, after they have left the transmitter, is completely independent of both transmitter and receiver. Due to conservation of energy, the amount of power passing through any spherical surface drawn around the source is the same. Because such a surface has an area proportional to the square of its distance from the source, the power density
Power density is the amount of power (time rate of energy transfer) per unit volume.
In energy transformers including batteries, fuel cells, motors, power supply units etc., power density refers to a volume, where it is often called volum ...
of EM radiation always decreases with the inverse square of the distance from the source; this is called the inverse-square law. This is in contrast to dipole parts of the EM field close to the source (the near-field), which vary in power according to an inverse cube power law, and thus do ''not'' transport a conserved amount of energy over distances, but instead fade with distance, with its energy (as noted) rapidly returning to the transmitter or absorbed by a nearby receiver (such as a transformer secondary coil).
The far-field (EMR) depends on a different mechanism for its production than the near-field, and upon different terms in Maxwell's equations. Whereas the magnetic part of the near-field is due to currents in the source, the magnetic field in EMR is due only to the local change in the electric field. In a similar way, while the electric field in the near-field is due directly to the charges and charge-separation in the source, the electric field in EMR is due to a change in the local magnetic field. Both processes for producing electric and magnetic EMR fields have a different dependence on distance than do near-field dipole electric and magnetic fields. That is why the EMR type of EM field becomes dominant in power "far" from sources. The term "far from sources" refers to how far from the source (moving at the speed of light) any portion of the outward-moving EM field is located, by the time that source currents are changed by the varying source potential, and the source has therefore begun to generate an outwardly moving EM field of a different phase.
A more compact view of EMR is that the far-field that composes EMR is generally that part of the EM field that has traveled sufficient distance from the source, that it has become completely disconnected from any feedback to the charges and currents that were originally responsible for it. Now independent of the source charges, the EM field, as it moves farther away, is dependent only upon the accelerations of the charges that produced it. It no longer has a strong connection to the direct fields of the charges, or to the velocity of the charges (currents).
In the Liénard–Wiechert potential formulation of the electric and magnetic fields due to motion of a single particle (according to Maxwell's equations), the terms associated with acceleration of the particle are those that are responsible for the part of the field that is regarded as electromagnetic radiation. By contrast, the term associated with the changing static electric field of the particle and the magnetic term that results from the particle's uniform velocity, are both associated with the electromagnetic near-field, and do not comprise EM radiation.
Properties

Electrodynamics
In physics, electromagnetism is an interaction that occurs between particles with electric charge. It is the second-strongest of the four fundamental interactions, after the strong force, and it is the dominant force in the interactions o ...
is the physics
Physics is the natural science that studies matter, its fundamental constituents, its motion and behavior through space and time, and the related entities of energy and force. "Physical science is that department of knowledge which r ...
of electromagnetic radiation, and electromagnetism
In physics, electromagnetism is an interaction that occurs between particles with electric charge. It is the second-strongest of the four fundamental interactions, after the strong force, and it is the dominant force in the interactions of ...
is the physical phenomenon associated with the theory of electrodynamics. Electric and magnetic fields obey the properties of superposition. Thus, a field due to any particular particle or time-varying electric or magnetic field contributes to the fields present in the same space due to other causes. Further, as they are vector
Vector most often refers to:
*Euclidean vector, a quantity with a magnitude and a direction
*Vector (epidemiology), an agent that carries and transmits an infectious pathogen into another living organism
Vector may also refer to:
Mathematic ...
fields, all magnetic and electric field vectors add together according to vector addition
In mathematics, physics, and engineering, a Euclidean vector or simply a vector (sometimes called a geometric vector or spatial vector) is a geometric object that has magnitude (or length) and direction. Vectors can be added to other vectors a ...
. For example, in optics two or more coherent light waves may interact and by constructive or destructive interference
Interference is the act of interfering, invading, or poaching. Interference may also refer to:
Communications
* Interference (communication), anything which alters, modifies, or disrupts a message
* Adjacent-channel interference, caused by extr ...
yield a resultant irradiance deviating from the sum of the component irradiances of the individual light waves.
The electromagnetic fields of light are not affected by traveling through static electric or magnetic fields in a linear medium such as a vacuum. However, in nonlinear media, such as some crystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macro ...
s, interactions can occur between light and static electric and magnetic fields—these interactions include the Faraday effect
The Faraday effect or Faraday rotation, sometimes referred to as the magneto-optic Faraday effect (MOFE), is a physical magneto-optical phenomenon. The Faraday effect causes a polarization rotation which is proportional to the projection of the ...
and the Kerr effect.
In refraction
In physics, refraction is the redirection of a wave as it passes from one medium to another. The redirection can be caused by the wave's change in speed or by a change in the medium. Refraction of light is the most commonly observed phenome ...
, a wave crossing from one medium to another of different density
Density (volumetric mass density or specific mass) is the substance's mass per unit of volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' can also be used. Mathematical ...
alters its speed and direction upon entering the new medium. The ratio of the refractive indices of the media determines the degree of refraction, and is summarized by Snell's law. Light of composite wavelengths (natural sunlight) disperses into a visible spectrum
A spectrum (plural ''spectra'' or ''spectrums'') is a condition that is not limited to a specific set of values but can vary, without gaps, across a continuum. The word was first used scientifically in optics to describe the rainbow of colors ...
passing through a prism, because of the wavelength-dependent refractive index
In optics, the refractive index (or refraction index) of an optical medium is a dimensionless number that gives the indication of the light bending ability of that medium.
The refractive index determines how much the path of light is bent, or ...
of the prism
Prism usually refers to:
* Prism (optics), a transparent optical component with flat surfaces that refract light
* Prism (geometry), a kind of polyhedron
Prism may also refer to:
Science and mathematics
* Prism (geology), a type of sedimentary ...
material (dispersion
Dispersion may refer to:
Economics and finance
* Dispersion (finance), a measure for the statistical distribution of portfolio returns
* Price dispersion, a variation in prices across sellers of the same item
*Wage dispersion, the amount of variat ...
); that is, each component wave within the composite light is bent a different amount.
EM radiation exhibits both wave properties and particle
In the physical sciences, a particle (or corpuscule in older texts) is a small localized object which can be described by several physical or chemical properties, such as volume, density, or mass.
They vary greatly in size or quantity, from ...
properties at the same time (see wave-particle duality). Both wave and particle characteristics have been confirmed in many experiments. Wave characteristics are more apparent when EM radiation is measured over relatively large timescales and over large distances while particle characteristics are more evident when measuring small timescales and distances. For example, when electromagnetic radiation is absorbed by matter, particle-like properties will be more obvious when the average number of photons in the cube of the relevant wavelength is much smaller than 1. It is not so difficult to experimentally observe non-uniform deposition of energy when light is absorbed, however this alone is not evidence of "particulate" behavior. Rather, it reflects the quantum nature of ''matter''. Demonstrating that the light itself is quantized, not merely its interaction with matter, is a more subtle affair.
Some experiments display both the wave and particle natures of electromagnetic waves, such as the self-interference of a single photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
. When a single photon is sent through an interferometer, it passes through both paths, interfering with itself, as waves do, yet is detected by a photomultiplier A photomultiplier is a device that converts incident photons into an electrical signal.
Kinds of photomultiplier include:
* Photomultiplier tube, a vacuum tube converting incident photons into an electric signal. Photomultiplier tubes (PMTs for sh ...
or other sensitive detector only once.
A quantum theory
Quantum theory may refer to:
Science
*Quantum mechanics, a major field of physics
*Old quantum theory, predating modern quantum mechanics
* Quantum field theory, an area of quantum mechanics that includes:
** Quantum electrodynamics
** Quantum ...
of the interaction between electromagnetic radiation and matter such as electrons is described by the theory of quantum electrodynamics
In particle physics, quantum electrodynamics (QED) is the relativistic quantum field theory of electrodynamics. In essence, it describes how light and matter interact and is the first theory where full agreement between quantum mechanics and spec ...
.
Electromagnetic waves can be polarized, reflected, refracted, diffracted
Diffraction is defined as the interference or bending of waves around the corners of an obstacle or through an aperture into the region of geometrical shadow of the obstacle/aperture. The diffracting object or aperture effectively becomes a s ...
or interfere with each other.
Wave model
 In homogeneous, isotropic media, electromagnetic radiation is a
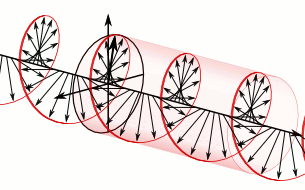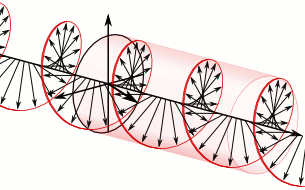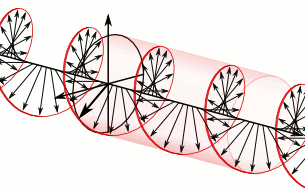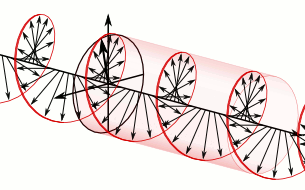
In homogeneous, isotropic media, electromagnetic radiation is a transverse wave
In physics, a transverse wave is a wave whose oscillations are perpendicular to the direction of the wave's advance. This is in contrast to a longitudinal wave which travels in the direction of its oscillations. Water waves are an example of t ...
, meaning that its oscillations are perpendicular to the direction of energy transfer and travel. It comes from the following equations:These equations predicate that any electromagnetic wave must be a transverse wave
In physics, a transverse wave is a wave whose oscillations are perpendicular to the direction of the wave's advance. This is in contrast to a longitudinal wave which travels in the direction of its oscillations. Water waves are an example of t ...
, where the electric field and the magnetic field are both perpendicular to the direction of wave propagation.
The electric and magnetic parts of the field in an electromagnetic wave stand in a fixed ratio of strengths to satisfy the two Maxwell equations
Maxwell's equations, or Maxwell–Heaviside equations, are a set of coupled partial differential equations that, together with the Lorentz force law, form the foundation of classical electromagnetism, classical optics, and electric circuits.
T ...
that specify how one is produced from the other. In dissipation-less (lossless) media, these E and B fields are also in phase, with both reaching maxima and minima at the same points in space (see illustrations). A common misconception is that the E and B fields in electromagnetic radiation are out of phase because a change in one produces the other, and this would produce a phase difference between them as sinusoidal functions (as indeed happens in electromagnetic induction, and in the near-field close to antennas). However, in the far-field EM radiation which is described by the two source-free Maxwell curl operator equations, a more correct description is that a time-change in one type of field is proportional to a space-change in the other. These derivatives require that the E and B fields in EMR are in-phase (see mathematics section below).
An important aspect of light's nature is its frequency
Frequency is the number of occurrences of a repeating event per unit of time. It is also occasionally referred to as ''temporal frequency'' for clarity, and is distinct from ''angular frequency''. Frequency is measured in hertz (Hz) which is eq ...
. The frequency of a wave is its rate of oscillation and is measured in hertz
The hertz (symbol: Hz) is the unit of frequency in the International System of Units (SI), equivalent to one event (or cycle) per second. The hertz is an SI derived unit whose expression in terms of SI base units is s−1, meaning that o ...
, the SI unit of frequency, where one hertz is equal to one oscillation per second. Light usually has multiple frequencies that sum to form the resultant wave. Different frequencies undergo different angles of refraction, a phenomenon known as dispersion
Dispersion may refer to:
Economics and finance
* Dispersion (finance), a measure for the statistical distribution of portfolio returns
* Price dispersion, a variation in prices across sellers of the same item
*Wage dispersion, the amount of variat ...
.
A monochromatic wave (a wave of a single frequency) consists of successive troughs and crests, and the distance between two adjacent crests or troughs is called the wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, t ...
. Waves of the electromagnetic spectrum vary in size, from very long radio waves longer than a continent to very short gamma rays smaller than atom nuclei. Frequency is inversely proportional to wavelength, according to the equation:
:
where ''v'' is the speed of the wave ('' c'' in a vacuum or less in other media), ''f'' is the frequency and λ is the wavelength. As waves cross boundaries between different media, their speeds change but their frequencies remain constant.
Electromagnetic waves in free space must be solutions of Maxwell's electromagnetic wave equation. Two main classes of solutions are known, namely plane waves and spherical waves. The plane waves may be viewed as the limiting case of spherical waves at a very large (ideally infinite) distance from the source. Both types of waves can have a waveform which is an arbitrary time function (so long as it is sufficiently differentiable to conform to the wave equation). As with any time function, this can be decomposed by means of Fourier analysis into its frequency spectrum
The power spectrum S_(f) of a time series x(t) describes the distribution of power into frequency components composing that signal. According to Fourier analysis, any physical signal can be decomposed into a number of discrete frequencies, ...
, or individual sinusoidal components, each of which contains a single frequency, amplitude and phase. Such a component wave is said to be ''monochromatic''. A monochromatic electromagnetic wave can be characterized by its frequency or wavelength, its peak amplitude, its phase relative to some reference phase, its direction of propagation, and its polarization.
Interference is the superposition of two or more waves resulting in a new wave pattern. If the fields have components in the same direction, they constructively interfere, while opposite directions cause destructive interference. An example of interference caused by EMR is electromagnetic interference (EMI) or as it is more commonly known as, radio-frequency interference (RFI). Additionally, multiple polarization signals can be combined (i.e. interfered) to form new states of polarization, which is known as parallel polarization state generation
Parallel is a geometric term of location which may refer to:
Computing
* Parallel algorithm
* Parallel computing
* Parallel metaheuristic
* Parallel (software), a UNIX utility for running programs in parallel
* Parallel Sysplex, a cluster of I ...
.
The energy in electromagnetic waves is sometimes called radiant energy
Radiant may refer to:
Computers, software, and video games
* Radiant (software), a content management system
* GtkRadiant, a level editor created by id Software for their games
* Radiant AI, a technology developed by Bethesda Softworks for '' ...
.
Particle model and quantum theory
An anomaly arose in the late 19th century involving a contradiction between the wave theory of light and measurements of the electromagnetic spectra that were being emitted by thermal radiators known as black bodies. Physicists struggled with this problem unsuccessfully for many years. It later became known as theultraviolet catastrophe
The ultraviolet catastrophe, also called the Rayleigh–Jeans catastrophe, was the prediction of late 19th century/early 20th century classical physics that an ideal black body at thermal equilibrium would emit an unbounded quantity of energy ...
. In 1900, Max Planck
Max Karl Ernst Ludwig Planck (, ; 23 April 1858 – 4 October 1947) was a German theoretical physicist whose discovery of energy quanta won him the Nobel Prize in Physics in 1918.
Planck made many substantial contributions to theoretical p ...
developed a new theory of black-body radiation that explained the observed spectrum. Planck's theory was based on the idea that black bodies emit light (and other electromagnetic radiation) only as discrete bundles or packets of energy. These packets were called quanta. In 1905, Albert Einstein
Albert Einstein ( ; ; 14 March 1879 – 18 April 1955) was a German-born theoretical physicist, widely acknowledged to be one of the greatest and most influential physicists of all time. Einstein is best known for developing the theory ...
proposed that light quanta be regarded as real particles. Later the particle of light was given the name photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
, to correspond with other particles being described around this time, such as the electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
and proton. A photon has an energy, ''E'', proportional to its frequency, ''f'', by
:
where ''h'' is Planck's constant, is the wavelength and ''c'' is the speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
. This is sometimes known as the Planck–Einstein equation. In quantum theory (see first quantization
A first quantization of a physical system is a possibly semiclassical treatment of quantum mechanics, in which particles or physical objects are treated using quantum wave functions but the surrounding environment (for example a potential well o ...
) the energy of the photons is thus directly proportional to the frequency of the EMR wave.
Likewise, the momentum ''p'' of a photon is also proportional to its frequency and inversely proportional to its wavelength:
:
The source of Einstein's proposal that light was composed of particles (or could act as particles in some circumstances) was an experimental anomaly not explained by the wave theory: the photoelectric effect
The photoelectric effect is the emission of electrons when electromagnetic radiation, such as light, hits a material. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, and solid sta ...
, in which light striking a metal surface ejected electrons from the surface, causing an electric current to flow across an applied voltage
Voltage, also known as electric pressure, electric tension, or (electric) potential difference, is the difference in electric potential between two points. In a static electric field, it corresponds to the work needed per unit of charge to ...
. Experimental measurements demonstrated that the energy of individual ejected electrons was proportional to the ''frequency
Frequency is the number of occurrences of a repeating event per unit of time. It is also occasionally referred to as ''temporal frequency'' for clarity, and is distinct from ''angular frequency''. Frequency is measured in hertz (Hz) which is eq ...
'', rather than the '' intensity'', of the light. Furthermore, below a certain minimum frequency, which depended on the particular metal, no current would flow regardless of the intensity. These observations appeared to contradict the wave theory, and for years physicists tried in vain to find an explanation. In 1905, Einstein explained this puzzle by resurrecting the particle theory of light to explain the observed effect. Because of the preponderance of evidence in favor of the wave theory, however, Einstein's ideas were met initially with great skepticism among established physicists. Eventually Einstein's explanation was accepted as new particle-like behavior of light was observed, such as the Compton effect
Compton scattering, discovered by Arthur Holly Compton, is the scattering of a high frequency photon after an interaction with a charged particle, usually an electron. If it results in a decrease in energy (increase in wavelength) of the photon ...
.
As a photon is absorbed by an atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, ...
, it excites the atom, elevating an electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
to a higher energy level
A quantum mechanical system or particle that is bound—that is, confined spatially—can only take on certain discrete values of energy, called energy levels. This contrasts with classical particles, which can have any amount of energy. The t ...
(one that is on average farther from the nucleus). When an electron in an excited molecule or atom descends to a lower energy level, it emits a photon of light at a frequency corresponding to the energy difference. Since the energy levels of electrons in atoms are discrete, each element and each molecule emits and absorbs its own characteristic frequencies. Immediate photon emission is called fluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, tha ...
, a type of photoluminescence. An example is visible light emitted from fluorescent paints, in response to ultraviolet (blacklight
A blacklight, also called a UV-A light, Wood's lamp, or ultraviolet light, is a lamp that emits long-wave (UV-A) ultraviolet light and very little visible light. One type of lamp has a violet filter material, either on the bulb or in a sepa ...
). Many other fluorescent emissions are known in spectral bands other than visible light. Delayed emission is called phosphorescence
Phosphorescence is a type of photoluminescence related to fluorescence. When exposed to light (radiation) of a shorter wavelength, a phosphorescent substance will glow, absorbing the light and reemitting it at a longer wavelength. Unlike fluo ...
.
Wave–particle duality
The modern theory that explains the nature of light includes the notion of wave–particle duality. More generally, the theory states that everything has both a particle nature and a wave nature, and various experiments can be done to bring out one or the other. The particle nature is more easily discerned using an object with a large mass. A bold proposition byLouis de Broglie
Louis Victor Pierre Raymond, 7th Duc de Broglie (, also , or ; 15 August 1892 – 19 March 1987) was a French physicist and aristocrat who made groundbreaking contributions to Old quantum theory, quantum theory. In his 1924 PhD thesis, he pos ...
in 1924 led the scientific community to realize that matter (e.g. electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
s) also exhibits wave–particle duality.
Wave and particle effects of electromagnetic radiation
Together, wave and particle effects fully explain the emission and absorption spectra of EM radiation. The matter-composition of the medium through which the light travels determines the nature of the absorption and emission spectrum. These bands correspond to the allowed energy levels in the atoms. Dark bands in the absorption spectrum are due to the atoms in an intervening medium between source and observer. The atoms absorb certain frequencies of the light between emitter and detector/eye, then emit them in all directions. A dark band appears to the detector, due to the radiation scattered out of the beam. For instance, dark bands in the light emitted by a distant star are due to the atoms in the star's atmosphere. A similar phenomenon occurs for emission, which is seen when an emitting gas glows due to excitation of the atoms from any mechanism, including heat. As electrons descend to lower energy levels, a spectrum is emitted that represents the jumps between the energy levels of the electrons, but lines are seen because again emission happens only at particular energies after excitation. An example is the emission spectrum of nebulae. Rapidly moving electrons are most sharply accelerated when they encounter a region of force, so they are responsible for producing much of the highest frequency electromagnetic radiation observed in nature. These phenomena can aid various chemical determinations for the composition of gases lit from behind (absorption spectra) and for glowing gases (emission spectra). Spectroscopy (for example) determines whatchemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
s comprise a particular star. Spectroscopy is also used in the determination of the distance of a star, using the red shift.
Propagation speed
When any wire (or other conducting object such as an antenna) conductsalternating current
Alternating current (AC) is an electric current which periodically reverses direction and changes its magnitude continuously with time in contrast to direct current (DC) which flows only in one direction. Alternating current is the form in whic ...
, electromagnetic radiation is propagated at the same frequency as the current. In many such situations it is possible to identify an electrical dipole moment that arises from separation of charges due to the exciting electrical potential, and this dipole moment oscillates in time, as the charges move back and forth. This oscillation at a given frequency gives rise to changing electric and magnetic fields, which then set the electromagnetic radiation in motion.
At the quantum level, electromagnetic radiation is produced when the wavepacket of a charged particle oscillates or otherwise accelerates. Charged particles in a stationary state
A stationary state is a quantum state with all observables independent of time. It is an eigenvector of the energy operator (instead of a quantum superposition of different energies). It is also called energy eigenvector, energy eigenstate, ener ...
do not move, but a superposition of such states may result in a transition state that has an electric dipole moment that oscillates in time. This oscillating dipole moment is responsible for the phenomenon of radiative transition between quantum states of a charged particle. Such states occur (for example) in atoms when photons are radiated as the atom shifts from one stationary state to another.
As a wave, light is characterized by a velocity (the speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
), wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, t ...
, and frequency
Frequency is the number of occurrences of a repeating event per unit of time. It is also occasionally referred to as ''temporal frequency'' for clarity, and is distinct from ''angular frequency''. Frequency is measured in hertz (Hz) which is eq ...
. As particles, light is a stream of photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they a ...
s. Each has an energy related to the frequency of the wave given by Planck's relation ''E = hf'', where ''E'' is the energy of the photon, ''h'' is Planck's constant, 6.626 × 10−34 J·s, and ''f'' is the frequency of the wave.
One rule is obeyed regardless of circumstances: EM radiation in a vacuum travels at the speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
, ''relative to the observer'', regardless of the observer's velocity.
In a medium (other than vacuum), velocity factor or refractive index
In optics, the refractive index (or refraction index) of an optical medium is a dimensionless number that gives the indication of the light bending ability of that medium.
The refractive index determines how much the path of light is bent, or ...
are considered, depending on frequency and application. Both of these are ratios of the speed in a medium to speed in a vacuum.
Special theory of relativity
By the late nineteenth century, various experimental anomalies could not be explained by the simple wave theory. One of these anomalies involved a controversy over the speed of light. The speed of light and other EMR predicted by Maxwell's equations did not appear unless the equations were modified in a way first suggested by FitzGerald and Lorentz (seehistory of special relativity
The history of special relativity consists of many theoretical results and empirical findings obtained by Albert A. Michelson, Hendrik Lorentz, Henri Poincaré and others. It culminated in the theory of special relativity proposed by Albert Eins ...
), or else otherwise that speed would depend on the speed of observer relative to the "medium" (called luminiferous aether
Luminiferous aether or ether ("luminiferous", meaning "light-bearing") was the postulated medium for the propagation of light. It was invoked to explain the ability of the apparently wave-based light to propagate through empty space (a vacuum), so ...
) which supposedly "carried" the electromagnetic wave (in a manner analogous to the way air carries sound waves). Experiments failed to find any observer effect. In 1905, Einstein proposed that space and time appeared to be velocity-changeable entities for light propagation and all other processes and laws. These changes accounted for the constancy of the speed of light and all electromagnetic radiation, from the viewpoints of all observers—even those in relative motion.
History of discovery
Electromagnetic radiation of wavelengths other than those of visible light were discovered in the early 19th century. The discovery ofinfrared
Infrared (IR), sometimes called infrared light, is electromagnetic radiation (EMR) with wavelengths longer than those of visible light. It is therefore invisible to the human eye. IR is generally understood to encompass wavelengths from around ...
radiation is ascribed to astronomer William Herschel
Frederick William Herschel (; german: Friedrich Wilhelm Herschel; 15 November 1738 – 25 August 1822) was a German-born British astronomer and composer. He frequently collaborated with his younger sister and fellow astronomer Caroline ...
, who published his results in 1800 before the Royal Society of London
The Royal Society, formally The Royal Society of London for Improving Natural Knowledge, is a learned society and the United Kingdom's national academy of sciences. The society fulfils a number of roles: promoting science and its benefits, re ...
. Herschel used a glass prism
Prism usually refers to:
* Prism (optics), a transparent optical component with flat surfaces that refract light
* Prism (geometry), a kind of polyhedron
Prism may also refer to:
Science and mathematics
* Prism (geology), a type of sedimentary ...
to refract
In physics, refraction is the redirection of a wave as it passes from one medium to another. The redirection can be caused by the wave's change in speed or by a change in the medium. Refraction of light is the most commonly observed phenomeno ...
light from the Sun
The Sun is the star at the center of the Solar System. It is a nearly perfect ball of hot plasma, heated to incandescence by nuclear fusion reactions in its core. The Sun radiates this energy mainly as light, ultraviolet, and infrared radi ...
and detected invisible rays that caused heating beyond the red part of the spectrum, through an increase in the temperature recorded with a thermometer
A thermometer is a device that measures temperature or a temperature gradient (the degree of hotness or coldness of an object). A thermometer has two important elements: (1) a temperature sensor (e.g. the bulb of a mercury-in-glass thermometer ...
. These "calorific rays" were later termed infrared.
In 1801, German physicist Johann Wilhelm Ritter discovered ultraviolet
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation ...
in an experiment similar to Herschel's, using sunlight and a glass prism. Ritter noted that invisible rays near the violet edge of a solar spectrum dispersed by a triangular prism darkened silver chloride
Silver chloride is a chemical compound with the chemical formula Ag Cl. This white crystalline solid is well known for its low solubility in water (this behavior being reminiscent of the chlorides of Tl+ and Pb2+). Upon illumination or heating, ...
preparations more quickly than did the nearby violet light. Ritter's experiments were an early precursor to what would become photography. Ritter noted that the ultraviolet rays (which at first were called "chemical rays") were capable of causing chemical reactions.
 In 1862–64
In 1862–64 James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish mathematician and scientist responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism and li ...
developed equations for the electromagnetic field which suggested that waves in the field would travel with a speed that was very close to the known speed of light. Maxwell therefore suggested that visible light (as well as invisible infrared and ultraviolet rays by inference) all consisted of propagating disturbances (or radiation) in the electromagnetic field. Radio waves were first produced deliberately by Heinrich Hertz
Heinrich Rudolf Hertz ( ; ; 22 February 1857 – 1 January 1894) was a German physicist who first conclusively proved the existence of the electromagnetic waves predicted by James Clerk Maxwell's equations of electromagnetism. The unit ...
in 1887, using electrical circuits calculated to produce oscillations at a much lower frequency than that of visible light, following recipes for producing oscillating charges and currents suggested by Maxwell's equations. Hertz also developed ways to detect these waves, and produced and characterized what were later termed radio waves and microwave
Microwave is a form of electromagnetic radiation with wavelengths ranging from about one meter to one millimeter corresponding to frequencies between 300 MHz and 300 GHz respectively. Different sources define different frequency ra ...
s. Jeans, James (1947The Growth of Physical Science
Cambridge University Press Wilhelm Röntgen discovered and named
X-rays
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nbs ...
. After experimenting with high voltages applied to an evacuated tube on 8 November 1895, he noticed a fluorescence on a nearby plate of coated glass. In one month, he discovered X-rays' main properties.
The last portion of the EM spectrum to be discovered was associated with radioactivity. Henri Becquerel
Antoine Henri Becquerel (; 15 December 1852 – 25 August 1908) was a French engineer, physicist, Nobel laureate, and the first person to discover evidence of radioactivity. For work in this field he, along with Marie Skłodowska-Curie and Pie ...
found that uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weak ...
salts caused fogging of an unexposed photographic plate through a covering paper in a manner similar to X-rays, and Marie Curie
Marie Salomea Skłodowska–Curie ( , , ; born Maria Salomea Skłodowska, ; 7 November 1867 – 4 July 1934) was a Polish and naturalized-French physicist and chemist who conducted pioneering research on radioactivity. She was the first ...
discovered that only certain elements gave off these rays of energy, soon discovering the intense radiation of radium
Radium is a chemical element with the symbol Ra and atomic number 88. It is the sixth element in group 2 of the periodic table, also known as the alkaline earth metals. Pure radium is silvery-white, but it readily reacts with nitrogen (rathe ...
. The radiation from pitchblende was differentiated into alpha rays (alpha particle
Alpha particles, also called alpha rays or alpha radiation, consist of two protons and two neutrons bound together into a particle identical to a helium-4 nucleus. They are generally produced in the process of alpha decay, but may also be pr ...
s) and beta rays ( beta particles) by Ernest Rutherford
Ernest Rutherford, 1st Baron Rutherford of Nelson, (30 August 1871 – 19 October 1937) was a New Zealand physicist who came to be known as the father of nuclear physics.
''Encyclopædia Britannica'' considers him to be the greatest ...
through simple experimentation in 1899, but these proved to be charged particulate types of radiation. However, in 1900 the French scientist Paul Villard discovered a third neutrally charged and especially penetrating type of radiation from radium, and after he described it, Rutherford realized it must be yet a third type of radiation, which in 1903 Rutherford named gamma ray
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
s. In 1910 British physicist William Henry Bragg
Sir William Henry Bragg (2 July 1862 – 12 March 1942) was an English physicist, chemist, mathematician, and active sportsman who uniquelyThis is still a unique accomplishment, because no other parent-child combination has yet shared a Nob ...
demonstrated that gamma rays are electromagnetic radiation, not particles, and in 1914 Rutherford and Edward Andrade
Edward Neville da Costa Andrade FRS (27 December 1887 – 6 June 1971) was an English physicist, writer, and poet. He told ''The Literary Digest'' his name was pronounced "as written, i.e., like ''air raid'', with ''and'' substituted for ''air' ...
measured their wavelengths, finding that they were similar to X-rays but with shorter wavelengths and higher frequency, although a 'cross-over' between X and gamma rays makes it possible to have X-rays with a higher energy (and hence shorter wavelength) than gamma rays and vice versa. The origin of the ray differentiates them, gamma rays tend to be natural phenomena originating from the unstable nucleus of an atom and X-rays are electrically generated (and hence man-made) unless they are as a result of bremsstrahlung
''Bremsstrahlung'' (), from "to brake" and "radiation"; i.e., "braking radiation" or "deceleration radiation", is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typicall ...
X-radiation caused by the interaction of fast moving particles (such as beta particles) colliding with certain materials, usually of higher atomic numbers.
Electromagnetic spectrum

radio
Radio is the technology of signaling and communicating using radio waves. Radio waves are electromagnetic waves of frequency between 30 hertz (Hz) and 300 gigahertz (GHz). They are generated by an electronic device called a transmi ...
, microwave
Microwave is a form of electromagnetic radiation with wavelengths ranging from about one meter to one millimeter corresponding to frequencies between 300 MHz and 300 GHz respectively. Different sources define different frequency ra ...
, infrared
Infrared (IR), sometimes called infrared light, is electromagnetic radiation (EMR) with wavelengths longer than those of visible light. It is therefore invisible to the human eye. IR is generally understood to encompass wavelengths from around ...
, visible, ultraviolet
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation ...
, X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
s and gamma rays
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
. Arbitrary electromagnetic waves can be expressed by Fourier analysis in terms of sinusoidal monochromatic waves, which in turn can each be classified into these regions of the EMR spectrum.
For certain classes of EM waves, the waveform is most usefully treated as ''random'', and then spectral analysis must be done by slightly different mathematical techniques appropriate to random or stochastic processes. In such cases, the individual frequency components are represented in terms of their ''power'' content, and the phase information is not preserved. Such a representation is called the power spectral density
The power spectrum S_(f) of a time series x(t) describes the distribution of power into frequency components composing that signal. According to Fourier analysis, any physical signal can be decomposed into a number of discrete frequencies, ...
of the random process. Random electromagnetic radiation requiring this kind of analysis is, for example, encountered in the interior of stars, and in certain other very wideband forms of radiation such as the Zero point wave field of the electromagnetic vacuum.
The behavior of EM radiation and its interaction with matter depends on its frequency, and changes qualitatively as the frequency changes. Lower frequencies have longer wavelengths, and higher frequencies have shorter wavelengths, and are associated with photons of higher energy. There is no fundamental limit known to these wavelengths or energies, at either end of the spectrum, although photons with energies near the Planck energy or exceeding it (far too high to have ever been observed) will require new physical theories to describe.
Radio and microwave
When radio waves impinge upon a conductor, they couple to the conductor, travel along it and induce an electric current on the conductor surface by moving the electrons of the conducting material in correlated bunches of charge. Such effects can cover macroscopic distances in conductors (such as radio antennas), since the wavelength of radiowaves is long. Electromagnetic radiation phenomena with wavelengths ranging from as long as one meter to as short as one millimeter are called microwaves; with frequencies between 300 MHz (0.3 GHz) and 300 GHz. At radio and microwave frequencies, EMR interacts with matter largely as a bulk collection of charges which are spread out over large numbers of affected atoms. Inelectrical conductor
In physics and electrical engineering, a conductor is an object or type of material that allows the flow of charge (electric current) in one or more directions. Materials made of metal are common electrical conductors. Electric current is gene ...
s, such induced bulk movement of charges ( electric currents) results in absorption of the EMR, or else separations of charges that cause generation of new EMR (effective reflection of the EMR). An example is absorption or emission of radio waves by antennas, or absorption of microwaves by water or other molecules with an electric dipole moment, as for example inside a microwave oven
A microwave oven (commonly referred to as a microwave) is an electric oven that heats and cooks food by exposing it to electromagnetic radiation in the microwave frequency range. This induces polar molecules in the food to rotate and produce ...
. These interactions produce either electric currents or heat, or both.
Infrared
Like radio and microwave, infrared (IR) also is reflected by metals (and also most EMR, well into the ultraviolet range). However, unlike lower-frequency radio and microwave radiation, Infrared EMR commonly interacts with dipoles present in single molecules, which change as atoms vibrate at the ends of a single chemical bond. It is consequently absorbed by a wide range of substances, causing them to increase in temperature as the vibrations dissipate as heat. The same process, run in reverse, causes bulk substances to radiate in the infrared spontaneously (seethermal radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of particles in matter. Thermal radiation is generated when heat from the movement of charges in the material (electrons and protons in common forms of matter) i ...
section below).
Infrared radiation is divided into spectral subregions. While different subdivision schemes exist, the spectrum is commonly divided as near-infrared (0.75–1.4 μm), short-wavelength infrared (1.4–3 μm), mid-wavelength infrared (3–8 μm), long-wavelength infrared (8–15 μm) and far infrared
Far infrared (FIR) is a region in the infrared spectrum of electromagnetic radiation. Far infrared is often defined as any radiation with a wavelength of 15 micrometers (μm) to 1 mm (corresponding to a range of about 20 THz to ...
(15–1000 μm).
Visible light
Natural sources produce EM radiation across the spectrum. EM radiation with awavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, t ...
between approximately 400 nm and 700 nm is directly detected by the human eye and perceived as visible light. Other wavelengths, especially nearby infrared (longer than 700 nm) and ultraviolet (shorter than 400 nm) are also sometimes referred to as light.
As frequency increases into the visible range, photons have enough energy to change the bond structure of some individual molecules. It is not a coincidence that this happens in the visible range, as the mechanism of vision involves the change in bonding of a single molecule, retinal
Retinal (also known as retinaldehyde) is a polyene chromophore. Retinal, bound to proteins called opsins, is the chemical basis of visual phototransduction, the light-detection stage of visual perception (vision).
Some microorganisms use reti ...
, which absorbs a single photon. The change in retinal causes a change in the shape of the rhodopsin protein it is contained in, which starts the biochemical process that causes the retina
The retina (from la, rete "net") is the innermost, light-sensitive layer of tissue of the eye of most vertebrates and some molluscs. The optics of the eye create a focused two-dimensional image of the visual world on the retina, which then ...
of the human eye to sense the light.
Photosynthesis
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored i ...
becomes possible in this range as well, for the same reason. A single molecule of chlorophyll is excited by a single photon. In plant tissues that conduct photosynthesis, carotenoids
Carotenoids (), also called tetraterpenoids, are yellow, orange, and red organic pigments that are produced by plants and algae, as well as several bacteria, and fungi. Carotenoids give the characteristic color to pumpkins, carrots, parsnips, co ...
act to quench electronically excited chlorophyll produced by visible light in a process called non-photochemical quenching
Non-photochemical quenching (NPQ) is a mechanism employed by plants and algae to protect themselves from the adverse effects of high light intensity. It involves the quenching of singlet excited state chlorophylls (Chl) via enhanced internal con ...
, to prevent reactions that would otherwise interfere with photosynthesis at high light levels.
Animals that detect infrared make use of small packets of water that change temperature, in an essentially thermal process that involves many photons.
Infrared, microwaves and radio waves are known to damage molecules and biological tissue only by bulk heating, not excitation from single photons of the radiation.
Visible light is able to affect only a tiny percentage of all molecules. Usually not in a permanent or damaging way, rather the photon excites an electron which then emits another photon when returning to its original position. This is the source of color produced by most dyes. Retinal
Retinal (also known as retinaldehyde) is a polyene chromophore. Retinal, bound to proteins called opsins, is the chemical basis of visual phototransduction, the light-detection stage of visual perception (vision).
Some microorganisms use reti ...
is an exception. When a photon is absorbed, the retinal permanently changes structure from cis to trans, and requires a protein to convert it back, i.e. reset it to be able to function as a light detector again.
Limited evidence indicate that some reactive oxygen species
In chemistry, reactive oxygen species (ROS) are highly reactive chemicals formed from diatomic oxygen (). Examples of ROS include peroxides, superoxide, hydroxyl radical, singlet oxygen, and alpha-oxygen.
The reduction of molecular oxygen () p ...
are created by visible light in skin, and that these may have some role in photoaging, in the same manner as ultraviolet A
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation i ...
.
Ultraviolet
As frequency increases into the ultraviolet, photons now carry enough energy (about threeelectron volt
In physics, an electronvolt (symbol eV, also written electron-volt and electron volt) is the measure of an amount of kinetic energy gained by a single electron accelerating from rest through an electric potential difference of one volt in vacuum ...
s or more) to excite certain doubly bonded molecules into permanent chemical rearrangement. In DNA, this causes lasting damage. DNA is also indirectly damaged by reactive oxygen species produced by ultraviolet A (UVA), which has energy too low to damage DNA directly. This is why ultraviolet at all wavelengths can damage DNA, and is capable of causing cancer, and (for UVB
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation i ...
) skin burns (sunburn) that are far worse than would be produced by simple heating (temperature increase) effects. This property of causing molecular damage that is out of proportion to heating effects, is characteristic of all EMR with frequencies at the visible light range and above. These properties of high-frequency EMR are due to quantum effects that permanently damage materials and tissues at the molecular level.
At the higher end of the ultraviolet range, the energy of photons becomes large enough to impart enough energy to electrons to cause them to be liberated from the atom, in a process called photoionisation. The energy required for this is always larger than about 10 electron volt
In physics, an electronvolt (symbol eV, also written electron-volt and electron volt) is the measure of an amount of kinetic energy gained by a single electron accelerating from rest through an electric potential difference of one volt in vacuum ...
(eV) corresponding with wavelengths smaller than 124 nm (some sources suggest a more realistic cutoff of 33 eV, which is the energy required to ionize water). This high end of the ultraviolet spectrum with energies in the approximate ionization range, is sometimes called "extreme UV." Ionizing UV is strongly filtered by the Earth's atmosphere.
X-rays and gamma rays
Electromagnetic radiation composed of photons that carry minimum-ionization energy, or more, (which includes the entire spectrum with shorter wavelengths), is therefore termed ionizing radiation. (Many other kinds of ionizing radiation are made of non-EM particles). Electromagnetic-type ionizing radiation extends from the extreme ultraviolet to all higher frequencies and shorter wavelengths, which means that allX-rays
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nbs ...
and gamma rays
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
qualify. These are capable of the most severe types of molecular damage, which can happen in biology to any type of biomolecule, including mutation and cancer, and often at great depths below the skin, since the higher end of the X-ray spectrum, and all of the gamma ray spectrum, penetrate matter.
Atmosphere and magnetosphere
nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
, and then (for wavelengths in the upper UV) from the electronic excitation of dioxygen
There are several known allotropes of oxygen. The most familiar is molecular oxygen (O2), present at significant levels in Earth's atmosphere and also known as dioxygen or triplet oxygen. Another is the highly reactive ozone (O3). Others are:
* ...
and finally ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lo ...
at the mid-range of UV. Only 30% of the Sun's ultraviolet light reaches the ground, and almost all of this is well transmitted.
Visible light is well transmitted in air, as it is not energetic enough to excite nitrogen, oxygen, or ozone, but too energetic to excite molecular vibrational frequencies of water vapor.
Absorption bands in the infrared are due to modes of vibrational excitation in water vapor. However, at energies too low to excite water vapor, the atmosphere becomes transparent again, allowing free transmission of most microwave and radio waves.
Finally, at radio wavelengths longer than 10 m or so (about 30 MHz), the air in the lower atmosphere remains transparent to radio, but plasma in certain layers of the ionosphere begins to interact with radio waves (see skywave). This property allows some longer wavelengths (100 m or 3 MHz) to be reflected and results in shortwave radio beyond line-of-sight. However, certain ionospheric effects begin to block incoming radiowaves from space, when their frequency is less than about 10 MHz (wavelength longer than about 30 m).
Thermal and electromagnetic radiation as a form of heat
The basic structure ofmatter
In classical physics and general chemistry, matter is any substance that has mass and takes up space by having volume. All everyday objects that can be touched are ultimately composed of atoms, which are made up of interacting subatomic part ...
involves charged particles bound together. When electromagnetic radiation impinges on matter, it causes the charged particles to oscillate and gain energy. The ultimate fate of this energy depends on the context. It could be immediately re-radiated and appear as scattered, reflected, or transmitted radiation. It may get dissipated into other microscopic motions within the matter, coming to thermal equilibrium
Two physical systems are in thermal equilibrium if there is no net flow of thermal energy between them when they are connected by a path permeable to heat. Thermal equilibrium obeys the zeroth law of thermodynamics. A system is said to be i ...
and manifesting itself as thermal energy
The term "thermal energy" is used loosely in various contexts in physics and engineering. It can refer to several different well-defined physical concepts. These include the internal energy or enthalpy of a body of matter and radiation; heat, de ...
, or even kinetic energy
In physics, the kinetic energy of an object is the energy that it possesses due to its motion.
It is defined as the work needed to accelerate a body of a given mass from rest to its stated velocity. Having gained this energy during its acc ...
, in the material. With a few exceptions related to high-energy photons (such as fluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, tha ...
, harmonic generation
Harmonic generation (HG, also called multiple harmonic generation) is a nonlinear optical process in which n photons with the same frequency interact with a nonlinear material, are "combined", and generate a new photon with n times the energy of ...
, photochemical reaction Organic photochemistry encompasses organic reactions that are induced by the action of light. The absorption of ultraviolet light by organic molecules often leads to reactions. In the earliest days, sunlight was employed, while in more modern times ...
s, the photovoltaic effect
The photovoltaic effect is the generation of voltage and electric current in a material upon exposure to light. It is a physical and chemical phenomenon.
The photovoltaic effect is closely related to the photoelectric effect. For both phenomena, ...
for ionizing radiations at far ultraviolet, X-ray and gamma radiation), absorbed electromagnetic radiation simply deposits its energy by heating the material. This happens for infrared, microwave and radio wave radiation. Intense radio waves can thermally burn living tissue and can cook food. In addition to infrared laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The fi ...
s, sufficiently intense visible and ultraviolet lasers can easily set paper afire.
Ionizing radiation creates high-speed electrons in a material and breaks chemical bonds, but after these electrons collide many times with other atoms eventually most of the energy becomes thermal energy all in a tiny fraction of a second. This process makes ionizing radiation far more dangerous per unit of energy than non-ionizing radiation. This caveat also applies to UV, even though almost all of it is not ionizing, because UV can damage molecules due to electronic excitation, which is far greater per unit energy than heating effects.
Infrared radiation in the spectral distribution of a black body
A black body or blackbody is an idealized physical body that absorbs all incident electromagnetic radiation, regardless of frequency or angle of incidence. The name "black body" is given because it absorbs all colors of light. A black body ...
is usually considered a form of heat, since it has an equivalent temperature and is associated with an entropy change per unit of thermal energy. However, "heat" is a technical term in physics and thermodynamics and is often confused with thermal energy. Any type of electromagnetic energy can be transformed into thermal energy in interaction with matter. Thus, ''any'' electromagnetic radiation can "heat" (in the sense of increase the thermal energy
The term "thermal energy" is used loosely in various contexts in physics and engineering. It can refer to several different well-defined physical concepts. These include the internal energy or enthalpy of a body of matter and radiation; heat, de ...
temperature of) a material, when it is absorbed.
The inverse or time-reversed process of absorption is thermal radiation. Much of the thermal energy in matter consists of random motion of charged particles, and this energy can be radiated away from the matter. The resulting radiation may subsequently be absorbed by another piece of matter, with the deposited energy heating the material.
The electromagnetic radiation in an opaque cavity at thermal equilibrium is effectively a form of thermal energy, having maximum radiation entropy.
Biological effects
Bioelectromagnetics
Bioelectromagnetics, also known as bioelectromagnetism, is the study of the interaction between electromagnetic fields and biological entities. Areas of study include electromagnetic fields produced by living cells, tissues or organisms, th ...
is the study of the interactions and effects of EM radiation on living organisms. The effects of electromagnetic radiation upon living cells, including those in humans, depends upon the radiation's power and frequency. For low-frequency radiation (radio waves to visible light) the best-understood effects are those due to radiation power alone, acting through heating when radiation is absorbed. For these thermal effects, frequency is important as it affects the intensity of the radiation and penetration into the organism (for example, microwaves penetrate better than infrared). It is widely accepted that low frequency fields that are too weak to cause significant heating could not possibly have any biological effect.
Despite the commonly accepted results, some research has been conducted to show that weaker ''non-thermal'' electromagnetic fields (including weak ELF magnetic fields, although the latter does not strictly qualify as EM radiation) and modulated RF and microwave fields have biological effects. Fundamental mechanisms of the interaction between biological material and electromagnetic fields at non-thermal levels are not fully understood.
The World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level of ...
has classified radio frequency electromagnetic radiation as Group 2B – possibly carcinogenic. This group contains possible carcinogens such as lead, DDT, and styrene. For example, epidemiological studies looking for a relationship between cell phone use and brain cancer development have been largely inconclusive, save to demonstrate that the effect, if it exists, cannot be a large one.
At higher frequencies (visible and beyond), the effects of individual photons begin to become important, as these now have enough energy individually to directly or indirectly damage biological molecules. All UV frequencies have been classed as Group 1 carcinogens by the World Health Organization. Ultraviolet radiation from sun exposure is the primary cause of skin cancer.
Thus, at UV frequencies and higher (and probably somewhat also in the visible range), electromagnetic radiation does more damage to biological systems than simple heating predicts. This is most obvious in the "far" (or "extreme") ultraviolet. UV, with X-ray and gamma radiation, are referred to as ionizing radiation due to the ability of photons of this radiation to produce ion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
s and free radical
A daughter category of ''Ageing'', this category deals only with the biological aspects of ageing.
Ageing
Ailments of unknown cause
Biogerontology
Biological processes
Causes of death
Cellular processes
Gerontology
Life extension
Metabo ...
s in materials (including living tissue). Since such radiation can severely damage life at energy levels that produce little heating, it is considered far more dangerous (in terms of damage-produced per unit of energy, or power) than the rest of the electromagnetic spectrum.
Use as weapon
The heat ray is an application of EMR that makes use of microwave frequencies to create an unpleasant heating effect in the upper layer of the skin. A publicly known heat ray weapon called theActive Denial System
The Active Denial System (ADS) is a non-lethal directed-energy weapon developed by the U.S. military, designed for area denial, perimeter security and crowd control. Informally, the weapon is also called the heat ray since it works by heating t ...
was developed by the US military as an experimental weapon to deny the enemy access to an area. A death ray
The death ray or death beam was a theoretical particle beam or electromagnetic weapon first theorized around the 1920s and 1930s. Around that time, notable inventors such as Guglielmo Marconi, Nikola Tesla, Harry Grindell Matthews, Edwin R. Sco ...
is a theoretical weapon that delivers heat ray based on electromagnetic energy at levels that are capable of injuring human tissue. An inventor of a death ray, Harry Grindell Matthews
Harry Grindell Matthews (17 March 1880 – 11 September 1941) was an English inventor who claimed to have invented a death ray in the 1920s.
Earlier life and inventions
Harry Grindell Matthews was born on 17 March 1880 in Winterbourne, Glo ...
, claimed to have lost sight in his left eye while working on his death ray weapon based on a microwave magnetron
The cavity magnetron is a high-power vacuum tube used in early radar systems and currently in microwave ovens and linear particle accelerators. It generates microwaves using the interaction of a stream of electrons with a magnetic field while ...
from the 1920s (a normal microwave oven
A microwave oven (commonly referred to as a microwave) is an electric oven that heats and cooks food by exposing it to electromagnetic radiation in the microwave frequency range. This induces polar molecules in the food to rotate and produce ...
creates a tissue damaging cooking effect inside the oven at around 2 kV/m).
Derivation from electromagnetic theory
Electromagnetic waves are predicted by the classical laws of electricity and magnetism, known asMaxwell's equations
Maxwell's equations, or Maxwell–Heaviside equations, are a set of coupled partial differential equations that, together with the Lorentz force law, form the foundation of classical electromagnetism, classical optics, and electric circuits.
...
. There are nontrivial solutions of the homogeneous Maxwell's equations (without charges or currents), describing ''waves'' of changing electric and magnetic fields. Beginning with Maxwell's equations in free space
A vacuum is a space devoid of matter. The word is derived from the Latin adjective ''vacuus'' for "vacant" or "void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often dis ...
:
where
* and are the electric field (measured in V/m or N/ C) and the magnetic field (measured in T or Wb/m2), respectively;
* yields the divergence
In vector calculus, divergence is a vector operator that operates on a vector field, producing a scalar field giving the quantity of the vector field's source at each point. More technically, the divergence represents the volume density of t ...
and the curl of a vector field
* and are partial derivatives
In mathematics, a partial derivative of a function of several variables is its derivative with respect to one of those variables, with the others held constant (as opposed to the total derivative, in which all variables are allowed to vary). Pa ...
(rate of change in time, with location fixed) of the magnetic and electric field;
* is the permeability of a vacuum (4 × 10−7 ( H/m)), and is the permittivity
In electromagnetism, the absolute permittivity, often simply called permittivity and denoted by the Greek letter ''ε'' (epsilon), is a measure of the electric polarizability of a dielectric. A material with high permittivity polarizes more in ...
of a vacuum (8.85×10−12 ( F/m));
Besides the trivial solution
useful solutions can be derived with the following vector identity
The following are important identities involving derivatives and integrals in vector calculus.
Operator notation
Gradient
For a function f(x, y, z) in three-dimensional Cartesian coordinate variables, the gradient is the vector field:
\o ...
, valid for all vectors in some vector field:
Taking the curl of the second Maxwell equation () yields:
Evaluating the left hand side of () with the above identity and simplifying using (), yields:
Evaluating the right hand side of () by exchanging the sequence of derivations and inserting the fourth yields:
Combining () and () again, gives a vector-valued differential equation
In mathematics, a differential equation is an equation that relates one or more unknown functions and their derivatives. In applications, the functions generally represent physical quantities, the derivatives represent their rates of change, an ...
for the electric field, solving the homogeneous Maxwell equations:
Taking the curl of the fourth Maxwell equation () results in a similar differential equation for a magnetic field solving the homogeneous Maxwell equations:
Both differential equations have the form of the general wave equation
The (two-way) wave equation is a second-order linear partial differential equation for the description of waves or standing wave fields — as they occur in classical physics — such as mechanical waves (e.g. water waves, sound waves and seism ...
for waves propagating with speed where is a function of time and location, which gives the amplitude of the wave at some time at a certain location:
This is also written as:
where denotes the so-called d'Alembert operator
In special relativity, electromagnetism and wave theory, the d'Alembert operator (denoted by a box: \Box), also called the d'Alembertian, wave operator, box operator or sometimes quabla operator (''cf''. nabla symbol) is the Laplace operator of Mi ...
, which in Cartesian coordinates is given as:
Comparing the terms for the speed of propagation, yields in the case of the electric and magnetic fields:
This is the speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
in vacuum. Thus Maxwell's equations connect the vacuum permittivity , the vacuum permeability
The vacuum magnetic permeability (variously ''vacuum permeability'', ''permeability of free space'', ''permeability of vacuum''), also known as the magnetic constant, is the magnetic permeability in a classical vacuum. It is a physical constant, ...
, and the speed of light, ''c''0, via the above equation. This relationship had been discovered by Wilhelm Eduard Weber
Wilhelm Eduard Weber (; ; 24 October 1804 – 23 June 1891) was a German physicist and, together with Carl Friedrich Gauss, inventor of the first electromagnetic telegraph.
Biography of Wilhelm
Early years
Weber was born in Schlossstrasse i ...
and Rudolf Kohlrausch prior to the development of Maxwell's electrodynamics, however Maxwell was the first to produce a field theory consistent with waves traveling at the speed of light.
These are only two equations versus the original four, so more information pertains to these waves hidden within Maxwell's equations. A generic vector wave for the electric field has the form
Here, is the constant amplitude, is any second differentiable function, is a unit vector in the direction of propagation, and is a position vector. is a generic solution to the wave equation. In other words,
for a generic wave traveling in the direction.
From the first of Maxwell's equations, we get
Thus,
which implies that the electric field is orthogonal to the direction the wave propagates. The second of Maxwell's equations yields the magnetic field, namely,
Thus,
The remaining equations will be satisfied by this choice of .
The electric and magnetic field waves in the far-field travel at the speed of light. They have a special restricted orientation and proportional magnitudes, , which can be seen immediately from the Poynting vector. The electric field, magnetic field, and direction of wave propagation are all orthogonal, and the wave propagates in the same direction as . Also, E and B far-fields in free space, which as wave solutions depend primarily on these two Maxwell equations, are in-phase with each other. This is guaranteed since the generic wave solution is first order in both space and time, and the curl operator on one side of these equations results in first-order spatial derivatives of the wave solution, while the time-derivative on the other side of the equations, which gives the other field, is first-order in time, resulting in the same phase shift
In physics and mathematics, the phase of a periodic function F of some real variable t (such as time) is an angle-like quantity representing the fraction of the cycle covered up to t. It is denoted \phi(t) and expressed in such a scale that it ...
for both fields in each mathematical operation.
From the viewpoint of an electromagnetic wave traveling forward, the electric field might be oscillating up and down, while the magnetic field oscillates right and left. This picture can be rotated with the electric field oscillating right and left and the magnetic field oscillating down and up. This is a different solution that is traveling in the same direction. This arbitrariness in the orientation with respect to propagation direction is known as polarization. On a quantum level, it is described as photon polarization
Photon polarization is the quantum mechanical description of the classical polarized sinusoidal plane electromagnetic wave. An individual photon
can be described as having right or left circular polarization, or a superposition of the two. Equ ...
. The direction of the polarization is defined as the direction of the electric field.
More general forms of the second-order wave equations given above are available, allowing for both non-vacuum propagation media and sources. Many competing derivations exist, all with varying levels of approximation and intended applications. One very general example is a form of the electric field equation, which was factorized into a pair of explicitly directional wave equations, and then efficiently reduced into a single uni-directional wave equation by means of a simple slow-evolution approximation.
See also
*Antenna measurement
Antenna measurement techniques refers to the testing of antennas to ensure that the antenna meets specifications or simply to characterize it. Typical parameters of antennas are gain, bandwidth, radiation pattern, beamwidth, polarization, and imp ...
* Bioelectromagnetics
Bioelectromagnetics, also known as bioelectromagnetism, is the study of the interaction between electromagnetic fields and biological entities. Areas of study include electromagnetic fields produced by living cells, tissues or organisms, th ...
* Bolometer
A bolometer is a device for measuring radiant heat by means of a material having a temperature-dependent electrical resistance. It was invented in 1878 by the American astronomer Samuel Pierpont Langley.
Principle of operation
A bolometer ...
* CONELRAD
CONELRAD (''Control of Electromagnetic Radiation'') was a method of emergency broadcasting to the public of the United States in the event of enemy attack during the Cold War. It was intended to allow continuous broadcast of civil defense informa ...
* Electromagnetic pulse
An electromagnetic pulse (EMP), also a transient electromagnetic disturbance (TED), is a brief burst of electromagnetic energy. Depending upon the source, the origin of an EMP can be natural or artificial, and can occur as an electromagnetic fi ...
* Electromagnetic radiation and health
Electromagnetic radiation can be classified into two types: ionizing radiation and non-ionizing radiation, based on the capability of a single photon with more than 10 eV energy to ionize atoms or break chemical bonds. Extreme ultra ...
* Evanescent wave coupling
* Finite-difference time-domain method
Finite-difference time-domain (FDTD) or Yee's method (named after the Chinese American applied mathematician Kane S. Yee, born 1934) is a numerical analysis technique used for modeling computational electrodynamics (finding approximate solutions t ...
* Gravitational wave
* Helicon
* Impedance of free space
* Radiation reaction
* Health effects of sunlight exposure
The ultraviolet radiation in sunlight has both positive and negative health effects, as it is required for the synthesis of vitamin D3 and is a mutagen. A dietary supplement can supply vitamin D without this mutagenic effect, but supplementatio ...
* Sinusoidal plane-wave solutions of the electromagnetic wave equation
References
*Further reading
* * * * * *External links
The Feynman Lectures on Physics Vol. I Ch. 28: Electromagnetic Radiation
*
''Electromagnetic Waves from Maxwell's Equations''
o
Project PHYSNET
"Electromagnetic radiation"
in the ''
Encyclopædia Britannica
The (Latin for "British Encyclopædia") is a general knowledge English-language encyclopaedia. It is published by Encyclopædia Britannica, Inc.; the company has existed since the 18th century, although it has changed ownership various t ...
''
{{Authority control
Heinrich Hertz
Radiation