Dess–Martin periodinane on:
[Wikipedia]
[Google]
[Amazon]
Dess–Martin periodinane (DMP) is a chemical reagent used in the Dess–Martin oxidation, oxidizing primary alcohols to 
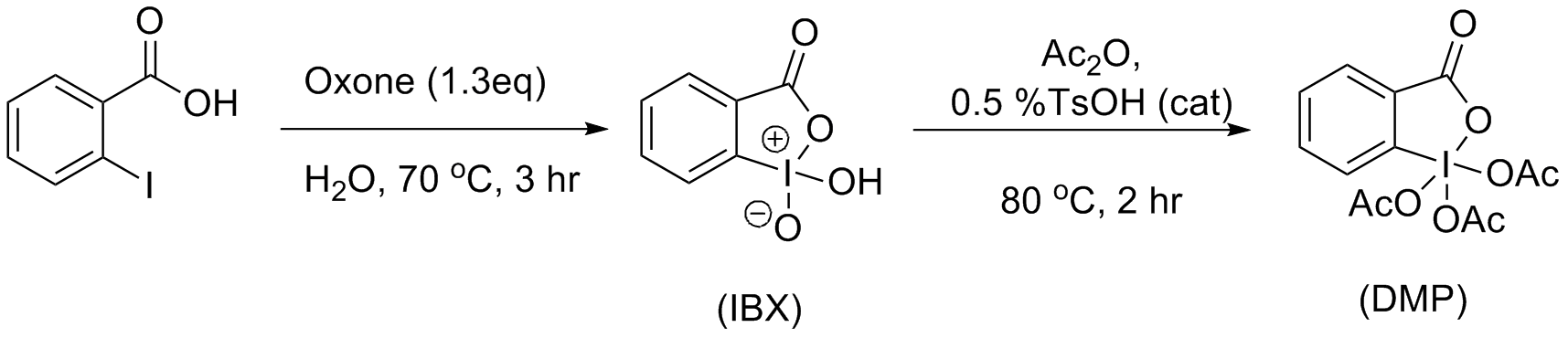 The classic method presented by R. K. Boeckman and J. J. Mullins involved heating a solution of
The classic method presented by R. K. Boeckman and J. J. Mullins involved heating a solution of 
 Schreiber and coworkers have shown that water increases the rate of the oxidation reaction. Dess and Martin had originally observed that the oxidation of ethanol was increased when there was an extra equivalent of ethanol. It is believed that the rate of dissociation of the final acetate ligand from the iodine is increased, because of the electron-donating ability of the hydroxyl group (thus weakening the I-OAc bond).
:
Schreiber and coworkers have shown that water increases the rate of the oxidation reaction. Dess and Martin had originally observed that the oxidation of ethanol was increased when there was an extra equivalent of ethanol. It is believed that the rate of dissociation of the final acetate ligand from the iodine is increased, because of the electron-donating ability of the hydroxyl group (thus weakening the I-OAc bond).
:


aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl gro ...
s and secondary alcohol
Alcohol most commonly refers to:
* Alcohol (chemistry), an organic compound in which a hydroxyl group is bound to a carbon atom
* Alcohol (drug), an intoxicant found in alcoholic drinks
Alcohol may also refer to:
Chemicals
* Ethanol, one of sev ...
s to ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double b ...
s. This periodinane Periodinanes also known as λ5-iodanes are organoiodine compounds with iodine in the +5 oxidation state. These compounds are described as hypervalent because the iodine center has more than 8 valence electrons.
Periodinane compounds
The λ5-iodane ...
has several advantages over chromium
Chromium is a chemical element with the symbol Cr and atomic number 24. It is the first element in group 6. It is a steely-grey, lustrous, hard, and brittle transition metal.
Chromium metal is valued for its high corrosion resistance and hard ...
- and DMSO
Dimethyl sulfoxide (DMSO) is an organosulfur compound with the formula ( CH3)2. This colorless liquid is the sulfoxide most widely used commercially. It is an important polar aprotic solvent that dissolves both polar and nonpolar compounds ...
-based oxidants that include milder conditions (room temperature, neutral pH), shorter reaction times, higher yields, simplified workups, high chemoselectivity, tolerance of sensitive functional groups, and a long shelf life. However, use on an industrial scale is made difficult by its cost and its potentially explosive nature. It is named after the American chemists Daniel Benjamin Dess and James Cullen Martin
James Cullen Martin (January 14, 1928 – April 20, 1999) was an American chemist. Known in the field as "J.C.", he specialized in physical organic chemistry with an emphasis on main group element chemistry.
Martin received his undergradua ...
who developed the reagent in 1983. It is based on IBX
2-Iodoxybenzoic acid (IBX) is an organic compound used in organic synthesis as an oxidizing agent. This periodinane is especially suited to oxidize alcohols to aldehydes. IBX is prepared from 2-iodobenzoic acid, potassium bromate, and sulfuric ...
, but due to the acetate groups attached to the central iodine atom, DMP is much more reactive than IBX and is much more soluble in organic solvents.
:
Preparation
The most friendly synthesis of IBX has been determined to be treating 2-iodobenzoic acid withoxone
Potassium peroxymonosulfate is widely used as an oxidizing agent. It is the potassium salt of peroxymonosulfuric acid. Usually potassium peroxymonosulfate refers to the triple salt known as oxone.
The standard electrode potential for potassium ...
in water, at elevated temperatures for 3 hours. IBX is then acylated using Ireland and Liu’s
modifications from the original procedure. These modifications allowed for higher yields and a simplified work up procedure. The resulted solids can be obtained via filtration and washing with ether. Ireland and Liu used a catalytic amount of tosylic acid, which allowed the reaction to complete in less than 2 hours (compared to the classic synthesis, utilizing 24 hours) and in yields exceeding 90%.
: The classic method presented by R. K. Boeckman and J. J. Mullins involved heating a solution of
The classic method presented by R. K. Boeckman and J. J. Mullins involved heating a solution of potassium bromate
Potassium bromate (KBrO3), is a bromate of potassium and takes the form of white crystals or powder. It is a strong oxidizing agent.
It is a toxic and carcinogenic compound.
Preparation
Potassium bromate is produced when bromine is passed throu ...
, sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular fo ...
, 2-iodobenzoic acid to afford IBX (1-hydroxy-1,2-benziodoxol-3(1H)-one 1-oxide, 2-iodoxybenzoic acid
2-Iodoxybenzoic acid (IBX) is an organic compound used in organic synthesis as an oxidizing agent. This periodinane is especially suited to oxidize alcohols to aldehydes. IBX is prepared from 2-iodobenzoic acid, potassium bromate, and sulfuric a ...
). IBX was then acylated using acetic acid and acetic anhydride
Acetic anhydride, or ethanoic anhydride, is the chemical compound with the formula (CH3CO)2O. Commonly abbreviated Ac2O, it is the simplest isolable anhydride of a carboxylic acid and is widely used as a reagent in organic synthesis. It is a co ...
.
:Oxidation mechanism
Dess–Martin periodinane is mainly used as anoxidant
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
for complex, sensitive and multifunctional alcohol
Alcohol most commonly refers to:
* Alcohol (chemistry), an organic compound in which a hydroxyl group is bound to a carbon atom
* Alcohol (drug), an intoxicant found in alcoholic drinks
Alcohol may also refer to:
Chemicals
* Ethanol, one of sev ...
s. One of the reasons for its effectiveness is its high selectivity towards complexation of the hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydrox ...
group, which allows alcohols to rapidly perform ligand
In coordination chemistry, a ligand is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's elect ...
exchange; the first step in the oxidation reaction.
Proton NMR
Proton nuclear magnetic resonance (proton NMR, hydrogen-1 NMR, or 1H NMR) is the application of nuclear magnetic resonance in NMR spectroscopy with respect to hydrogen-1 nuclei within the molecules of a substance, in order to determine the struct ...
has indicated that using one equivalent of alcohol forms the intermediate diacetoxyalkoxyperiodinane. The acetate then acts as a base to deprotonate the α-H from the alcohol to afford the carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
compound, iodinane, and acetic acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main componen ...
.
When a diol
A diol is a chemical compound containing two hydroxyl groups ( groups). An aliphatic diol is also called a glycol. This pairing of functional groups is pervasive, and many subcategories have been identified.
The most common industrial diol is e ...
or more than one equivalent of alcohol is used, acetoxydialkoxyperiodinane is formed instead. Due to the labile nature of this particular periodinane, oxidation occurs much faster.
: Schreiber and coworkers have shown that water increases the rate of the oxidation reaction. Dess and Martin had originally observed that the oxidation of ethanol was increased when there was an extra equivalent of ethanol. It is believed that the rate of dissociation of the final acetate ligand from the iodine is increased, because of the electron-donating ability of the hydroxyl group (thus weakening the I-OAc bond).
:
Schreiber and coworkers have shown that water increases the rate of the oxidation reaction. Dess and Martin had originally observed that the oxidation of ethanol was increased when there was an extra equivalent of ethanol. It is believed that the rate of dissociation of the final acetate ligand from the iodine is increased, because of the electron-donating ability of the hydroxyl group (thus weakening the I-OAc bond).
:
Chemoselectivity
Using the standard Dess–Martin periodinane conditions, alcohols can be oxidized to aldehydes/ketones without affectingfuran
Furan is a heterocyclic organic compound, consisting of a five-membered aromatic ring with four carbon atoms and one oxygen atom. Chemical compounds containing such rings are also referred to as furans.
Furan is a colorless, flammable, highl ...
rings, sulfides
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds la ...
, vinyl ethers, and secondary amides
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it is ...
. Allylic alcohols are easily oxidized using DMP, which are typically difficult to convert to their respective carbonyls using the typical oxidants.
Myers and coworkers determined that DMP could oxidize N-protected-amino alcohols, without epimerization (unlike most other oxidants, including Swern oxidation). These protected amino alcohols can be very important in the pharmaceutical industry.
Benzylic and allylic alcohols react faster than saturated alcohols, while DMP oxidizes aldoximes and ketoximes to their respective aldehydes and ketones, faster than a primary, secondary or benzylic alcohol to its respective carbonyl.
One example of the Dess–Martin oxidation involves transforming a sensitive α-β-unsaturated alcohol to its corresponding aldehyde. This moiety has been found in several natural products and due to its high functionality, it could be a valuable synthetic building block in organic synthesis. Thongsornkleeb and Danheiser oxidized this sensitive alcohol by employing the Dess Martin Oxidation and altering the work up procedure (diluting with pentanes, washing with poly( 4-vinylpyridine) to remove the acetic acid generated during the reaction, filtering and concentrating via distillation.
:
''t''-Butyl DMP
Difluoro and monofluoro alcohols are more difficult to oxidize.Swern oxidation
The Swern oxidation, named after Daniel Swern, is a chemical reaction whereby a primary or secondary alcohol is oxidized to an aldehyde or ketone using oxalyl chloride, dimethyl sulfoxide (DMSO) and an organic base, such as triethylamine. It is one ...
has been used, but a large excess of the oxidant had to be employed, and in some cases did not give reproducible results. Linderman and Graves found DMP was successful in most cases but could not tolerate the presence of nucleophilic
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are ...
functional groups in the alcohol, as these reacted with DMP by displacing acetate. Using the compound shown below produced the desired carbonyls in high yields as the addition of the ''tert''-butoxy group, due to its steric bulk
Steric effects arise from the spatial arrangement of atoms. When atoms come close together there is a rise in the energy of the molecule. Steric effects are nonbonding interactions that influence the shape ( conformation) and reactivity of ions ...
, minimizes these side reactions.
:
See also
*Alcohol oxidation Alcohol oxidation is a class of organic reactions in which the alcohol functional group is converted into another functional group (e.g., aldehyde, ketone, carboxylic acid) in which carbon carries a higher oxidation state.
Through a variety of m ...
* Pyridinium chlorochromate
* Jones oxidation
The Jones oxidation is an organic reaction for the oxidation of primary and secondary alcohols to carboxylic acids and ketones, respectively. It is named after its discoverer, Sir Ewart Jones. The reaction was an early method for the oxidation of ...
* Oppenauer oxidation
Oppenauer oxidation, named after , is a gentle method for selectively oxidizing secondary alcohols to ketones.
The reaction is the opposite Meerwein–Ponndorf–Verley reduction. The alcohol is oxidized with aluminium isopropoxide in excess ac ...
* Pfitzner–Moffatt oxidation
The Pfitzner–Moffatt oxidation, sometimes referred to as simply the Moffatt oxidation, is a chemical reaction for the oxidation of primary and secondary alcohols to aldehydes and ketones, respectively. The oxidant is a combination of dimethyl ...
* Parikh–Doering oxidation
The Parikh– Doering oxidation is an oxidation reaction that transforms primary and secondary alcohols into aldehydes and ketones, respectively. The procedure uses dimethyl sulfoxide (DMSO) as the oxidant and the solvent, activated by the sul ...
* Albright-Goldman oxidation
* Swern oxidation
The Swern oxidation, named after Daniel Swern, is a chemical reaction whereby a primary or secondary alcohol is oxidized to an aldehyde or ketone using oxalyl chloride, dimethyl sulfoxide (DMSO) and an organic base, such as triethylamine. It is one ...
* Corey–Kim oxidation
* Ley oxidation
Ley may refer to:
Toponyms
* Ley (landform), name for a crag, rock or cliff in the north German language area
* Ley (crater), crater on the Moon
* Ley, Moselle, commune in France
* Ley Hill, hill in England
People
* Ley Matampi (born 19 ...
( TPAP oxidation)
* TEMPO
In musical terminology, tempo ( Italian, 'time'; plural ''tempos'', or ''tempi'' from the Italian plural) is the speed or pace of a given piece. In classical music, tempo is typically indicated with an instruction at the start of a piece (ofte ...
oxidation
References
External links
Oxidizing agents Periodinanes Acetates Iodine heterocycles Lactones Heterocyclic compounds with 2 rings {{DEFAULTSORT:Dess-Martin periodinane