Condensed formula on:
[Wikipedia]
[Google]
[Amazon]
 The structural formula of a
The structural formula of a


 Electrons are usually shown as colored in circles. One circle indicates one electron. Two circles indicate a pair of electrons. Typically, a pair of electrons will also indicate a negative charge. By using the colored circles, the number of electrons in the valence shell of each respective atom is indicated providing further descriptive information regarding the reactive capacity of that atom in the molecule.
Electrons are usually shown as colored in circles. One circle indicates one electron. Two circles indicate a pair of electrons. Typically, a pair of electrons will also indicate a negative charge. By using the colored circles, the number of electrons in the valence shell of each respective atom is indicated providing further descriptive information regarding the reactive capacity of that atom in the molecule.


 Wavy single bonds represent unknown or unspecified stereochemistry or a mixture of isomers. For example, the above diagram shows the
Wavy single bonds represent unknown or unspecified stereochemistry or a mixture of isomers. For example, the above diagram shows the 
File:Water with 4 single electrons.svg, The Lewis structure of water
CH3CH2OH ( ethanol)
Parentheses are used to indicate multiple identical groups, indicating attachment to the nearest non-hydrogen atom on the left when appearing within a formula, or to the atom on the right when appearing at the start of a formula:
: (CH3)2CHOH or CH(CH3)2OH (C=O
is implied through the O being placed in the brackets. For example:
CH3C(O)CH3 ( acetone)
Therefore, it is important to look to the left of the atom in the bracket to make sure what atom it is attached to. This is helpful when converting from condensed formula to another form of structural formula such as skeletal formula or Lewis structures. There are different ways to show the various functional groups in the condensed formulas such as aldehyde as CHO , CO2H or COOH , Esters as CO2R or COOR . However, the use of condensed formulas does not give an immediate idea of the molecular geometry of the compound or the number of bonds between the carbons, it needs to be recognized based on the number of atoms attached to the carbons and if there are any charges on the carbon.
Image:Isobutanol.svg, Skeletal formula of isobutanol, (CH3)2CHCH2OH
Image:Newman projection butane -sc.svg, Newman projection of


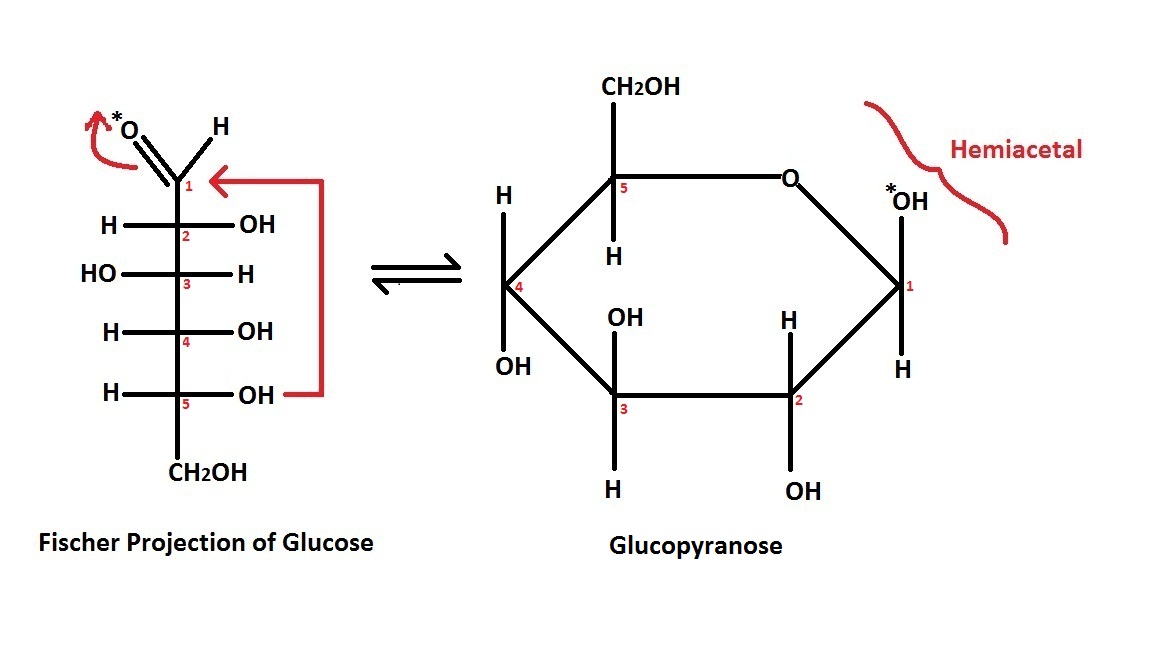
Image:beta-D-glucose Haworth formula.svg, Haworth projection of beta-D-Glucose
Image:DGlucose Fischer.svg, Fischer projection of D-Glucose
The Importance of Structural Formulas
*
{{DEFAULTSORT:Structural Formula Chemical formulas Chemical structures
chemical compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ...
is a graphic representation of the molecular structure (determined by structural chemistry
Structural chemistry is a part of chemistry and deals with spatial structures of molecules (in the gaseous, liquid or solid state) and solids (with extended structures that cannot be subdivided into molecules).
The main tasks are:
* The formulat ...
methods), showing how the atoms are possibly arranged in the real three-dimensional space
Space is the boundless three-dimensional extent in which objects and events have relative position and direction. In classical physics, physical space is often conceived in three linear dimensions, although modern physicists usually consider ...
. The chemical bond
A chemical bond is a lasting attraction between atoms or ions that enables the formation of molecules and crystals. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds, or through the sharing of ...
ing within the molecule is also shown, either explicitly or implicitly. Unlike other chemical formula
In chemistry, a chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, ...
types, which have a limited number of symbols and are capable of only limited descriptive power, structural formulas provide a more complete geometric representation of the molecular structure. For example, many chemical compounds exist in different isomer
In chemistry, isomers are molecules or polyatomic ions with identical molecular formulae – that is, same number of atoms of each element – but distinct arrangements of atoms in space. Isomerism is existence or possibility of isomers.
Iso ...
ic forms, which have different enantiomer
In chemistry, an enantiomer ( /ɪˈnænti.əmər, ɛ-, -oʊ-/ ''ih-NAN-tee-ə-mər''; from Ancient Greek ἐνάντιος ''(enántios)'' 'opposite', and μέρος ''(méros)'' 'part') – also called optical isomer, antipode, or optical ant ...
ic structures but the same molecular formula. There are multiple types of ways to draw these structural formulas such as: Lewis Structures
Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures (LEDS), are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons tha ...
, condensed formulas, skeletal formulas, Newman projections, Cyclohexane conformations, Haworth projections, and Fischer projections.
Several systematic chemical naming formats, as in chemical databases, are used that are equivalent to, and as powerful as, geometric structures. These chemical nomenclature
A chemical nomenclature is a set of rules to generate systematic names for chemical compounds. The nomenclature used most frequently worldwide is the one created and developed by the International Union of Pure and Applied Chemistry (IUPAC).
The ...
systems include SMILES, InChI
The International Chemical Identifier (InChI or ) is a textual identifier for chemical substances, designed to provide a standard way to encode molecular information and to facilitate the search for such information in databases and on the we ...
and CML. These systematic chemical names can be converted to structural formulas and vice versa, but chemists nearly always describe a chemical reaction or synthesis
Synthesis or synthesize may refer to:
Science Chemistry and biochemistry
*Chemical synthesis, the execution of chemical reactions to form a more complex molecule from chemical precursors
** Organic synthesis, the chemical synthesis of organ ...
using structural formulas rather than chemical names, because the structural formulas allow the chemist to visualize the molecules and the structural changes that occur in them during chemical reactions. ChemSketch and ChemDraw are popular downloads/websites that allow users to draw reactions and structural formulas, typically in the Lewis Structure style.
Structures in Structural Formulas
Bonds
Bonds are often shown as a line that connects one atom to another. One line indicates asingle bond
In chemistry, a single bond is a chemical bond between two atoms involving two valence electrons. That is, the atoms share one pair of electrons where the bond forms. Therefore, a single bond is a type of covalent bond. When shared, each of th ...
. Two lines indicate a double bond, and three lines indicate a triple bond. In some structures the atoms in between each bond are specified and shown. However, in some structures, the carbon molecules are not written out specifically. Instead, these carbons are indicated by a corner that forms when two lines connect. Additionally, Hydrogen atoms are implied and not usually drawn out. These can be inferred based on how many other atoms the carbon is attached to. For example, if Carbon A is attached to one other Carbon B, Carbon A will have three hydrogens in order to fill its octet.


Electrons
 Electrons are usually shown as colored in circles. One circle indicates one electron. Two circles indicate a pair of electrons. Typically, a pair of electrons will also indicate a negative charge. By using the colored circles, the number of electrons in the valence shell of each respective atom is indicated providing further descriptive information regarding the reactive capacity of that atom in the molecule.
Electrons are usually shown as colored in circles. One circle indicates one electron. Two circles indicate a pair of electrons. Typically, a pair of electrons will also indicate a negative charge. By using the colored circles, the number of electrons in the valence shell of each respective atom is indicated providing further descriptive information regarding the reactive capacity of that atom in the molecule.
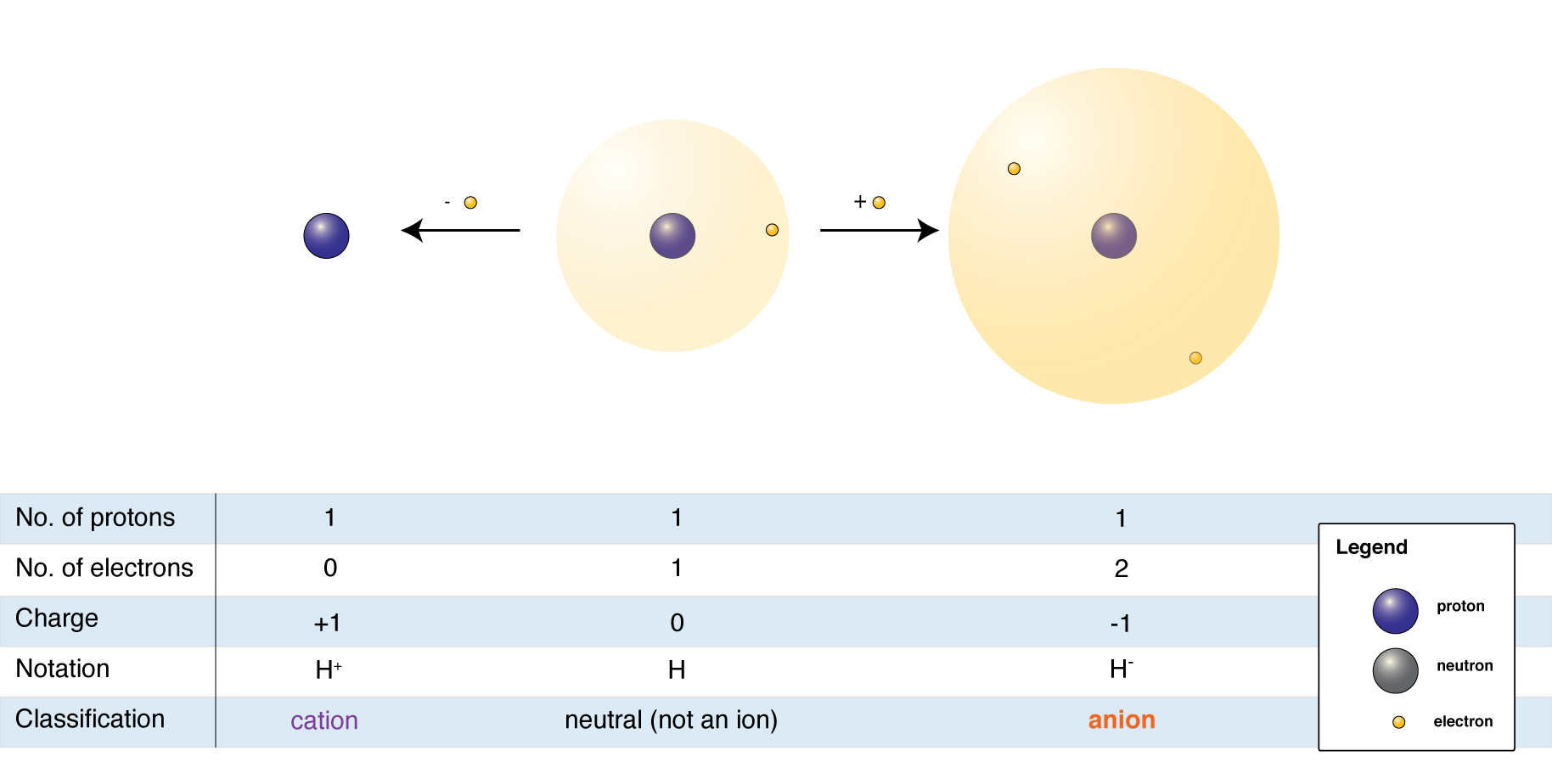
Charges
Often times, atoms will have a positive or negative charge as their octet may not be complete. If the atom is missing a pair of electrons or has a proton, it will have a positive charge. If the atom has electrons that are not bonded to another atom, there will be a negative charge. In structural formulas, the positive charge is indicated by ⊕ , and the negative charge is indicated by ⊖ .
Stereochemistry (Skeletal Formula)
Chirality
Chirality is a property of asymmetry important in several branches of science. The word ''chirality'' is derived from the Greek (''kheir''), "hand", a familiar chiral object.
An object or a system is ''chiral'' if it is distinguishable from ...
in skeletal formulas is indicated by the Natta projection method. Stereochemistry
Stereochemistry, a subdiscipline of chemistry, involves the study of the relative spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereois ...
is used to show the relative spatial arrangement of atoms in a molecule. Wedges are used to show this, and there are two types: dashed and filled. A filled wedge indicates that the atom is in the front of the molecule; it is pointing above the plane of the paper towards the front. A dashed wedge indicates that the atom is behind the molecule; it is pointing below the plane of the paper. When a straight, un-dashed line is used, the atom is in the plane of the paper. This spatial arrangement provides an idea of the molecule in a 3-dimensional space and there are constraints as to how the spatial arrangements can be arranged.
Unspecified Stereochemistry
fructose
Fructose, or fruit sugar, is a Ketose, ketonic monosaccharide, simple sugar found in many plants, where it is often bonded to glucose to form the disaccharide sucrose. It is one of the three dietary monosaccharides, along with glucose and galacto ...
molecule with a wavy bond to the HOCH2- group at the left. In this case the two possible ring structures are in chemical equilibrium with each other and also with the open-chain structure. The ring automatically opens and closes, sometimes closing with one stereochemistry and sometimes with the other.
Skeletal formulas can depict ''cis'' and ''trans'' isomers of alkenes. Wavy single bonds are the standard way to represent unknown or unspecified stereochemistry or a mixture of isomers (as with tetrahedral stereocenters). A crossed double-bond has been used sometimes, but is no longer considered an acceptable style for general use.
Lewis structures
Lewis structures (or "Lewis dot structures") are flat graphical formulas that show atom connectivity andlone pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC ''Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. Lone ...
or unpaired electrons, but not three-dimensional structure. This notation is mostly used for small molecules. Each line represents the two electrons of a single bond
In chemistry, a single bond is a chemical bond between two atoms involving two valence electrons. That is, the atoms share one pair of electrons where the bond forms. Therefore, a single bond is a type of covalent bond. When shared, each of th ...
. Two or three parallel lines between pairs of atoms represent double or triple bonds, respectively. Alternatively, pairs of dots may be used to represent bonding pairs. In addition, all non-bonded electrons (paired or unpaired) and any formal charges on atoms are indicated. Through the use of Lewis structures, the placement of electrons, whether it is in a bond or in lone pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC ''Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. Lone ...
s, will allow for the identification of the formal charge
In chemistry, a formal charge (F.C. or q), in the covalent view of chemical bonding, is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electroneg ...
s of the atoms in the molecule to understand the stability and determine the most likely molecule (based on molecular geometry difference) that would be formed in a reaction. Lewis structures do give some thought to the geometry of the molecule as often times, the bonds are drawn at certain angles to represent the molecule in real life. Lewis structure is best used to calculate formal charges or how atoms bond to each other as both electrons and bonds are shown. Lewis structures give an idea of the molecular
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and bioche ...
and electronic geometry which varies based on the presence of bonds and lone pairs and through this one could determine the bond angles and hybridization
Hybridization (or hybridisation) may refer to:
*Hybridization (biology), the process of combining different varieties of organisms to create a hybrid
*Orbital hybridization, in chemistry, the mixing of atomic orbitals into new hybrid orbitals
*Nu ...
as well.
Condensed formulas
In early organic-chemistry publications, where use of graphics was strongly limited, a typographic system arose to describe organic structures in a line of text. Although this system tends to be problematic in application to cyclic compounds, it remains a convenient way to represent simple structures: :2-propanol
Isopropyl alcohol (IUPAC name propan-2-ol and also called isopropanol or 2-propanol) is a colorless, flammable organic compound with a pungent alcoholic odor. As an isopropyl group linked to a hydroxyl group (chemical formula ) it is the sim ...
)
In all cases, all atoms are shown, including hydrogen atoms. It is also helpful to show the carbonyls where the
Carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
s as Skeletal formulas
Skeletal formulas are the standard notation for more complex organic molecules. In this type of diagram, first used by the organic chemistFriedrich August Kekulé von Stradonitz Friedrich may refer to:
Names
* Friedrich (surname), people with the surname ''Friedrich''
* Friedrich (given name), people with the given name ''Friedrich''
Other
* Friedrich (board game), a board game about Frederick the Great and the Seven Year ...
, the carbon atoms are implied to be located at the vertices (corners) and ends of line segments rather than being indicated with the atomic symbol C. Hydrogen atoms attached to carbon atoms are not indicated: each carbon atom is understood to be associated with enough hydrogen atoms to give the carbon atom four bonds. The presence of a positive or negative charge at a carbon atom takes the place of one of the implied hydrogen atoms. Hydrogen atoms attached to atoms other than carbon must be written explicitly. An additional feature of skeletal formulas is that by adding certain structures the stereochemistry
Stereochemistry, a subdiscipline of chemistry, involves the study of the relative spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereois ...
, that is the three-dimensional structure, of the compound can be determined. Often times, the skeletal formula can indicate stereochemistry through the use of wedges instead of lines. Solid wedges represent bonds pointing above the plane of the paper, whereas dashed wedges represent bonds pointing below the plane.
Perspective drawings
Newman projection and sawhorse projection
The Newman projection and the sawhorse projection are used to depict specificconformers
In chemistry, conformational isomerism is a form of stereoisomerism in which the isomers can be interconverted just by rotations about formally single bonds (refer to figure on single bond rotation). While any two arrangements of atoms in a molec ...
or to distinguish vicinal stereochemistry. In both cases, two specific carbon atoms and their connecting bond are the center of attention. The only difference is a slightly different perspective: the Newman projection looking straight down the bond of interest, the sawhorse projection looking at the same bond but from a somewhat oblique vantage point. In the Newman projection, a circle is used to represent a plane perpendicular to the bond, distinguishing the substituents on the front carbon from the substituents on the back carbon. In the sawhorse projection, the front carbon is usually on the left and is always slightly lower. Sometimes, an arrow is used to indicate the front carbon. The sawhorse projection is very similar to a skeletal formula, and it can even use wedges instead of lines to indicate the stereochemistry of the molecule. The sawhorse projection is set apart from the skeletal formulas because the sawhorse projection is not a very good indicator of molecule geometry and molecular arrangement. Both a Newman and Sawhorse Projection can be used to create a Fischer Projection.
butane
Butane () or ''n''-butane is an alkane with the formula C4H10. Butane is a gas at room temperature and atmospheric pressure. Butane is a highly flammable, colorless, easily liquefied gas that quickly vaporizes at room temperature. The name but ...
Image:Sawhorse projection butane -sc.svg, Sawhorse projection of butane
Cyclohexane conformations
Certain conformations ofcyclohexane
Cyclohexane is a cycloalkane with the molecular formula . Cyclohexane is non-polar. Cyclohexane is a colorless, flammable liquid with a distinctive detergent-like odor, reminiscent of cleaning products (in which it is sometimes used). Cyclohexan ...
and other small-ring compounds can be shown using a standard convention. For example, the standard chair conformation of cyclohexane involves a perspective view from slightly above the average plane of the carbon atoms and indicates clearly which groups are axial
Axial may refer to:
* one of the anatomical directions describing relationships in an animal body
* In geometry:
:* a geometric term of location
:* an axis of rotation
* In chemistry, referring to an axial bond
* a type of modal frame, in music
* ...
(pointing vertically up or down) and which are equatorial Equatorial may refer to something related to:
*Earth's equator
**the tropics, the Earth's equatorial region
**tropical climate
*the Celestial equator
** equatorial orbit
**equatorial coordinate system
** equatorial mount, of telescopes
* equatorial ...
(almost horizontal, slightly slanted up or down). Bonds in front may or may not be highlighted with stronger lines or wedges. The conformations progress as follows: chair to half-chair to twist-boat to boat to twist-boat to half-chair to chair. The cyclohexane conformations may also be used to show the potential energy present at each stage as shown in the diagram. The chair conformations (A) have the lowest energy, whereas the half-chair conformations (D) have the highest energy. There is a peak/local maximum at the boat conformation (C), and there are valleys/local minimums at the twist-boat conformations (B). In addition, cyclohexane conformations can be used to indicate if the molecule has any 1,3 diaxial-interactions which are steric interactions between axial substituents on the 1,3, and 5 carbons.Haworth projection
The Haworth projection is used for cyclicsugar
Sugar is the generic name for sweet-tasting, soluble carbohydrates, many of which are used in food. Simple sugars, also called monosaccharides, include glucose, fructose, and galactose. Compound sugars, also called disaccharides or double ...
s. Axial and equatorial positions are not distinguished; instead, substituents are positioned directly above or below the ring atom to which they are connected. Hydrogen substituents are typically omitted.
However, an important thing to keep in mind while reading an Haworth projection is that the ring structures are not flat. Therefore, Haworth does not provide 3-D shape. Sir Norman Haworth, was a British Chemist, who won a Noble Prize for his work on Carbohydrates and discovering the structure of Vitamin C. During his discovery, he also deducted different structural formulas which are now referred to as Haworth Projections. In a Haworth Projection a pyranose sugar is depicted as a hexagon and a furanose sugar is depicted as a pentagon. Usually an oxygen is placed at the upper right corner in pyranose and in the upper center in a furanose sugar. The thinner bonds at the top of the ring refer to the bonds as being farther away and the thicker bonds at the bottom of the ring refer to the end of the ring that is closer to the viewer.
Fischer projection
The Fischer projection is mostly used for linear monosaccharides. At any given carbon center, vertical bond lines are equivalent to stereochemical hashed markings, directed away from the observer, while horizontal lines are equivalent to wedges, pointing toward the observer. The projection is unrealistic, as a saccharide would never adopt this multiplyeclipsed
In chemistry an eclipsed conformation is a conformation in which two substituents X and Y on adjacent atoms A, B are in closest proximity, implying that the torsion angle X–A–B–Y is 0°. Such a conformation can exist in any open chain, ...
conformation. Nonetheless, the Fischer projection is a simple way of depicting multiple sequential stereocenters that does not require or imply any knowledge of actual conformation. A Fischer projection will restrict a 3-D molecule to 2-D, and therefore, there are limitations to changing the configuration of the chiral centers. Fischer projections are used to determine the R and S configuration on a chiral carbon and it is done using the Cahn Ingold Prelog rules. It is a convenient way to represent and distinguish between enantiomer
In chemistry, an enantiomer ( /ɪˈnænti.əmər, ɛ-, -oʊ-/ ''ih-NAN-tee-ə-mər''; from Ancient Greek ἐνάντιος ''(enántios)'' 'opposite', and μέρος ''(méros)'' 'part') – also called optical isomer, antipode, or optical ant ...
s and diastereomers.
Limitations
A structural formula is a simplified model that cannot represent certain aspects of chemical structures. For example, formalized bonding may not be applicable to dynamic systems such asdelocalized bond
In theoretical chemistry, a conjugated system is a system of connected p-orbitals with delocalized electrons in a molecule, which in general lowers the overall energy of the molecule and increases stability. It is conventionally represented as ...
s. Aromaticity is such a case and relies on convention to represent the bonding. Different styles of structural formulas may represent aromaticity in different ways, leading to different depictions of the same chemical compound. Another example is formal double bonds where the electron density is spread outside the formal bond, leading to partial double bond character and slow inter-conversion at room temperature. For all dynamic effects, temperature will affect the inter-conversion rates and may change how the structure should be represented. There is no explicit temperature associated with a structural formula, although many assume that it would be standard temperature
Standard temperature and pressure (STP) are standard sets of conditions for experimental measurements to be established to allow comparisons to be made between different sets of data. The most used standards are those of the International Union o ...
.
See also
* Molecular graph * Chemical formula * Valency interaction formula * Side chain * Chemical structureNotes
References
External links
The Importance of Structural Formulas
*
{{DEFAULTSORT:Structural Formula Chemical formulas Chemical structures