Coronavirus disease 2019 (COVID-19) is a
contagious disease caused by a
virus
A virus is a submicroscopic infectious agent that replicates only inside the living cells of an organism. Viruses infect all life forms, from animals and plants to microorganisms, including bacteria and archaea.
Since Dmitri Ivanovsk ...
, the
severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The first known case was
identified in Wuhan, China, in December 2019.
The disease quickly spread worldwide, resulting in the
COVID-19 pandemic
The COVID-19 pandemic, also known as the coronavirus pandemic, is an ongoing global pandemic of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The novel virus was first identi ...
.
The
symptoms of COVID‑19
Signs and symptoms are the observed or detectable signs, and experienced symptoms of an illness, injury, or condition. A sign for example may be a higher or lower temperature than normal, raised or lowered blood pressure or an abnormality showi ...
are variable but often include fever, cough, headache, fatigue,
breathing difficulties,
loss of smell
Anosmia, also known as smell blindness, is the loss of the ability to detect one or more smells. Anosmia may be temporary or permanent. It differs from hyposmia, which is a decreased sensitivity to some or all smells.
Anosmia can be due to a num ...
, and
loss of taste. Symptoms may begin one to fourteen days
after exposure to the virus. At least a third of people who are infected
do not develop noticeable symptoms. Of those who develop symptoms noticeable enough to be classified as patients, most (81%) develop mild to moderate symptoms (up to mild
pneumonia
Pneumonia is an inflammatory condition of the lung primarily affecting the small air sacs known as alveoli. Symptoms typically include some combination of productive or dry cough, chest pain, fever, and difficulty breathing. The severit ...
), while 14% develop severe symptoms (
dyspnea,
hypoxia, or more than 50% lung involvement on imaging), and 5% develop critical symptoms (
respiratory failure,
shock
Shock may refer to:
Common uses Collective noun
*Shock, a historic commercial term for a group of 60, see English numerals#Special names
* Stook, or shock of grain, stacked sheaves
Healthcare
* Shock (circulatory), circulatory medical emerge ...
, or
multiorgan dysfunction).
Older people
Old age refers to ages nearing or surpassing the life expectancy of human beings, and is thus the end of the human life cycle. Terms and euphemisms for people at this age include old people, the elderly (worldwide usage), OAPs (British usage ...
are at a higher risk of developing severe symptoms. Some people continue to experience a range of effects (
long COVID) for months after recovery, and damage to organs has been observed.
Multi-year studies are underway to further investigate the long-term effects of the disease.
when people breathe air contaminated by droplets and small
airborne
Airborne or Airborn may refer to:
Arts, entertainment, and media
Films
* ''Airborne'' (1962 film), a 1962 American film directed by James Landis
* ''Airborne'' (1993 film), a comedy–drama film
* ''Airborne'' (1998 film), an action film sta ...
particles containing the virus. The risk of breathing these is highest when people are in close proximity, but they can be inhaled over longer distances, particularly indoors. Transmission can also occur if contaminated fluids are splashed or sprayed in the eyes, nose, or mouth, or, more rarely, via contaminated surfaces. People remain contagious for up to 20 days and can spread the virus even if they do not develop symptoms.
to detect the virus's
nucleic acid include
real-time reverse transcription polymerase chain reaction (rRTPCR),
,
and
reverse transcription loop-mediated isothermal amplification
Reverse transcription loop-mediated isothermal amplification (RT-LAMP) is a one step nucleic acid amplification method to multiply specific sequences of RNA. It is used to diagnose infectious disease caused by RNA viruses.
It combines LAMP DNA-de ...
(RTLAMP)
from a
nasopharyngeal swab.
Several
COVID-19 vaccines have been approved and distributed in various countries, which have initiated
mass vaccination campaigns. Other
preventive measures include
physical or social distancing,
quarantining, ventilation of indoor spaces,
use of face masks or coverings in public, covering coughs and sneezes,
hand washing, and keeping unwashed hands away from the face. While work is underway to
develop drugs that inhibit the virus, the primary
treatment is symptomatic. Management involves the
treatment of symptoms through
supportive care,
isolation, and
experimental measures.
Nomenclature
During the initial outbreak in
Wuhan, the virus and disease were commonly referred to as "coronavirus" and "Wuhan coronavirus", with the disease sometimes called "Wuhan pneumonia". In the past, many diseases have been named after geographical locations, such as the
Spanish flu
The 1918–1920 influenza pandemic, commonly known by the misnomer Spanish flu or as the Great Influenza epidemic, was an exceptionally deadly global influenza pandemic caused by the H1N1 influenza A virus. The earliest documented case wa ...
,
Middle East respiratory syndrome, and
Zika virus.
In January 2020, the
World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level o ...
(WHO) recommended 2019-nCoV and 2019-nCoV acute respiratory disease as interim names for the virus and disease per 2015 guidance and international guidelines against using geographical locations or groups of people in disease and virus names to prevent
social stigma.
The official names COVID‑19 and SARS-CoV-2 were issued by the WHO on 11 February 2020 with COVID-19 being shorthand for "coronavirus disease 2019".
The WHO additionally uses "the COVID‑19 virus" and "the virus responsible for COVID‑19" in public communications.
Signs and symptoms
Cause
COVID‑19 is caused by infection with a
strain of
coronavirus known as 'Severe Acute Respiratory Syndrome coronavirus 2' (
SARS-CoV-2
Severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) is a strain of coronavirus that causes COVID-19 (coronavirus disease 2019), the respiratory illness responsible for the ongoing COVID-19 pandemic. The virus previously had a ...
).
Transmission

Virology

Severe acute respiratory syndrome coronavirus2 (SARS-CoV-2) is a
novel severe acute respiratory syndrome coronavirus. It was first isolated from three people with pneumonia connected to the
cluster
may refer to:
Science and technology Astronomy
* Cluster (spacecraft), constellation of four European Space Agency spacecraft
* Asteroid cluster, a small asteroid family
* Cluster II (spacecraft), a European Space Agency mission to study th ...
of acute respiratory illness cases in Wuhan.
All structural features of the novel SARS-CoV-2 virus particle occur in related
coronaviruses in nature.
Outside the human body, the virus is destroyed by household soap, which bursts its
protective bubble.
SARS-CoV-2 is closely related to the original
SARS-CoV.
It is thought to have an animal (
zoonotic) origin. Genetic analysis has revealed that the coronavirus genetically clusters with the genus ''
Betacoronavirus'', in subgenus
''Sarbecovirus'' (lineage B) together with two bat-derived strains. It is 96% identical at the whole
genome
In the fields of molecular biology and genetics, a genome is all the genetic information of an organism. It consists of nucleotide sequences of DNA (or RNA in RNA viruses). The nuclear genome includes protein-coding genes and non-coding ...
level to other bat coronavirus samples (BatCov
RaTG13).
The structural proteins of SARS-CoV-2 include
membrane glycoprotein (M),
envelope protein
A viral envelope is the outermost layer of many types of viruses. It protects the genetic material in their life cycle when traveling between host cells. Not all viruses have envelopes.
Numerous human pathogenic viruses in circulation are encase ...
(E),
nucleocapsid protein (N), and the
spike protein (S). The M protein of SARS-CoV-2 is about 98% similar to the M protein of bat SARS-CoV, maintains around 98% homology with pangolin SARS-CoV, and has 90% homology with the M protein of SARS-CoV; whereas, the similarity is only around 38% with the M protein of MERS-CoV.
SARS-CoV-2 variants
The many thousands of SARS-CoV-2 variants are grouped into either
clades or
lineages.
The WHO, in collaboration with partners, expert networks, national authorities, institutions and researchers, have established nomenclature systems for naming and tracking SARS-CoV-2 genetic lineages by
GISAID,
Nextstrain and
Pango. The expert group convened by the WHO recommended the labelling of variants using letters of the
Greek alphabet
The Greek alphabet has been used to write the Greek language since the late 9th or early 8th century BCE. It is derived from the earlier Phoenician alphabet, and was the earliest known alphabetic script to have distinct letters for vowels as ...
, for example,
Alpha
Alpha (uppercase , lowercase ; grc, ἄλφα, ''álpha'', or ell, άλφα, álfa) is the first letter of the Greek alphabet. In the system of Greek numerals, it has a value of one. Alpha is derived from the Phoenician letter aleph , whi ...
,
Beta,
Delta, and
Gamma
Gamma (uppercase , lowercase ; ''gámma'') is the third letter of the Greek alphabet. In the system of Greek numerals it has a value of 3. In Ancient Greek, the letter gamma represented a voiced velar stop . In Modern Greek, this letter r ...
, giving the justification that they "will be easier and more practical to discussed by non-scientific audiences."
Nextstrain divides the variants into five clades (19A, 19B, 20A, 20B, and 20C), while
GISAID divides them into seven (L, O, V, S, G, GH, and GR).
The Pango tool groups variants into
lineages, with many circulating lineages being classed under the B.1 lineage.
Several notable variants of SARS-CoV-2 emerged throughout 2020.
emerged among
minks and mink farmers in
Denmark
)
, song = ( en, "King Christian stood by the lofty mast")
, song_type = National and royal anthem
, image_map = EU-Denmark.svg
, map_caption =
, subdivision_type = Sovereign state
, subdivision_name = Kingdom of Denmark
, establish ...
. After strict quarantines and a mink euthanasia campaign, the cluster was assessed to no longer be circulating among humans in Denmark as of 1 February 2021.
, there are five dominant variants of SARS-CoV-2 spreading among global populations: the
Alpha variant (B.1.1.7, formerly called the UK variant), first found in London and Kent, the
Beta variant (B.1.351, formerly called the South Africa variant), the
Gamma variant (P.1, formerly called the Brazil variant), the
Delta variant (B.1.617.2, formerly called the India variant), and the
Omicron variant (B.1.1.529), which had spread to 57 countries as of 7 December.
Pathophysiology

The SARS-CoV-2 virus can infect a wide range of cells and systems of the body. COVID‑19 is most known for affecting the upper respiratory tract (sinuses, nose, and throat) and the lower respiratory tract (windpipe and lungs).
The lungs are the organs most affected by COVID‑19 because the virus accesses host cells via the
receptor
Receptor may refer to:
*Sensory receptor, in physiology, any structure which, on receiving environmental stimuli, produces an informative nerve impulse
*Receptor (biochemistry), in biochemistry, a protein molecule that receives and responds to a n ...
for the enzyme
angiotensin-converting enzyme 2 (ACE2), which is most abundant on the surface of
type II alveolar cells of the lungs. The virus uses a special surface glycoprotein called a "
spike" to connect to the ACE2 receptor and enter the host cell.
Respiratory tract
Following viral entry, COVID‑19 infects the ciliated epithelium of the nasopharynx and upper airways.
Nervous system
One common symptom, loss of smell, results from
infection of the support cells of the olfactory epithelium, with subsequent damage to the
olfactory neurons
An olfactory receptor neuron (ORN), also called an olfactory sensory neuron (OSN), is a sensory neuron within the olfactory system.
Structure
Humans have between 10 and 20 million olfactory receptor neurons (ORNs). In vertebrates, ORNs are b ...
.
The involvement of both the central and peripheral nervous system in COVID‑19 has been reported in many medical publications.
It is clear that many people with
COVID-19 exhibit neurological or mental health issues. The virus is not detected in the
central nervous system
The central nervous system (CNS) is the part of the nervous system consisting primarily of the brain and spinal cord. The CNS is so named because the brain integrates the received information and coordinates and influences the activity of all p ...
(CNS) of the majority of COVID-19 patients with
neurological issues. However, SARS-CoV-2 has been detected at low levels in the brains of those who have died from COVID‑19, but these results need to be confirmed.
While virus has been detected in
cerebrospinal fluid
Cerebrospinal fluid (CSF) is a clear, colorless body fluid found within the tissue that surrounds the brain and spinal cord of all vertebrates.
CSF is produced by specialised ependymal cells in the choroid plexus of the ventricles of the ...
of autopsies, the exact mechanism by which it invades the CNS remains unclear and may first involve invasion of peripheral nerves given the low levels of ACE2 in the brain. The virus may also enter the bloodstream from the lungs and cross the blood–brain barrier to gain access to the CNS, possibly within an infected white blood cell.

Research conducted when Alpha was the dominant variant has suggested COVID-19 may cause brain damage. It is unknown if such damage is temporary or permanent, and whether Omicron has similar effects. Observed individuals infected with COVID-19 (most with mild cases) experienced an additional 0.2% to 2% of brain tissue lost in regions of the brain connected to the sense of smell compared with uninfected individuals, and the overall effect on the brain was equivalent on average to at least one extra year of normal ageing; infected individuals also scored lower on several cognitive tests. All effects were more pronounced among older ages.
Gastrointestinal tract
The virus also affects gastrointestinal organs as ACE2 is abundantly expressed in the
glandular cells of
gastric,
duodenal and
rectal epithelium as well as
endothelial
The endothelium is a single layer of squamous endothelial cells that line the interior surface of blood vessels and lymphatic vessels. The endothelium forms an interface between circulating blood or lymph in the lumen and the rest of the ve ...
cells and
enterocytes of the
small intestine.
Cardiovascular system
The virus can cause
acute myocardial injury and chronic damage to the
cardiovascular system.
An acute cardiac injury was found in 12% of infected people admitted to the hospital in Wuhan, China,
and is more frequent in severe disease. Rates of cardiovascular symptoms are high, owing to the systemic inflammatory response and immune system disorders during disease progression, but acute myocardial injuries may also be related to ACE2 receptors in the heart.
ACE2 receptors are highly expressed in the heart and are involved in heart function.
A high incidence of
thrombosis
Thrombosis (from Ancient Greek "clotting") is the formation of a blood clot inside a blood vessel, obstructing the flow of blood through the circulatory system. When a blood vessel (a vein or an artery) is injured, the body uses platelets (th ...
and
venous thromboembolism
Venous thrombosis is blockage of a vein caused by a thrombus (blood clot). A common form of venous thrombosis is deep vein thrombosis (DVT), when a blood clot forms in the deep veins. If a thrombus breaks off (embolizes) and flows to the lungs to ...
occurs in people transferred to
intensive care unit
220px, Intensive care unit
An intensive care unit (ICU), also known as an intensive therapy unit or intensive treatment unit (ITU) or critical care unit (CCU), is a special department of a hospital or health care facility that provides intensi ...
s with COVID‑19 infections, and may be related to poor prognosis. Blood vessel dysfunction and clot formation (as suggested by high
D-dimer
D-dimer (or D dimer) is a fibrin degradation product (or FDP), a small protein fragment present in the blood after a blood clot is degraded by fibrinolysis. It is so named because it contains two D fragments of the fibrin protein joined by a cros ...
levels caused by blood clots) may have a significant role in mortality, incidences of clots leading to
pulmonary embolisms, and
ischaemic events within the brain found as complications leading to death in people infected with COVID‑19.
[ Infection may initiate a chain of vasoconstrictive responses within the body, including pulmonary vasoconstriction a possible mechanism in which oxygenation decreases during pneumonia.]arterioles
An arteriole is a small-diameter blood vessel in the microcirculation that extends and branches out from an artery and leads to capillaries.
Arterioles have muscular walls (usually only one to two layers of smooth muscle cells) and are the pri ...
and capillaries was found in brain tissue samples of people who died from COVID‑19.
COVID‑19 may also cause substantial structural changes to blood cell
A blood cell, also called a hematopoietic cell, hemocyte, or hematocyte, is a cell produced through hematopoiesis and found mainly in the blood. Major types of blood cells include red blood cells (erythrocytes), white blood cells (leukocytes) ...
s, sometimes persisting for months after hospital discharge. A low level of blood lymphocytes may result from the virus acting through ACE2-related entry into lymphocytes.
Other organs
Another common cause of death is complications related to the kidney
The kidneys are two reddish-brown bean-shaped organs found in vertebrates. They are located on the left and right in the retroperitoneal space, and in adult humans are about in length. They receive blood from the paired renal arteries; blo ...
s.
Immunopathology
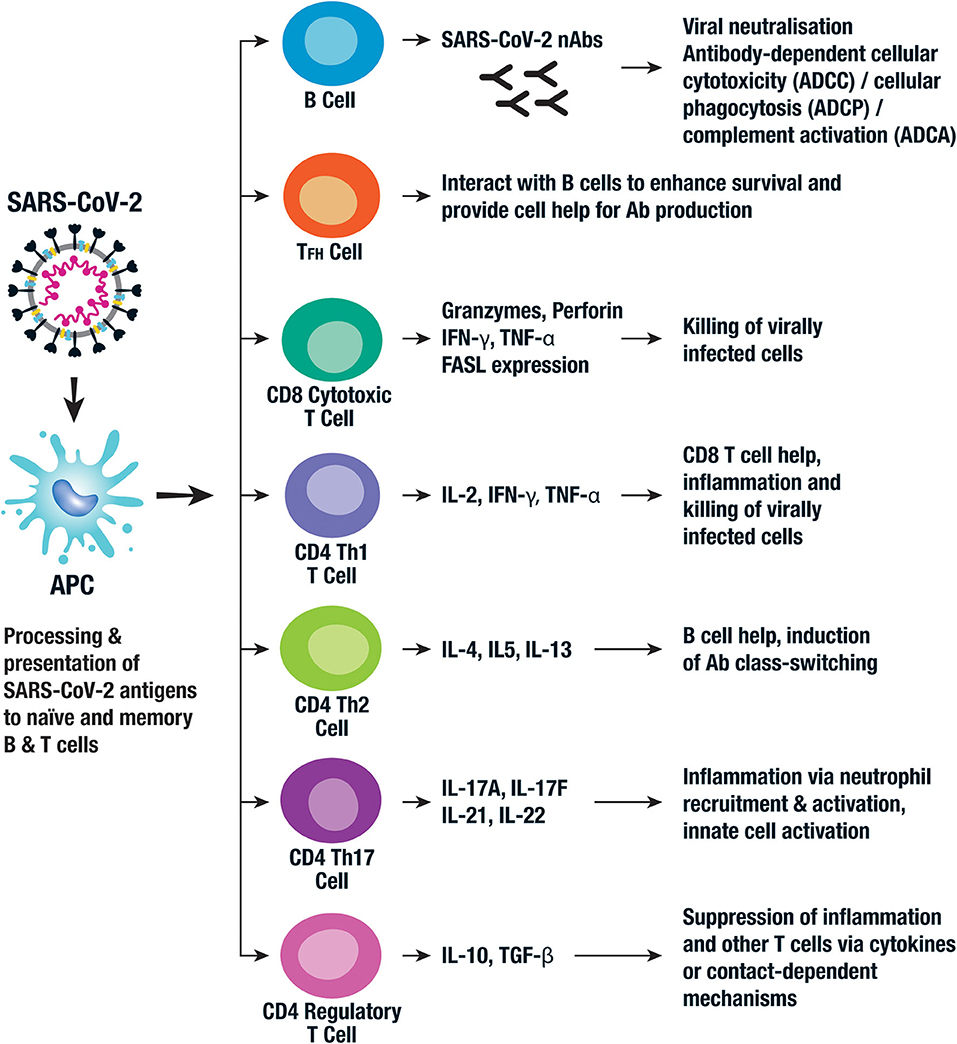 Although SARS-CoV-2 has a tropism for ACE2-expressing epithelial cells of the respiratory tract, people with severe COVID‑19 have symptoms of systemic hyperinflammation. Clinical laboratory findings of elevated IL2, IL7, IL6, granulocyte-macrophage colony-stimulating factor (GMCSF), interferon gamma-induced protein10 (IP10), monocyte chemoattractant protein1 (MCP1), macrophage inflammatory protein 1alpha (MIP1alpha), and tumour necrosis factor (TNFα) indicative of cytokine release syndrome (CRS) suggest an underlying immunopathology.
Although SARS-CoV-2 has a tropism for ACE2-expressing epithelial cells of the respiratory tract, people with severe COVID‑19 have symptoms of systemic hyperinflammation. Clinical laboratory findings of elevated IL2, IL7, IL6, granulocyte-macrophage colony-stimulating factor (GMCSF), interferon gamma-induced protein10 (IP10), monocyte chemoattractant protein1 (MCP1), macrophage inflammatory protein 1alpha (MIP1alpha), and tumour necrosis factor (TNFα) indicative of cytokine release syndrome (CRS) suggest an underlying immunopathology.serum
Serum may refer to:
* Serum (blood), plasma from which the clotting proteins have been removed
**Antiserum, blood serum with specific antibodies for passive immunity
* Serous fluid, any clear bodily fluid
*Truth serum, a drug that is likely to mak ...
biomarkers of CRS, including elevated C-reactive protein (CRP), lactate dehydrogenase (LDH), D-dimer
D-dimer (or D dimer) is a fibrin degradation product (or FDP), a small protein fragment present in the blood after a blood clot is degraded by fibrinolysis. It is so named because it contains two D fragments of the fibrin protein joined by a cros ...
, and ferritin
Ferritin is a universal intracellular protein that stores iron and releases it in a controlled fashion. The protein is produced by almost all living organisms, including archaea, bacteria, algae, higher plants, and animals. It is the primary ...
.
Systemic inflammation results in vasodilation, allowing inflammatory lymphocytic and monocytic infiltration of the lung and the heart. In particular, pathogenic GM-CSF-secreting T cells were shown to correlate with the recruitment of inflammatory IL-6-secreting monocyte
Monocytes are a type of leukocyte or white blood cell. They are the largest type of leukocyte in blood and can differentiate into macrophages and conventional dendritic cells. As a part of the vertebrate innate immune system monocytes also ...
s and severe lung pathology in people with COVID‑19. Lymphocytic infiltrates have also been reported at autopsy.
Viral and host factors
Virus proteins
 Multiple viral and host factors affect the pathogenesis of the virus. The S-protein, otherwise known as the spike protein, is the viral component that attaches to the host receptor via the
Multiple viral and host factors affect the pathogenesis of the virus. The S-protein, otherwise known as the spike protein, is the viral component that attaches to the host receptor via the ACE2
Angiotensin-converting enzyme 2 (ACE2) is an enzyme that can be found either attached to the membrane of cells (mACE2) in the intestines, kidney, testis, gallbladder, and heart or in a soluble form (sACE2). Both membrane bound and soluble ACE2 ...
receptors. It includes two subunits: S1 and S2. S1 determines the virus-host range and cellular tropism via the receptor-binding domain. S2 mediates the membrane fusion of the virus to its potential cell host via the H1 and HR2, which are heptad repeat
The heptad repeat is an example of a structural motif that consists of a repeating pattern of seven amino acids:
''a b c d e f g''
H P P H C P C
where H represents hydrophobic residues, C represents, typically, charged residues, and P repres ...
regions. Studies have shown that S1 domain induced IgG
Immunoglobulin G (Ig G) is a type of antibody. Representing approximately 75% of serum antibodies in humans, IgG is the most common type of antibody found in blood circulation. IgG molecules are created and released by plasma B cells. Each IgG an ...
and IgA Iga may refer to:
Arts and entertainment
* Ambush at Iga Pass, a 1958 Japanese film
* Iga no Kagemaru, Japanese manga series
* Iga, a set of characters from the Japanese novel '' The Kouga Ninja Scrolls''
Biology
* ''Iga'' (beetle), a g ...
antibody levels at a much higher capacity. It is the focus spike proteins expression that are involved in many effective COVID‑19 vaccines.
Host factors
Human angiotensin converting enzyme 2
Angiotensin-converting enzyme 2 (ACE2) is an enzyme that can be found either attached to the membrane of cells (mACE2) in the intestines, kidney, testis, gallbladder, and heart or in a soluble form (sACE2). Both membrane bound and soluble ACE2 ...
(hACE2) is the host factor that SARS-CoV-2 virus targets causing COVID‑19. Theoretically, the usage of angiotensin receptor blockers (ARB) and ACE inhibitors upregulating ACE2 expression might increase morbidity with COVID‑19, though animal data suggest some potential protective effect of ARB; however no clinical studies have proven susceptibility or outcomes. Until further data is available, guidelines and recommendations for hypertensive patients remain.
The effect of the virus on ACE2 cell surfaces leads to leukocytic infiltration, increased blood vessel permeability, alveolar wall permeability, as well as decreased secretion of lung surfactants. These effects cause the majority of the respiratory symptoms. However, the aggravation of local inflammation causes a cytokine storm eventually leading to a systemic inflammatory response syndrome.
Among healthy adults not exposed to SARS-CoV-2, about 35% have CD4+ T cells that recognise the SARS-CoV-2 S protein (particularly the S2 subunit) and about 50% react to other proteins of the virus, suggesting cross-reactivity from previous common colds caused by other coronaviruses.
It is unknown whether different persons use similar antibody genes in response to COVID‑19.
Host cytokine response
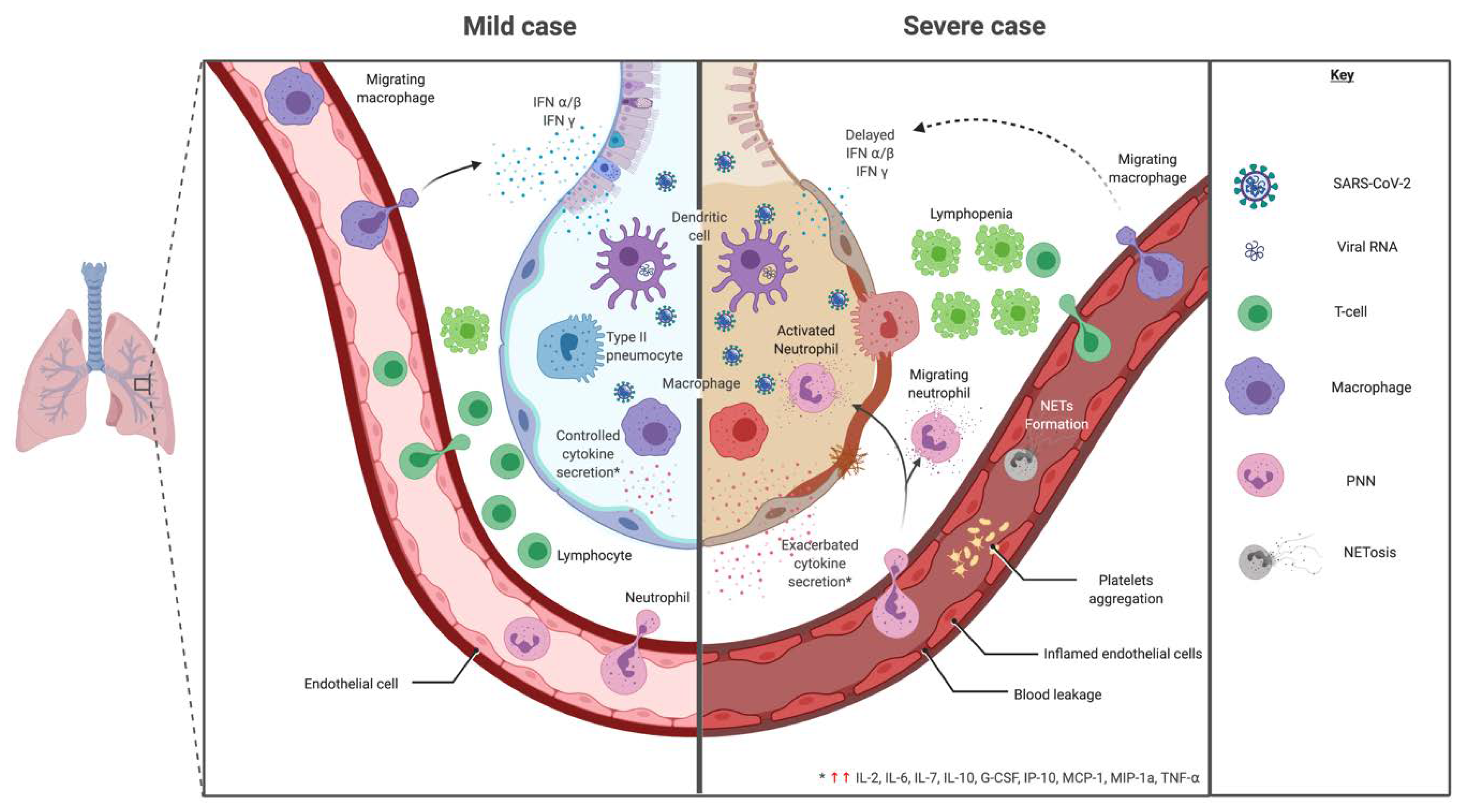 The severity of the inflammation can be attributed to the severity of what is known as the
The severity of the inflammation can be attributed to the severity of what is known as the cytokine storm
A cytokine storm, also called hypercytokinemia, is a physiological reaction in humans and other animals in which the innate immune system causes an uncontrolled and excessive release of pro-inflammatory signaling molecules called cytokines. Norma ...
.interferon-gamma
Interferon gamma (IFN-γ) is a dimerized soluble cytokine that is the only member of the type II class of interferons. The existence of this interferon, which early in its history was known as immune interferon, was described by E. F. Wheelock ...
, interferon-inducible protein 10, and monocyte chemoattractant protein1 were all associated with COVID‑19 disease severity. Treatment has been proposed to combat the cytokine storm as it remains to be one of the leading causes of morbidity and mortality in COVID‑19 disease.
A cytokine storm is due to an acute hyperinflammatory response that is responsible for clinical illness in an array of diseases but in COVID‑19, it is related to worse prognosis and increased fatality. The storm causes acute respiratory distress syndrome, blood clotting events such as strokes, myocardial infarction, encephalitis, acute kidney injury, and vasculitis. The production of IL-1, IL-2, IL-6, TNF-alpha, and interferon-gamma
Interferon gamma (IFN-γ) is a dimerized soluble cytokine that is the only member of the type II class of interferons. The existence of this interferon, which early in its history was known as immune interferon, was described by E. F. Wheelock ...
, all crucial components of normal immune responses, inadvertently become the causes of a cytokine storm. The cells of the central nervous system
The central nervous system (CNS) is the part of the nervous system consisting primarily of the brain and spinal cord. The CNS is so named because the brain integrates the received information and coordinates and influences the activity of all p ...
, the microglia, neuron
A neuron, neurone, or nerve cell is an electrically excitable cell that communicates with other cells via specialized connections called synapses. The neuron is the main component of nervous tissue in all animals except sponges and placozoa ...
s, and astrocytes, are also involved in the release of pro-inflammatory cytokines affecting the nervous system, and effects of cytokine storms toward the CNS are not uncommon.
Pregnancy response
There are many unknowns for pregnant women during the COVID-19 pandemic. Given that they are prone to have complications and severe disease infection with other types of coronaviruses, they have been identified as a vulnerable group and advised to take supplementary preventive measures.
Diagnosis
COVID‑19 can provisionally be diagnosed on the basis of symptoms and confirmed using reverse transcription polymerase chain reaction (RT-PCR) or other nucleic acid testing of infected secretions.antibodies
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of ...
produced by the body in response to the infection.
Viral testing
The standard methods of testing for presence of SARS-CoV-2 are nucleic acid tests,Public Health England
Public Health England (PHE) was an executive agency of the Department of Health and Social Care in England which began operating on 1 April 2013 to protect and improve health and wellbeing and reduce health inequalities. Its formation came as ...
and approved for use in the UK.University of Oxford
, mottoeng = The Lord is my light
, established =
, endowment = £6.1 billion (including colleges) (2019)
, budget = £2.145 billion (2019–20)
, chancellor ...
's CEBM has pointed to mounting evidence
Imaging
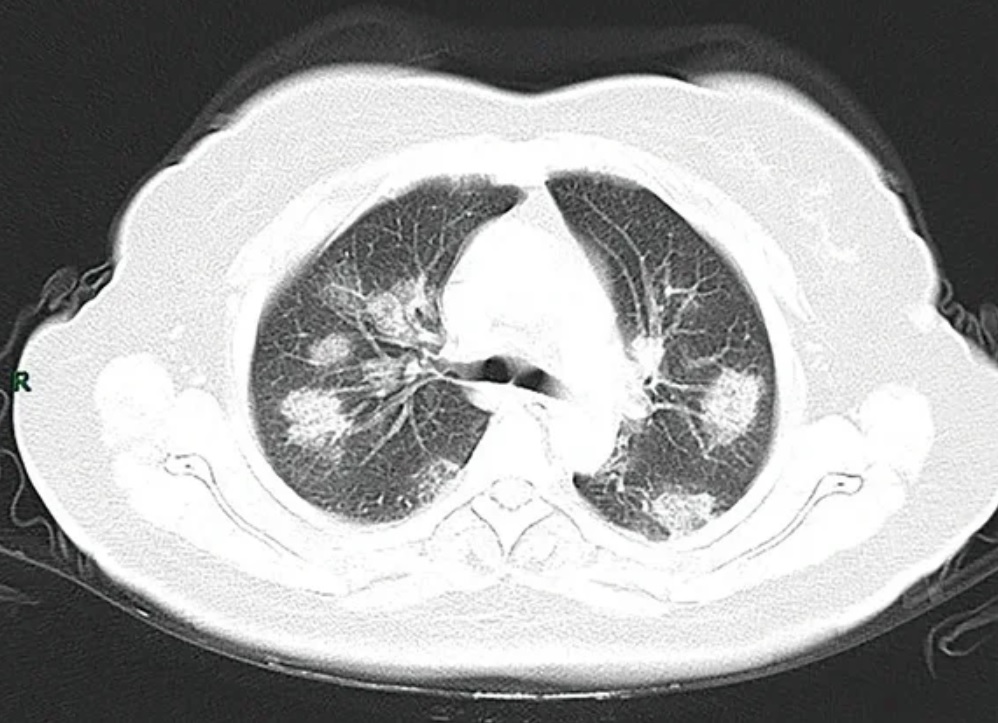

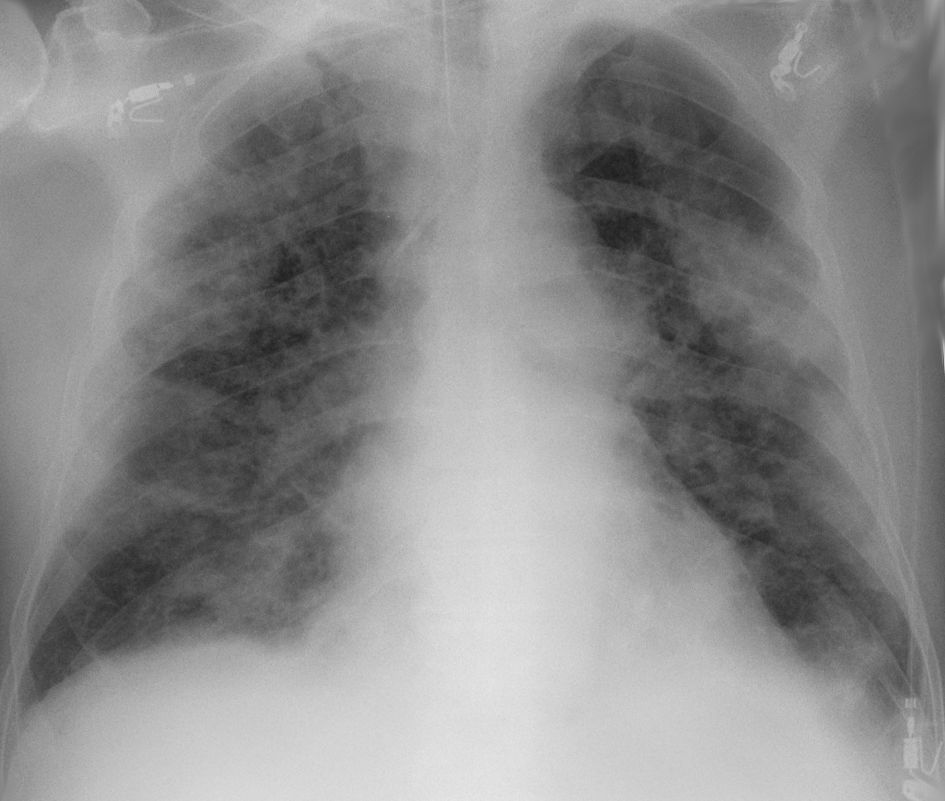 Chest CT scans may be helpful to diagnose COVID‑19 in individuals with a high clinical suspicion of infection but are not recommended for routine screening.
Chest CT scans may be helpful to diagnose COVID‑19 in individuals with a high clinical suspicion of infection but are not recommended for routine screening.COVID‑19 datasets
COVID-19 datasets are public databases for sharing case data and medical information related to the COVID-19 pandemic.
Aggregate statistics
United States
Volunteer/non-government
U.S. Department of Health & Human Services
Global
...
that include imagery such as the Italian Radiological Society which has compiled an international online database of imaging findings for confirmed cases.
Coding
In late 2019, the WHO assigned emergency ICD-10
ICD-10 is the 10th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD), a medical classification list by the World Health Organization (WHO). It contains codes for diseases, signs and symptoms ...
disease codes U07.1 for deaths from lab-confirmed SARS-CoV-2 infection and U07.2 for deaths from clinically or epidemiologically diagnosed COVID‑19 without lab-confirmed SARS-CoV-2 infection.
Pathology
The main pathological findings at autopsy are:
* Macroscopy: pericarditis, lung consolidation
A pulmonary consolidation is a region of normally compressible lung tissue that has filled with liquid instead of air. The condition is marked by induration (swelling or hardening of normally soft tissue) of a normally aerated lung. It is conside ...
and pulmonary oedemafibrin
Fibrin (also called Factor Ia) is a fibrous, non-globular protein involved in the clotting of blood. It is formed by the action of the protease thrombin on fibrinogen, which causes it to polymerize. The polymerized fibrin, together with pl ...
exudationinflammation
Inflammation (from la, inflammatio) is part of the complex biological response of body tissues to harmful stimuli, such as pathogens, damaged cells, or irritants, and is a protective response involving immune cells, blood vessels, and molec ...
with lymphocytic
A lymphocyte is a type of white blood cell (leukocyte) in the immune system of most vertebrates. Lymphocytes include natural killer cells (which function in cell-mediated, cytotoxic innate immunity), T cells (for cell-mediated, cytotoxic adapt ...
infiltration
Infiltration may refer to:
Science, medicine, and engineering
*Infiltration (hydrology), downward movement of water into soil
*Infiltration (HVAC), a heating, ventilation, and air conditioning term for air leakage into buildings
*Infiltration (me ...
and multinucleated giant cell formationBAL
Bal may refer to:
* Bal (surname), a Dutch, Indian, and Turkish surname
* Bal, Iran (disambiguation)
* Bal, Zira, a village in Punjab, India
* ''Bal'' (film), a 2010 Turkish film
* Bäl, a settlement on the Swedish island of Gotland
* 8678 B ...
* Blood and vessels: disseminated intravascular coagulation (DIC); leukoerythroblastic reaction, endotheliitis Endotheliitis is an immune response within the endothelium in blood vessels, in which they become inflamed. The condition can cause oedema of the surrounding tissue, including the stroma, and can cause irritation and pain. If it is within the corne ...
,hemophagocytosis
Hemophagocytosis is phagocytosis by histiocytes of erythrocytes, leukocytes, platelets, and their precursors in bone marrow and other tissues.
It is part of the presentation of hemophagocytic lymphohistiocytosis and Macrophage activation s ...
infarction
Infarction is tissue death ( necrosis) due to inadequate blood supply to the affected area. It may be caused by artery blockages, rupture, mechanical compression, or vasoconstriction. The resulting lesion is referred to as an infarct
(from th ...
Prevention
 Preventive measures to reduce the chances of infection include getting vaccinated, staying at home, wearing a mask in public, avoiding crowded places, keeping distance from others, ventilating indoor spaces, managing potential exposure durations, washing hands with soap and water often and for at least twenty seconds, practising good respiratory hygiene, and avoiding touching the eyes, nose, or mouth with unwashed hands.
Preventive measures to reduce the chances of infection include getting vaccinated, staying at home, wearing a mask in public, avoiding crowded places, keeping distance from others, ventilating indoor spaces, managing potential exposure durations, washing hands with soap and water often and for at least twenty seconds, practising good respiratory hygiene, and avoiding touching the eyes, nose, or mouth with unwashed hands.National Institutes of Health
The National Institutes of Health, commonly referred to as NIH (with each letter pronounced individually), is the primary agency of the United States government responsible for biomedical and public health research. It was founded in the lat ...
guidelines do not recommend any medication for prevention of COVID‑19, before or after exposure to the SARS-CoV-2 virus, outside the setting of a clinical trial.
Vaccine

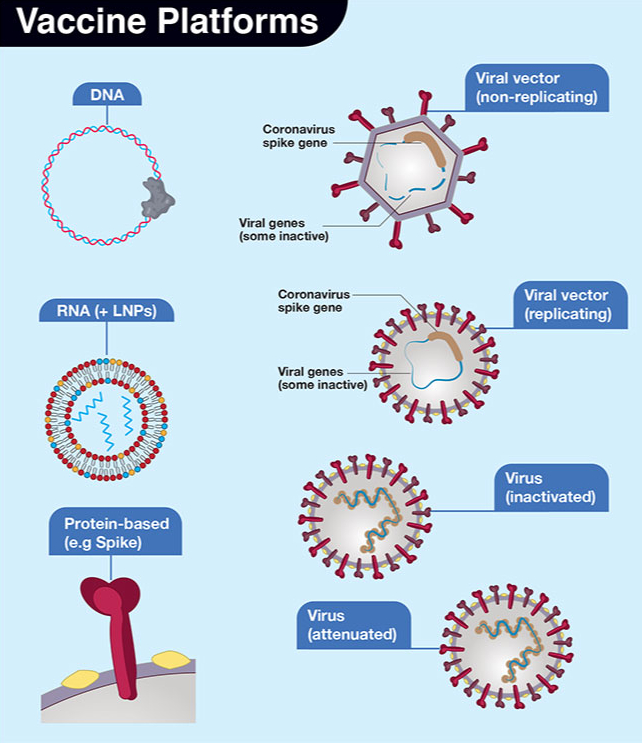
Face masks and respiratory hygiene
 The WHO and the US CDC recommend individuals wear non-medical face coverings in public settings where there is an increased risk of transmission and where social distancing measures are difficult to maintain.
The WHO and the US CDC recommend individuals wear non-medical face coverings in public settings where there is an increased risk of transmission and where social distancing measures are difficult to maintain.N95
An N95 filtering facepiece respirator, commonly abbreviated N95 respirator, is a particulate-filtering facepiece respirator that meets the U.S. National Institute for Occupational Safety and Health (NIOSH) NIOSH air filtration rating#NIOSH cl ...
or equivalent, in addition to other personal protective equipment
Personal protective equipment (PPE) is protective clothing, helmets, goggles, or other garments or equipment designed to protect the wearer's body from injury or infection. The hazards addressed by protective equipment include physical, ...
.
Indoor ventilation and avoiding crowded indoor spaces
The CDC recommends that crowded indoor spaces should be avoided.Displacement ventilation Displacement ventilation (DV) It is a room air distribution strategy where conditioned outdoor air is supplied at a low velocity from air supply diffusers located near floor level and extracted above the occupied zone, usually at ceiling height.
...
with large natural inlets can move stale air directly to the exhaust in laminar flow while significantly reducing the concentration of droplets and particles. Passive ventilation reduces energy consumption and maintenance costs but may lack controllability and heat recovery. Displacement ventilation can also be achieved mechanically with higher energy and maintenance costs. The use of large ducts and openings helps to prevent mixing in closed environments. Recirculation and mixing should be avoided because recirculation prevents dilution of harmful particles and redistributes possibly contaminated air, and mixing increases the concentration and range of infectious particles and keeps larger particles in the air.
Hand-washing and hygiene
 Thorough hand hygiene after any cough or sneeze is required.
Thorough hand hygiene after any cough or sneeze is required.hand sanitiser
Hand sanitizer (also known as hand antiseptic, hand disinfectant, hand rub, or handrub) is a liquid, gel or foam generally used to kill many viruses/ bacteria/microorganisms on the hands. In most settings, hand washing with soap and water is ge ...
with at least 60% alcohol. For areas where commercial hand sanitisers are not readily available, the WHO provides two formulations for local production. In these formulations, the antimicrobial activity arises from ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
or isopropanol. Hydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3 ...
is used to help eliminate bacterial spores in the alcohol; it is "not an active substance for hand antisepsis
An antiseptic (from Greek ἀντί ''anti'', "against" and σηπτικός ''sēptikos'', "putrefactive") is an antimicrobial substance or compound that is applied to living tissue/ skin to reduce the possibility of infection, sepsis, or pu ...
." Glycerol
Glycerol (), also called glycerine in British English and glycerin in American English, is a simple triol compound. It is a colorless, odorless, viscous liquid that is sweet-tasting and non-toxic. The glycerol backbone is found in lipids known ...
is added as a humectant.
Social distancing
Social distancing (also known as physical distancing) includes infection control actions intended to slow the spread of the disease by minimising close contact between individuals. Methods include quarantines; travel restrictions; and the closing of schools, workplaces, stadiums, theatres, or shopping centres. Individuals may apply social distancing methods by staying at home, limiting travel, avoiding crowded areas, using no-contact greetings, and physically distancing themselves from others. Many governments are mandating or recommending social distancing in regions affected by the outbreak.
Surface cleaning
After being expelled from the body, coronaviruses can survive on surfaces for hours to days. If a person touches the dirty surface, they may deposit the virus at the eyes, nose, or mouth where it can enter the body and cause infection.relative humidity
Humidity is the concentration of water vapor present in the air. Water vapor, the gaseous state of water, is generally invisible to the human eye. Humidity indicates the likelihood for precipitation, dew, or fog to be present.
Humidity dep ...
is low (<50%).ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
, 50–100 per cent isopropanol, 0.1 per cent sodium hypochlorite, 0.5 per cent hydrogen peroxide, 0.2–7.5 per cent povidone-iodine, or 50–200 ppm hypochlorous acid. Other solutions, such as benzalkonium chloride and chlorhexidine gluconate, are less effective. Ultraviolet germicidal irradiation
Ultraviolet germicidal irradiation (UVGI) is a disinfection method that uses short-wavelength ultraviolet ( ultraviolet C or UV-C) light to kill or inactivate microorganisms by destroying nucleic acids and disrupting their DNA, leaving them unab ...
may also be used,
Self-isolation
Self-isolation at home has been recommended for those diagnosed with COVID‑19 and those who suspect they have been infected. Health agencies have issued detailed instructions for proper self-isolation.
International travel-related control measures
A 2021 Cochrane rapid review found that based upon low-certainty evidence, international travel-related control measures such as restricting cross-border travel may help to contain the spread of COVID‑19.
Treatment

Prognosis and risk factors
The severity of COVID‑19 varies. The disease may take a mild course with few or no symptoms, resembling other common upper respiratory diseases such as the common cold. In 3–4% of cases (7.4% for those over age 65) symptoms are severe enough to cause hospitalisation.Tedros Adhanom
Tedros Adhanom Ghebreyesus ( ti, ቴዎድሮስ አድሓኖም ገብረኢየሱስ, sometimes spelt ti, ቴድሮስ ኣድሓኖም ገብረየሱስ, label=none; born 3 March 1965) is an Ethiopian public health official, researcher, and ...
warned that "to a significant number of people, the COVID virus poses a range of serious long-term effects." He has described the vast spectrum of COVID‑19 symptoms that fluctuate over time as "really concerning". They range from fatigue, a cough and shortness of breath, to inflammation and injury of major organsincluding the lungs and heart, and also neurological and psychologic effects. Symptoms often overlap and can affect any system in the body. Infected people have reported cyclical bouts of fatigue, headaches, months of complete exhaustion, mood swings, and other symptoms. Tedros therefore concluded that a strategy of achieving herd immunity
Herd immunity (also called herd effect, community immunity, population immunity, or mass immunity) is a form of indirect protection that applies only to contagious diseases. It occurs when a sufficient percentage of a population has become im ...
by infection, rather than vaccination, is "morally unconscionable and unfeasible".obesity
Obesity is a medical condition, sometimes considered a disease, in which excess body fat has accumulated to such an extent that it may negatively affect health. People are classified as obese when their body mass index (BMI)—a person's ...
, especially in conjunction with fatty liver disease, contributes to an increased health risk of COVID‑19.
Genetic risk factors
Genetics
Genetics is the study of genes, genetic variation, and heredity in organisms.Hartl D, Jones E (2005) It is an important branch in biology because heredity is vital to organisms' evolution. Gregor Mendel, a Moravian Augustinian friar work ...
plays an important role in the ability to fight off Covid.type I interferon
The type-I interferons (IFN) are cytokines which play essential roles in inflammation, immunoregulation, tumor cells recognition, and T-cell responses. In the human genome, a cluster of thirteen functional IFN genes is located at the 9p21.3 cyto ...
s or produce auto-antibodies against these may get much sicker from COVID‑19. Genetic screening is able to detect interferon effector genes. Some genetic variants are risk factors in specific populations. For instance, and allele
An allele (, ; ; modern formation from Greek ἄλλος ''állos'', "other") is a variation of the same sequence of nucleotides at the same place on a long DNA molecule, as described in leading textbooks on genetics and evolution.
::"The chrom ...
of the DOCK2 gene (dedicator of cytokinesis 2 gene) is a common risk factor in Asian populations but much less common in Europe. The mutation leads to lower expression of DOCK2 especially in younger patients with severe Covid. In fact, many other genes and genetic variants have been found that determine the outcome of SARS-CoV-2 infections.
Children
While very young children have experienced lower rates of infection, older children have a rate of infection that is similar to the population as a whole. Children are likely to have milder symptoms and are at lower risk of severe disease than adults.
Complications
 Complications may include
Complications may include pneumonia
Pneumonia is an inflammatory condition of the lung primarily affecting the small air sacs known as alveoli. Symptoms typically include some combination of productive or dry cough, chest pain, fever, and difficulty breathing. The severit ...
, acute respiratory distress syndrome (ARDS), multi-organ failure, septic shock, and death.thrombosis
Thrombosis (from Ancient Greek "clotting") is the formation of a blood clot inside a blood vessel, obstructing the flow of blood through the circulatory system. When a blood vessel (a vein or an artery) is injured, the body uses platelets (th ...
, particularly venous thromboembolism
Venous thrombosis is blockage of a vein caused by a thrombus (blood clot). A common form of venous thrombosis is deep vein thrombosis (DVT), when a blood clot forms in the deep veins. If a thrombus breaks off (embolizes) and flows to the lungs to ...
.elevated liver enzymes
In medicine, the presence of elevated transaminases, commonly the transaminases alanine transaminase (ALT) and aspartate transaminase (AST), may be an indicator of liver dysfunction. Other terms include transaminasemia, transaminitis, and elevated ...
, reflecting liver injury.seizure
An epileptic seizure, informally known as a seizure, is a period of symptoms due to abnormally excessive or synchronous neuronal activity in the brain. Outward effects vary from uncontrolled shaking movements involving much of the body with lo ...
, stroke, encephalitis, and Guillain–Barré syndrome (which includes loss of motor functions).paediatric multisystem inflammatory syndrome
Multisystem inflammatory syndrome in children (MIS-C), or paediatric inflammatory multisystem syndrome (PIMS / PIMS-TS), or systemic inflammatory syndrome in COVID-19 (SISCoV), is a rare systemic illness involving persistent fever and extreme i ...
, which has symptoms similar to Kawasaki disease, which can be fatal. In very rare cases, acute encephalopathy can occur, and it can be considered in those who have been diagnosed with COVID‑19 and have an altered mental status.
In the case of pregnant women, it is important to note that, according to the US Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georg ...
, pregnant women are at increased risk of becoming seriously ill from COVID‑19.premature delivery
Preterm birth, also known as premature birth, is the birth of a baby at fewer than 37 weeks gestational age, as opposed to full-term delivery at approximately 40 weeks. Extreme preterm is less than 28 weeks, very early preterm birth is between 2 ...
and intrauterine growth restriction
Intrauterine growth restriction (IUGR), or fetal growth restriction, refers to poor growth of a fetus while in the womb during pregnancy. IUGR is defined by clinical features of malnutrition and evidence of reduced growth regardless of an infant's ...
.aspergillosis
Aspergillosis is a fungal infection of usually the lungs, caused by the genus ''Aspergillus'', a common mould that is breathed in frequently from the air around, but does not usually affect most people. It generally occurs in people with lung dis ...
, candidiasis, cryptococcosis
Cryptococcosis is a potentially fatal fungal infection of mainly the lungs, presenting as a pneumonia, and brain, where it appears as a meningitis. Cough, difficulty breathing, chest pain and fever are seen when the lungs are infected. When the ...
and mucormycosis
Mucormycosis, also known as black fungus, is a serious fungal infection that comes under fulminant fungal sinusitis, usually in people who are immunocompromised. It is curable only when diagnosed early. Symptoms depend on where in the body the ...
have been recorded in patients recovering from COVID‑19.
Longer-term effects
Some early studies suggest that 10–20% of people with COVID‑19 will experience symptoms lasting longer than a month.DLCO DLCO or TLCO ( diffusing capacity or transfer factor of the lung for carbon monoxide (CO),) is the extent to which oxygen passes from the air sacs of the lungs into the blood. Commonly, it refers to the test used to determine this parameter. It was ...
, even in asymptomatic people, but with the suggestion of continuing improvement with the passing of more time.cognitive deficit
Cognitive deficit is an inclusive term to describe any characteristic that acts as a barrier to the cognition process.
The term may describe
* deficits in overall intelligence (as with intellectual disabilities),
* specific and restricted defic ...
, dementia
Dementia is a disorder which manifests as a set of related symptoms, which usually surfaces when the brain is damaged by injury or disease. The symptoms involve progressive impairments in memory, thinking, and behavior, which negatively affe ...
, psychotic disorders, and epilepsy
Epilepsy is a group of non-communicable neurological disorders characterized by recurrent epileptic seizures. Epileptic seizures can vary from brief and nearly undetectable periods to long periods of vigorous shaking due to abnormal electrica ...
or seizures persists at an increased level two years after infection.
Immunity
 The
The immune response
An immune response is a reaction which occurs within an organism for the purpose of defending against foreign invaders. These invaders include a wide variety of different microorganisms including viruses, bacteria, parasites, and fungi which coul ...
by humans to SARS-CoV-2 virus occurs as a combination of the cell-mediated immunity
Cell-mediated immunity or cellular immunity is an immune response that does not involve antibodies. Rather, cell-mediated immunity is the activation of phagocytes, antigen-specific cytotoxic T-lymphocytes, and the release of various cytokines ...
and antibody production, just as with most other infections. B cells interact with T cells and begin dividing before selection into the plasma cell, partly on the basis of their affinity for antigen. Since SARS-CoV-2 has been in the human population only since December 2019, it remains unknown if the immunity
Immunity may refer to:
Medicine
* Immunity (medical), resistance of an organism to infection or disease
* ''Immunity'' (journal), a scientific journal published by Cell Press
Biology
* Immune system
Engineering
* Radiofrequence immunity de ...
is long-lasting in people who recover from the disease.
Mortality
Several measures are commonly used to quantify mortality. These numbers vary by region and over time and are influenced by the volume of testing, healthcare system quality, treatment options, time since the initial outbreak, and population characteristics such as age, sex, and overall health.
The mortality rate
Mortality rate, or death rate, is a measure of the number of deaths (in general, or due to a specific cause) in a particular population, scaled to the size of that population, per unit of time. Mortality rate is typically expressed in units of d ...
reflects the number of deaths within a specific demographic group divided by the population of that demographic group. Consequently, the mortality rate reflects the prevalence as well as the severity of the disease within a given population. Mortality rates are highly correlated to age, with relatively low rates for young people and relatively high rates among the elderly.
Case fatality rate
The case fatality rate (CFR) reflects the number of deaths divided by the number of diagnosed cases within a given time interval. Based on Johns Hopkins University statistics, the global death-to-case ratio is (/) as of . The number varies by region.
Cumulative confirmed COVID-19 cases.svg, Total confirmed cases over time
World map of total confirmed COVID-19 cases per million people.svg, Total confirmed cases of COVID‑19 per million people
Daily and total confirmed COVID-19 deaths, World.svg, Total deaths over time
World map of total confirmed COVID-19 deaths per million people by country.svg, Total confirmed deaths due to COVID‑19 per million people
Infection fatality rate
A key metric in gauging the severity of COVID‑19 is the infection fatality rate (IFR), also referred to as the ''infection fatality ratio'' or ''infection fatality risk''. This metric is calculated by dividing the total number of deaths from the disease by the total number of infected individuals; hence, in contrast to the CFR, the IFR incorporates asymptomatic and undiagnosed infections as well as reported cases.
Estimates
 A December 2020 systematic review and meta-analysis estimated that population IFR during the first wave of the pandemic was about 0.5% to 1% in many locations (including France, Netherlands, New Zealand, and Portugal), 1% to 2% in other locations (Australia, England, Lithuania, and Spain), and exceeded 2% in Italy.
A December 2020 systematic review and meta-analysis estimated that population IFR during the first wave of the pandemic was about 0.5% to 1% in many locations (including France, Netherlands, New Zealand, and Portugal), 1% to 2% in other locations (Australia, England, Lithuania, and Spain), and exceeded 2% in Italy.[ ] Text was copied from this source, which is available under
Text was copied from this source, which is available under
Creative Commons Attribution 4.0 International License
That study also found that most of these differences in IFR reflected corresponding differences in the age composition of the population and age-specific infection rates; in particular, the metaregression estimate of IFR is very low for children and younger adults (e.g., 0.002% at age 10 and 0.01% at age 25) but increases progressively to 0.4% at age 55, 1.4% at age 65, 4.6% at age 75, and 15% at age 85.
Earlier estimates of IFR
At an early stage of the pandemic, the World Health Organization reported estimates of IFR between 0.3% and 1%. On 2July, The WHO's chief scientist reported that the average IFR estimate presented at a two-day WHO expert forum was about 0.6%.Bergamo province
The Province of Bergamo ( it, provincia di Bergamo; lmo, proìnsa de Bèrghem) is a province in the Lombardy region of Italy. It has a population of 1,112,187 (2017), an area of , and contains 243 ''comuni''. Its capital is the city of Bergamo.
...
, 0.6% of the population has died. In September 2020, the U.S. Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georg ...
(CDC) reported preliminary estimates of age-specific IFRs for public health planning purposes.
Sex differences
COVID‑19 case fatality rates are higher among men than women in most countries. However, in a few countries like India, Nepal, Vietnam, and Slovenia the fatality cases are higher in women than men.Chinese Center for Disease Control and Prevention
The Chinese Center for Disease Control and Prevention (CCDC; ) is an institution directly under the National Health Commission, based in Changping District, Beijing, China.
Established in 1983, it works to protect public health and safety ...
reported the death rate was 2.8% for men and 1.7% for women.
Ethnic differences
In the US, a greater proportion of deaths due to COVID‑19 have occurred among African Americans and other minority groups.health insurance
Health insurance or medical insurance (also known as medical aid in South Africa) is a type of insurance that covers the whole or a part of the risk of a person incurring medical expenses. As with other types of insurance, risk is shared among m ...
and care of underlying conditions such as diabetes
Diabetes, also known as diabetes mellitus, is a group of metabolic disorders characterized by a high blood sugar level ( hyperglycemia) over a prolonged period of time. Symptoms often include frequent urination, increased thirst and increased ...
,Black
Black is a color which results from the absence or complete absorption of visible light. It is an achromatic color, without hue, like white and grey. It is often used symbolically or figuratively to represent darkness. Black and white ha ...
, Asian, and other ethnic minority background. More severe impacts upon patients including the relative incidence of the necessity of hospitalisation requirements, and vulnerability to the disease has been associated via DNA analysis to be expressed in genetic variants at chromosomal region 3, features that are associated with European Neanderthal
Neanderthals (, also ''Homo neanderthalensis'' and erroneously ''Homo sapiens neanderthalensis''), also written as Neandertals, are an Extinction, extinct species or subspecies of archaic humans who lived in Eurasia until about 40,000 years ag ...
heritage. That structure imposes greater risks that those affected will develop a more severe form of the disease.
Comorbidities
Biological factors (immune response) and the general behaviour (habits) can strongly determine the consequences of COVID‑19.diabetes mellitus
Diabetes, also known as diabetes mellitus, is a group of metabolic disorders characterized by a high blood sugar level ( hyperglycemia) over a prolonged period of time. Symptoms often include frequent urination, increased thirst and increased ...
,comorbidity
In medicine, comorbidity - from Latin morbus ("sickness"), co ("together"), -ity (as if - several sicknesses together) - is the presence of one or more additional conditions often co-occurring (that is, concomitant or concurrent) with a primary ...
with the average person having 3.4 diseases.Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georg ...
(CDC), are: moderate or severe asthma
Asthma is a long-term inflammatory disease of the airways of the lungs. It is characterized by variable and recurring symptoms, reversible airflow obstruction, and easily triggered bronchospasms. Symptoms include episodes of wheezing, co ...
, pre-existing COPD, pulmonary fibrosis, cystic fibrosis. Evidence stemming from meta-analysis
A meta-analysis is a statistical analysis that combines the results of multiple scientific studies. Meta-analyses can be performed when there are multiple scientific studies addressing the same question, with each individual study reporting m ...
of several smaller research papers also suggests that smoking can be associated with worse outcomes. When someone with existing respiratory problems is infected with COVID‑19, they might be at greater risk for severe symptoms. COVID‑19 also poses a greater risk to people who misuse opioids and amphetamines
Substituted amphetamines are a class of compounds based upon the amphetamine structure; it includes all derivative compounds which are formed by replacing, or substituting, one or more hydrogen atoms in the amphetamine core structure with sub ...
, insofar as their drug use may have caused lung damage.
In August 2020, the CDC issued a caution that tuberculosis
Tuberculosis (TB) is an infectious disease usually caused by '' Mycobacterium tuberculosis'' (MTB) bacteria. Tuberculosis generally affects the lungs, but it can also affect other parts of the body. Most infections show no symptoms, ...
(TB) infections could increase the risk of severe illness or death. The WHO recommended that people with respiratory symptoms be screened for both diseases, as testing positive for COVID‑19 could not rule out co-infections. Some projections have estimated that reduced TB detection due to the pandemic could result in 6.3 million additional TB cases and 1.4 million TB-related deaths by 2025.
History
The virus is thought to be of natural animal origin, most likely through spillover infection.World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level o ...
indicated that the virus descended from a coronavirus that infects wild bats, and likely spread to humans through an intermediary wildlife host. There are several theories about where the index case originated and investigations into the origin of the pandemic are ongoing.Science
Science is a systematic endeavor that builds and organizes knowledge in the form of testable explanations and predictions about the universe.
Science may be as old as the human species, and some of the earliest archeological evidence ...
, virus transmission into humans occurred through two spillover events in November 2019 and was likely due to live wildlife trade on the Huanan wet market in the city of Wuhan (Hubei, China). Doubts about the conclusions have mostly centred on the precise site of spillover. Earlier phylogenetics
In biology, phylogenetics (; from Greek φυλή/ φῦλον [] "tribe, clan, race", and wikt:γενετικός, γενετικός [] "origin, source, birth") is the study of the evolutionary history and relationships among or within groups ...
estimated that SARS-CoV-2 arose in October or November 2019.Guangdong
Guangdong (, ), alternatively romanized as Canton or Kwangtung, is a coastal province in South China on the north shore of the South China Sea. The capital of the province is Guangzhou. With a population of 126.01 million (as of 2020 ...
before Wuhan. U.S intelligence agencies and other scientists have found that the virus may have been unintentionally leaked from a laboratory such as the Wuhan Institute of Virology, but that it was not developed as a biological weapon and is unlikely to have been genetically engineered.
The first confirmed human infections were in Wuhan. A study of the first 41 cases of confirmed COVID‑19, published in January 2020 in ''The Lancet'', reported the earliest date of onset of symptoms as 1December 2019.Milan
Milan ( , , Lombard: ; it, Milano ) is a city in northern Italy, capital of Lombardy, and the second-most populous city proper in Italy after Rome. The city proper has a population of about 1.4 million, while its metropolitan city ...
and Turin
Turin ( , Piedmontese language, Piedmontese: ; it, Torino ) is a city and an important business and cultural centre in Northern Italy. It is the capital city of Piedmont and of the Metropolitan City of Turin, and was the first Italian capital ...
, Italy, on 18 December 2019.
By December 2019, the spread of infection was almost entirely driven by human-to-human transmission.bronchoalveolar lavage fluid
Bronchoalveolar lavage (BAL) (also known as bronchoalveolar washing) is a diagnostic method of the lower respiratory system in which a bronchoscope is passed through the mouth or nose into an appropriate airway in the lungs, with a measured amount ...
(BAL) sample from an unresolved clinical case to sequencing company Vision Medicals. On 27 and 28 December, Vision Medicals informed the Wuhan Central Hospital and the Chinese CDC of the results of the test, showing a new coronavirus.Shenzhen
Shenzhen (; ; ; ), also historically known as Sham Chun, is a major sub-provincial city and one of the special economic zones of China. The city is located on the east bank of the Pearl River estuary on the central coast of southern provi ...
.Reuters
Reuters ( ) is a news agency owned by Thomson Reuters Corporation. It employs around 2,500 journalists and 600 photojournalists in about 200 locations worldwide. Reuters is one of the largest news agencies in the world.
The agency was est ...
'' reported that it had estimated the worldwide total number of deaths due to COVID‑19 to have exceeded five million.
Misinformation
After the initial outbreak of COVID‑19, misinformation
Misinformation is incorrect or misleading information. It differs from disinformation, which is ''deliberately'' deceptive. Rumors are information not attributed to any particular source, and so are unreliable and often unverified, but can turn ...
and disinformation
Disinformation is false information deliberately spread to deceive people. It is sometimes confused with misinformation, which is false information but is not deliberate.
The English word ''disinformation'' comes from the application of the ...
regarding the origin, scale, prevention, treatment, and other aspects of the disease rapidly spread online.Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georg ...
(CDC) published preliminary estimates of the risk of death by age groups in the United States, but those estimates were widely misreported and misunderstood.
Other species
Humans appear to be capable of spreading the virus to some other animals, a type of disease transmission referred to as zooanthroponosis.
Some pets, especially cats and ferrets, can catch this virus from infected humans.great ape
The Hominidae (), whose members are known as the great apes or hominids (), are a taxonomic family of primates that includes eight extant species in four genera: '' Pongo'' (the Bornean, Sumatran and Tapanuli orangutan); ''Gorilla'' (the ...
species such as orangutan
Orangutans are great apes native to the rainforests of Indonesia and Malaysia. They are now found only in parts of Borneo and Sumatra, but during the Pleistocene they ranged throughout Southeast Asia and South China. Classified in the genu ...
s can also be infected with the COVID‑19 virus.Denmark
)
, song = ( en, "King Christian stood by the lofty mast")
, song_type = National and royal anthem
, image_map = EU-Denmark.svg
, map_caption =
, subdivision_type = Sovereign state
, subdivision_name = Kingdom of Denmark
, establish ...
, a major producer of mink pelts, ordered the slaughter of all minks over fears of viral mutations,
Research
International research on vaccines and medicines in COVID‑19 is underway by government organisations, academic groups, and industry researchers.clinical trial
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments (such as novel vaccines, drugs, diet ...
s have been undertaken, with research happening on every continent except Antarctica
Antarctica () is Earth's southernmost and least-populated continent. Situated almost entirely south of the Antarctic Circle and surrounded by the Southern Ocean, it contains the geographic South Pole. Antarctica is the fifth-largest cont ...
. , more than 200 possible treatments have been studied in humans.
Transmission and prevention research
Modelling research has been conducted with several objectives, including predictions of the dynamics of transmission, diagnosis and prognosis of infection, estimation of the impact of interventions, or allocation of resources. Modelling studies are mostly based on compartmental models in epidemiology, estimating the number of infected people over time under given conditions. Several other types of models have been developed and used during the COVID‑19 including computational fluid dynamics
Computational fluid dynamics (CFD) is a branch of fluid mechanics that uses numerical analysis and data structures to analyze and solve problems that involve fluid flows. Computers are used to perform the calculations required to simulate ...
models to study the flow physics of COVID‑19, retrofits of crowd movement models to study occupant exposure, mobility-data based models to investigate transmission, or the use of macroeconomic
Macroeconomics (from the Greek prefix ''makro-'' meaning "large" + ''economics'') is a branch of economics dealing with performance, structure, behavior, and decision-making of an economy as a whole.
For example, using interest rates, taxes, an ...
models to assess the economic impact of the pandemic. Further, conceptual frameworks from crisis management research have been applied to better understand the effects of COVID‑19 on organisations worldwide.
Treatment-related research
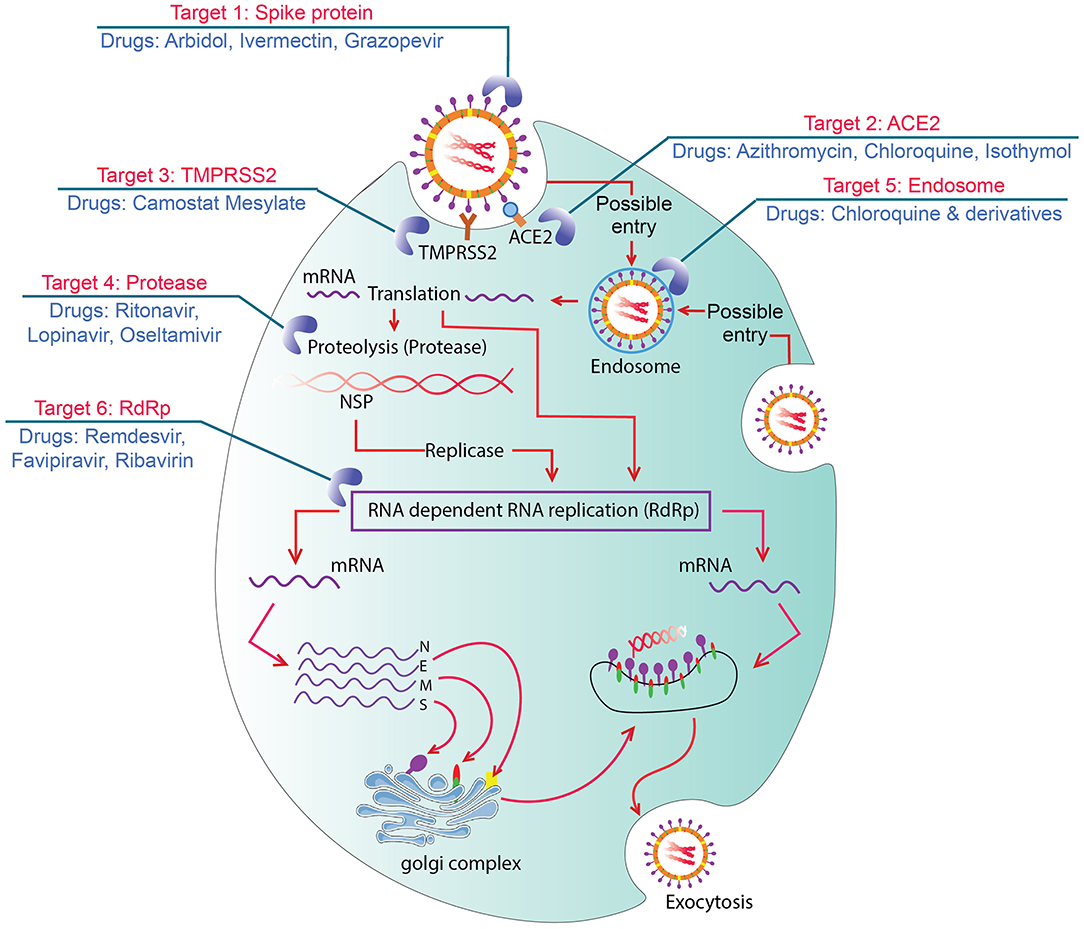 Repurposed antiviral drugs make up most of the research into COVID‑19 treatments.
Repurposed antiviral drugs make up most of the research into COVID‑19 treatments.bevacizumab
Bevacizumab, sold under the brand name Avastin among others, is a medication used to treat a number of types of cancers and a specific eye disease. For cancer, it is given by slow injection into a vein (intravenous) and used for colon cancer, ...
, and recombinant angiotensin-converting enzyme 2.World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level o ...
(WHO) initiated the Solidarity trial to assess the treatment effects of some promising drugs: an experimental drug called remdesivir; anti-malarial
Antimalarial medications or simply antimalarials are a type of antiparasitic chemical agent, often natural product, naturally derived, that can be used to treat or to prevent malaria, in the latter case, most often aiming at two susceptible target ...
drugs chloroquine and hydroxychloroquine; two anti-HIV drugs, lopinavir/ritonavir; and interferon-beta
The type-I interferons (IFN) are cytokines which play essential roles in inflammation, immunoregulation, tumor cells recognition, and T-cell responses. In the human genome, a cluster of thirteen functional IFN genes is located at the 9p21.3 cyto ...
.RECOVERY Trial
The Randomised Evaluation of COVID-19 Therapy (RECOVERY Trial) is a large-enrollment clinical trial of possible treatments for people in the United Kingdom admitted to hospital with severe COVID-19 infection. The trial was later expanded to In ...
in the United Kingdom showed that dexamethasone reduced mortality by one third for people who are critically ill on ventilators and one fifth for those receiving supplemental oxygen. Because this is a well-tested and widely available treatment, it was welcomed by the WHO, which is in the process of updating treatment guidelines to include dexamethasone and other steroids. Based on those preliminary results, dexamethasone treatment has been recommended by the NIH for patients with COVID‑19 who are mechanically ventilated or who require supplemental oxygen but not in patients with COVID‑19 who do not require supplemental oxygen.
In September 2020, the WHO released updated guidance on using corticosteroids for COVID‑19.European Medicines Agency
The European Medicines Agency (EMA) is an agency of the European Union (EU) in charge of the evaluation and supervision of medicinal products. Prior to 2004, it was known as the European Agency for the Evaluation of Medicinal Products or Eur ...
(EMA) endorsed the use of dexamethasone in adults and adolescents from twelve years of age and weighing at least who require supplemental oxygen therapy.[ Text was copied from this source which is European Medicines Agency. Reproduction is authorized provided the source is acknowledged.] Dexamethasone can be taken by mouth or given as an injection or infusion (drip) into a vein.Food and Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a federal agency of the Department of Health and Human Services. The FDA is responsible for protecting and promoting public health through the control and supervision of food ...
(FDA) issued an emergency use authorization for the investigational monoclonal antibody therapy bamlanivimab
Bamlanivimab is a monoclonal antibody developed by AbCellera Biologics and Eli Lilly as a treatment for COVID-19. The medication was granted an emergency use authorization (EUA) by the US Food and Drug Administration (FDA) in November 2020, an ...
for the treatment of mild-to-moderate COVID‑19.etesevimab
Bamlanivimab/etesevimab is a combination of two monoclonal antibodies, bamlanivimab and etesevimab, administered together via intravenous infusion as a treatment for COVID-19. Both types of antibody target the surface spike protein of SARS� ...
administered together for the treatment of mild to moderate COVID‑19 in people twelve years of age or older weighing at least who test positive for SARS‑CoV‑2 and who are at high risk for progressing to severe COVID‑19. The authorised use includes treatment for those who are 65 years of age or older or who have certain chronic medical conditions.
Cytokine storm
 A
A cytokine storm
A cytokine storm, also called hypercytokinemia, is a physiological reaction in humans and other animals in which the innate immune system causes an uncontrolled and excessive release of pro-inflammatory signaling molecules called cytokines. Norma ...
can be a complication in the later stages of severe COVID‑19. A cytokine storm is a potentially deadly immune reaction where a large amount of pro-inflammatory cytokines and chemokine
Chemokines (), or chemotactic cytokines, are a family of small cytokines or signaling proteins secreted by cells that induce directional movement of leukocytes, as well as other cell types, including endothelial and epithelial cells. In additi ...
s are released too quickly. A cytokine storm can lead to ARDS and multiple organ failure. Data collected from Jin Yin-tan Hospital in Wuhan, China indicates that patients who had more severe responses to COVID‑19 had greater amounts of pro-inflammatory cytokines and chemokines in their system than patients who had milder responses. These high levels of pro-inflammatory cytokines and chemokines indicate presence of a cytokine storm.
Tocilizumab has been included in treatment guidelines by China's National Health Commission after a small study was completed.[Various sources:
*
*
* ] The interleukin-6 receptor (IL-6R) antagonist was approved by the FDA to undergo a PhaseIII clinical trial assessing its effectiveness on COVID‑19 based on retrospective case studies for the treatment of steroid-refractory cytokine release syndrome induced by a different cause, CAR T cell therapy
A therapy or medical treatment (often abbreviated tx, Tx, or Tx) is the attempted remediation of a health problem, usually following a medical diagnosis.
As a rule, each therapy has indications and contraindications. There are many differe ...
, in 2017.Lenzilumab
Lenzilumab (INN; development code KB003) is a humanized monoclonal antibody (class IgG1 kappa) that targets colony stimulating factor 2 (CSF2)/granulocyte-macrophage colony stimulating factor (GM-CSF).
Pre-clinical evidence and clinical data imp ...
, an anti-GM-CSF monoclonal antibody, is protective in murine models for CAR T cell-induced CRS and neurotoxicity and is a viable therapeutic option due to the observed increase of pathogenic GM-CSF secreting Tcells in hospitalised patients with COVID‑19.
Passive antibodies
 Transferring purified and concentrated
Transferring purified and concentrated antibodies
An antibody (Ab), also known as an immunoglobulin (Ig), is a large, Y-shaped protein used by the immune system to identify and neutralize foreign objects such as pathogenic bacteria and viruses. The antibody recognizes a unique molecule of ...
produced by the immune systems of those who have recovered from COVID‑19 to people who need them is being investigated as a non-vaccine method of passive immunisation.convalescent serum
Antiserum is a blood serum containing monoclonal or polyclonal antibodies that is used to spread passive immunity to many diseases via blood donation (plasmapheresis). For example, convalescent serum, passive antibody transfusion from a previous ...
, which consists of the liquid portion of the blood from people who recovered from the infection and contains antibodies specific to this virus, which is then administered to active patients.
Bioethics
Since the outbreak of the COVID‑19 pandemic, scholars have explored the bioethics
Bioethics is both a field of study and professional practice, interested in ethical issues related to health (primarily focused on the human, but also increasingly includes animal ethics), including those emerging from advances in biology, me ...
, normative economics, and political theories
Political philosophy or political theory is the philosophical study of government, addressing questions about the nature, scope, and legitimacy of public agents and institutions and the relationships between them. Its topics include politics ...
of healthcare policies related to the public health crisis. Academics have pointed to the moral distress of healthcare workers, ethics of distributing scarce healthcare resources such as ventilators, and the global justice of vaccine diplomacies. The socio-economic inequalities between genders, races, groups with disabilities, communities, regions, countries, and continents have also drawn attention in academia and the general public.
Effects on other diseases
The use of social distancing and the wearing of surgical masks and similar precautions against COVID‑19 may have caused a drop in the spread of the common cold and the flu.
See also
* Coronavirus diseases, a group of closely related syndromes
* Disease X, a WHO term
*
*
References
Further reading
* Scholia
Scholia (singular scholium or scholion, from grc, σχόλιον, "comment, interpretation") are grammatical, critical, or explanatory comments – original or copied from prior commentaries – which are inserted in the margin of t ...
Q104287299.
*
*
External links
Health agencies
Coronavirus disease (COVID‑19)Facts
by the World Health Organization
The World Health Organization (WHO) is a specialized agency of the United Nations responsible for international public health. The WHO Constitution states its main objective as "the attainment by all peoples of the highest possible level o ...
(WHO)
Coronavirus 2019 (COVID-19)
by the US Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is the national public health agency of the United States. It is a United States federal agency, under the Department of Health and Human Services, and is headquartered in Atlanta, Georg ...
(CDC)
Coronavirus (COVID‑19)
by the UK National Health Service
The National Health Service (NHS) is the umbrella term for the publicly funded healthcare systems of the United Kingdom (UK). Since 1948, they have been funded out of general taxation. There are three systems which are referred to using the " ...
(NHS)
Directories
Coronavirus Resource Center
at the Center for Inquiry
*
COVID‑19 Resource Directory on OpenMD
Medical journals
Coronavirus Disease 2019 (COVID‑19)
by '' JAMA''
BMJ's Coronavirus (covid‑19) Hub
by the BMJ
Novel Coronavirus Information Center
by Elsevier
Elsevier () is a Dutch academic publishing company specializing in scientific, technical, and medical content. Its products include journals such as '' The Lancet'', '' Cell'', the ScienceDirect collection of electronic journals, '' Trends'', ...
COVID‑19 Resource Centre
by '' The Lancet''
Coronavirus (COVID‑19) Research Highlights
by Springer Nature
Coronavirus (Covid‑19)
by ''The New England Journal of Medicine
''The New England Journal of Medicine'' (''NEJM'') is a weekly medical journal published by the Massachusetts Medical Society. It is among the most prestigious peer-reviewed medical journals as well as the oldest continuously published one.
H ...
''
Covid‑19: Novel Coronavirus
by Wiley Publishing
Treatment guidelines
*
*
*
*
*
*
{{Authority control
Occupational safety and health
Vaccine-preventable diseases
Viral respiratory tract infections
Zoonoses
Public health
Coronavirus-associated diseases

 The SARS-CoV-2 virus can infect a wide range of cells and systems of the body. COVID‑19 is most known for affecting the upper respiratory tract (sinuses, nose, and throat) and the lower respiratory tract (windpipe and lungs). The lungs are the organs most affected by COVID‑19 because the virus accesses host cells via the
The SARS-CoV-2 virus can infect a wide range of cells and systems of the body. COVID‑19 is most known for affecting the upper respiratory tract (sinuses, nose, and throat) and the lower respiratory tract (windpipe and lungs). The lungs are the organs most affected by COVID‑19 because the virus accesses host cells via the  Research conducted when Alpha was the dominant variant has suggested COVID-19 may cause brain damage. It is unknown if such damage is temporary or permanent, and whether Omicron has similar effects. Observed individuals infected with COVID-19 (most with mild cases) experienced an additional 0.2% to 2% of brain tissue lost in regions of the brain connected to the sense of smell compared with uninfected individuals, and the overall effect on the brain was equivalent on average to at least one extra year of normal ageing; infected individuals also scored lower on several cognitive tests. All effects were more pronounced among older ages.
Research conducted when Alpha was the dominant variant has suggested COVID-19 may cause brain damage. It is unknown if such damage is temporary or permanent, and whether Omicron has similar effects. Observed individuals infected with COVID-19 (most with mild cases) experienced an additional 0.2% to 2% of brain tissue lost in regions of the brain connected to the sense of smell compared with uninfected individuals, and the overall effect on the brain was equivalent on average to at least one extra year of normal ageing; infected individuals also scored lower on several cognitive tests. All effects were more pronounced among older ages.
 Although SARS-CoV-2 has a tropism for ACE2-expressing epithelial cells of the respiratory tract, people with severe COVID‑19 have symptoms of systemic hyperinflammation. Clinical laboratory findings of elevated IL2, IL7, IL6, granulocyte-macrophage colony-stimulating factor (GMCSF), interferon gamma-induced protein10 (IP10), monocyte chemoattractant protein1 (MCP1), macrophage inflammatory protein 1alpha (MIP1alpha), and tumour necrosis factor (TNFα) indicative of cytokine release syndrome (CRS) suggest an underlying immunopathology.
Interferon alpha plays a complex, Janus-faced role in the pathogenesis of COVID-19. Although it promotes the elimination of virus-infected cells, it also upregulates the expression of ACE-2, thereby facilitating the SARS-Cov2 virus to enter cells and to replicate. A competition of negative feedback loops (via protective effects of interferon alpha) and positive feedback loops (via upregulation of ACE-2) is assumed to determine the fate of patients suffering from COVID-19.
Additionally, people with COVID‑19 and acute respiratory distress syndrome (ARDS) have classical
Although SARS-CoV-2 has a tropism for ACE2-expressing epithelial cells of the respiratory tract, people with severe COVID‑19 have symptoms of systemic hyperinflammation. Clinical laboratory findings of elevated IL2, IL7, IL6, granulocyte-macrophage colony-stimulating factor (GMCSF), interferon gamma-induced protein10 (IP10), monocyte chemoattractant protein1 (MCP1), macrophage inflammatory protein 1alpha (MIP1alpha), and tumour necrosis factor (TNFα) indicative of cytokine release syndrome (CRS) suggest an underlying immunopathology.
Interferon alpha plays a complex, Janus-faced role in the pathogenesis of COVID-19. Although it promotes the elimination of virus-infected cells, it also upregulates the expression of ACE-2, thereby facilitating the SARS-Cov2 virus to enter cells and to replicate. A competition of negative feedback loops (via protective effects of interferon alpha) and positive feedback loops (via upregulation of ACE-2) is assumed to determine the fate of patients suffering from COVID-19.
Additionally, people with COVID‑19 and acute respiratory distress syndrome (ARDS) have classical  Multiple viral and host factors affect the pathogenesis of the virus. The S-protein, otherwise known as the spike protein, is the viral component that attaches to the host receptor via the
Multiple viral and host factors affect the pathogenesis of the virus. The S-protein, otherwise known as the spike protein, is the viral component that attaches to the host receptor via the  The severity of the inflammation can be attributed to the severity of what is known as the
The severity of the inflammation can be attributed to the severity of what is known as the 

 Chest CT scans may be helpful to diagnose COVID‑19 in individuals with a high clinical suspicion of infection but are not recommended for routine screening. Bilateral multilobar ground-glass opacities with a peripheral, asymmetric, and posterior distribution are common in early infection. Subpleural dominance, crazy paving (lobular septal thickening with variable alveolar filling), and consolidation may appear as the disease progresses. Characteristic imaging features on chest radiographs and computed tomography (CT) of people who are symptomatic include asymmetric peripheral ground-glass opacities without pleural effusions.
Many groups have created
Chest CT scans may be helpful to diagnose COVID‑19 in individuals with a high clinical suspicion of infection but are not recommended for routine screening. Bilateral multilobar ground-glass opacities with a peripheral, asymmetric, and posterior distribution are common in early infection. Subpleural dominance, crazy paving (lobular septal thickening with variable alveolar filling), and consolidation may appear as the disease progresses. Characteristic imaging features on chest radiographs and computed tomography (CT) of people who are symptomatic include asymmetric peripheral ground-glass opacities without pleural effusions.
Many groups have created  Preventive measures to reduce the chances of infection include getting vaccinated, staying at home, wearing a mask in public, avoiding crowded places, keeping distance from others, ventilating indoor spaces, managing potential exposure durations, washing hands with soap and water often and for at least twenty seconds, practising good respiratory hygiene, and avoiding touching the eyes, nose, or mouth with unwashed hands.
Those diagnosed with COVID‑19 or who believe they may be infected are advised by the CDC to stay home except to get medical care, call ahead before visiting a healthcare provider, wear a face mask before entering the healthcare provider's office and when in any room or vehicle with another person, cover coughs and sneezes with a tissue, regularly wash hands with soap and water and avoid sharing personal household items.
The first COVID‑19 vaccine was granted regulatory approval on 2December 2020 by the UK medicines regulator MHRA. It was evaluated for emergency use authorization (EUA) status by the US FDA, and in several other countries. Initially, the US
Preventive measures to reduce the chances of infection include getting vaccinated, staying at home, wearing a mask in public, avoiding crowded places, keeping distance from others, ventilating indoor spaces, managing potential exposure durations, washing hands with soap and water often and for at least twenty seconds, practising good respiratory hygiene, and avoiding touching the eyes, nose, or mouth with unwashed hands.
Those diagnosed with COVID‑19 or who believe they may be infected are advised by the CDC to stay home except to get medical care, call ahead before visiting a healthcare provider, wear a face mask before entering the healthcare provider's office and when in any room or vehicle with another person, cover coughs and sneezes with a tissue, regularly wash hands with soap and water and avoid sharing personal household items.
The first COVID‑19 vaccine was granted regulatory approval on 2December 2020 by the UK medicines regulator MHRA. It was evaluated for emergency use authorization (EUA) status by the US FDA, and in several other countries. Initially, the US 

 The WHO and the US CDC recommend individuals wear non-medical face coverings in public settings where there is an increased risk of transmission and where social distancing measures are difficult to maintain. This recommendation is meant to reduce the spread of the disease by asymptomatic and pre-symptomatic individuals and is complementary to established preventive measures such as social distancing. Face coverings limit the volume and travel distance of expiratory droplets dispersed when talking, breathing, and coughing. A face covering without vents or holes will also filter out particles containing the virus from inhaled and exhaled air, reducing the chances of infection. However, if the mask includes an exhalation valve, a wearer that is infected (and possibly asymptomatic) may transmit the virus through the valve. Many countries and local jurisdictions encourage or mandate the use of face masks or cloth face coverings by members of the public to limit the spread of the virus.
Masks are also strongly recommended for those who may have been infected and those taking care of someone who may have the disease. When not wearing a mask, the CDC recommends covering the mouth and nose with a tissue when coughing or sneezing and recommends using the inside of the elbow if no tissue is available. Proper hand hygiene after any cough or sneeze is encouraged. Healthcare professionals interacting directly with people who have COVID‑19 are advised to use respirators at least as protective as NIOSH-certified
The WHO and the US CDC recommend individuals wear non-medical face coverings in public settings where there is an increased risk of transmission and where social distancing measures are difficult to maintain. This recommendation is meant to reduce the spread of the disease by asymptomatic and pre-symptomatic individuals and is complementary to established preventive measures such as social distancing. Face coverings limit the volume and travel distance of expiratory droplets dispersed when talking, breathing, and coughing. A face covering without vents or holes will also filter out particles containing the virus from inhaled and exhaled air, reducing the chances of infection. However, if the mask includes an exhalation valve, a wearer that is infected (and possibly asymptomatic) may transmit the virus through the valve. Many countries and local jurisdictions encourage or mandate the use of face masks or cloth face coverings by members of the public to limit the spread of the virus.
Masks are also strongly recommended for those who may have been infected and those taking care of someone who may have the disease. When not wearing a mask, the CDC recommends covering the mouth and nose with a tissue when coughing or sneezing and recommends using the inside of the elbow if no tissue is available. Proper hand hygiene after any cough or sneeze is encouraged. Healthcare professionals interacting directly with people who have COVID‑19 are advised to use respirators at least as protective as NIOSH-certified  Thorough hand hygiene after any cough or sneeze is required. The WHO also recommends that individuals wash hands often with soap and water for at least twenty seconds, especially after going to the toilet or when hands are visibly dirty, before eating and after blowing one's nose. When soap and water are not available, the CDC recommends using an alcohol-based
Thorough hand hygiene after any cough or sneeze is required. The WHO also recommends that individuals wash hands often with soap and water for at least twenty seconds, especially after going to the toilet or when hands are visibly dirty, before eating and after blowing one's nose. When soap and water are not available, the CDC recommends using an alcohol-based 
 Complications may include
Complications may include  The
The  A December 2020 systematic review and meta-analysis estimated that population IFR during the first wave of the pandemic was about 0.5% to 1% in many locations (including France, Netherlands, New Zealand, and Portugal), 1% to 2% in other locations (Australia, England, Lithuania, and Spain), and exceeded 2% in Italy.
A December 2020 systematic review and meta-analysis estimated that population IFR during the first wave of the pandemic was about 0.5% to 1% in many locations (including France, Netherlands, New Zealand, and Portugal), 1% to 2% in other locations (Australia, England, Lithuania, and Spain), and exceeded 2% in Italy.  Repurposed antiviral drugs make up most of the research into COVID‑19 treatments. Other candidates in trials include vasodilators, corticosteroids, immune therapies, lipoic acid,
Repurposed antiviral drugs make up most of the research into COVID‑19 treatments. Other candidates in trials include vasodilators, corticosteroids, immune therapies, lipoic acid,  A
A  Transferring purified and concentrated
Transferring purified and concentrated