Chromosomal crossover on:
[Wikipedia]
[Google]
[Amazon]
 Chromosomal crossover, or crossing over, is the exchange of genetic material during
Chromosomal crossover, or crossing over, is the exchange of genetic material during
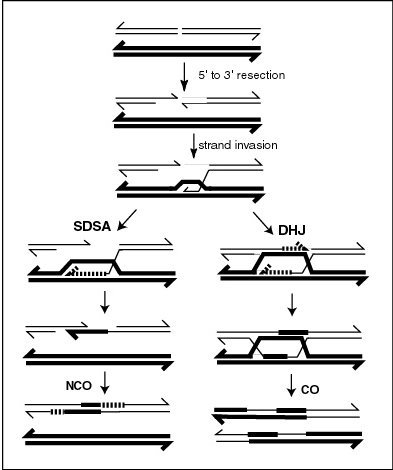 Meiotic recombination may be initiated by double-stranded breaks that are introduced into the DNA by exposure to DNA damaging agents, or the Spo11 protein. One or more exonucleases then digest the 5' ends generated by the double-stranded breaks to produce 3' single-stranded DNA tails (see diagram). The meiosis-specific recombinase Dmc1 and the general recombinase Rad51 coat the single-stranded DNA to form nucleoprotein filaments. The recombinases catalyze invasion of the opposite chromatid by the single-stranded DNA from one end of the break. Next, the 3' end of the invading DNA primes DNA synthesis, causing displacement of the complementary strand, which subsequently anneals to the single-stranded DNA generated from the other end of the initial double-stranded break. The structure that results is a ''cross-strand exchange'', also known as a Holliday junction. The contact between two chromatids that will soon undergo crossing-over is known as a '' chiasma''. The Holliday junction is a
Meiotic recombination may be initiated by double-stranded breaks that are introduced into the DNA by exposure to DNA damaging agents, or the Spo11 protein. One or more exonucleases then digest the 5' ends generated by the double-stranded breaks to produce 3' single-stranded DNA tails (see diagram). The meiosis-specific recombinase Dmc1 and the general recombinase Rad51 coat the single-stranded DNA to form nucleoprotein filaments. The recombinases catalyze invasion of the opposite chromatid by the single-stranded DNA from one end of the break. Next, the 3' end of the invading DNA primes DNA synthesis, causing displacement of the complementary strand, which subsequently anneals to the single-stranded DNA generated from the other end of the initial double-stranded break. The structure that results is a ''cross-strand exchange'', also known as a Holliday junction. The contact between two chromatids that will soon undergo crossing-over is known as a '' chiasma''. The Holliday junction is a
sexual reproduction
Sexual reproduction is a type of reproduction that involves a complex life cycle in which a gamete ( haploid reproductive cells, such as a sperm or egg cell) with a single set of chromosomes combines with another gamete to produce a zygote th ...
between two homologous chromosome
A couple of homologous chromosomes, or homologs, are a set of one maternal and one paternal chromosome that pair up with each other inside a cell during fertilization. Homologs have the same genes in the same loci where they provide points alon ...
s' non-sister chromatids that results in recombinant chromosome
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
s. It is one of the final phases of genetic recombination, which occurs in the ''pachytene'' stage of prophase I
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately res ...
of meiosis
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately r ...
during a process called synapsis. Synapsis begins before the synaptonemal complex develops and is not completed until near the end of prophase I. Crossover usually occurs when matching regions on matching chromosomes break and then reconnect to the other chromosome.
Crossing over was described, in theory, by Thomas Hunt Morgan
Thomas Hunt Morgan (September 25, 1866 – December 4, 1945) was an American evolutionary biologist, geneticist, embryologist, and science author who won the Nobel Prize in Physiology or Medicine in 1933 for discoveries elucidating the role that ...
. He relied on the discovery of Frans Alfons Janssens who described the phenomenon in 1909 and had called it "chiasmatypie". The term '' chiasma'' is linked, if not identical, to chromosomal crossover. Morgan immediately saw the great importance of Janssens' cytological interpretation of chiasmata to the experimental results of his research on the heredity of '' Drosophila''. The physical basis of crossing over was first demonstrated by Harriet Creighton
Harriet Baldwin Creighton (27 June 1909 – January 9, 2004) was an American botanist, geneticist and educator.
Background
Born in Delavan, Illinois, Creighton graduated from Wellesley College in 1929, and went on to complete her Ph.D. at Cor ...
and Barbara McClintock in 1931.
The linked frequency of crossing over between two gene
In biology, the word gene (from , ; "... Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a b ...
loci ( markers) is the '' crossing-over value'' . For fixed set of genetic and environmental conditions, recombination in a particular region of a linkage structure (chromosome
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
) tends to be constant and the same is then true for the crossing-over value which is used in the production of genetic maps.
Origins
There are two popular and overlapping theories that explain the origins of crossing-over, coming from the different theories on the origin ofmeiosis
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately r ...
. The first theory rests upon the idea that meiosis evolved as another method of DNA repair, and thus crossing-over is a novel way to replace possibly damaged sections of DNA. The second theory comes from the idea that meiosis evolved from bacterial transformation, with the function of propagating diversity.
In 1931, Barbara McClintock discovered a triploid maize plant. She made key findings regarding corn's karyotype, including the size and shape of the chromosomes. McClintock used the prophase and metaphase stages of mitosis to describe the morphology of corn's chromosomes, and later showed the first ever cytological demonstration of crossing over in meiosis. Working with student Harriet Creighton, McClintock also made significant contributions to the early understanding of codependency of linked genes.
DNA repair theory
Crossing over and DNA repair are very similar processes, which utilize many of the same protein complexes. In her report, "The Significance of Responses of the Genome to Challenge", McClintock studied corn to show how corn's genome would change itself to overcome threats to its survival. She used 450 self-pollinated plants that received from each parent a chromosome with a ruptured end. She used modified patterns of gene expression on different sectors of leaves of her corn plants to show that transposable elements ("controlling elements") hide in the genome, and their mobility allows them to alter the action of genes at different loci. These elements can also restructure the genome, anywhere from a few nucleotides to whole segments of chromosome. Recombinases and primases lay a foundation of nucleotides along the DNA sequence. One such particular protein complex that is conserved between processes is RAD51, a well conserved recombinase protein that has been shown to be crucial in DNA repair as well as cross over. Several other genes in ''D. melanogaster'' have been linked as well to both processes, by showing that mutants at these specific loci cannot undergo DNA repair or crossing over. Such genes include mei-41, mei-9, hdm, spnA, and brca2. This large group of conserved genes between processes supports the theory of a close evolutionary relationship. Furthermore, DNA repair and crossover have been found to favor similar regions on chromosomes. In an experiment using radiation hybrid mapping on wheat's (''Triticum aestivum L.'') 3B chromosome, crossing over and DNA repair were found to occur predominantly in the same regions. Furthermore, crossing over has been correlated to occur in response to stressful, and likely DNA damaging, conditionsLinks to bacterial transformation
The process of bacterial transformation also shares many similarities with chromosomal cross over, particularly in the formation of overhangs on the sides of the broken DNA strand, allowing for the annealing of a new strand. Bacterial transformation itself has been linked to DNA repair many times. The second theory comes from the idea that meiosis evolved from bacterial transformation, with the function of propagating genetic diversity. Thus, this evidence suggests that it is a question of whether cross over is linked to DNA repair or bacterial transformation, as the two do not appear to be mutually exclusive. It is likely that crossing over may have evolved from bacterial transformation, which in turn developed from DNA repair, thus explaining the links between all three processes.Chemistry
 Meiotic recombination may be initiated by double-stranded breaks that are introduced into the DNA by exposure to DNA damaging agents, or the Spo11 protein. One or more exonucleases then digest the 5' ends generated by the double-stranded breaks to produce 3' single-stranded DNA tails (see diagram). The meiosis-specific recombinase Dmc1 and the general recombinase Rad51 coat the single-stranded DNA to form nucleoprotein filaments. The recombinases catalyze invasion of the opposite chromatid by the single-stranded DNA from one end of the break. Next, the 3' end of the invading DNA primes DNA synthesis, causing displacement of the complementary strand, which subsequently anneals to the single-stranded DNA generated from the other end of the initial double-stranded break. The structure that results is a ''cross-strand exchange'', also known as a Holliday junction. The contact between two chromatids that will soon undergo crossing-over is known as a '' chiasma''. The Holliday junction is a
Meiotic recombination may be initiated by double-stranded breaks that are introduced into the DNA by exposure to DNA damaging agents, or the Spo11 protein. One or more exonucleases then digest the 5' ends generated by the double-stranded breaks to produce 3' single-stranded DNA tails (see diagram). The meiosis-specific recombinase Dmc1 and the general recombinase Rad51 coat the single-stranded DNA to form nucleoprotein filaments. The recombinases catalyze invasion of the opposite chromatid by the single-stranded DNA from one end of the break. Next, the 3' end of the invading DNA primes DNA synthesis, causing displacement of the complementary strand, which subsequently anneals to the single-stranded DNA generated from the other end of the initial double-stranded break. The structure that results is a ''cross-strand exchange'', also known as a Holliday junction. The contact between two chromatids that will soon undergo crossing-over is known as a '' chiasma''. The Holliday junction is a tetrahedral
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners. The tetrahedron is the simplest of all the ...
structure which can be 'pulled' by other recombinases, moving it along the four-stranded structure.
MSH4 and MSH5
The MSH4 and MSH5 proteins form a hetero-oligomeric structure ( heterodimer) in yeast and humans. In the yeast ''Saccharomyces cerevisiae
''Saccharomyces cerevisiae'' () (brewer's yeast or baker's yeast) is a species of yeast (single-celled fungus microorganisms). The species has been instrumental in winemaking, baking, and brewing since ancient times. It is believed to have b ...
'' MSH4 and MSH5 act specifically to facilitate crossovers between homologous chromosome
A couple of homologous chromosomes, or homologs, are a set of one maternal and one paternal chromosome that pair up with each other inside a cell during fertilization. Homologs have the same genes in the same loci where they provide points alon ...
s during meiosis
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately r ...
. The MSH4/MSH5 complex binds and stabilizes double Holliday junctions and promotes their resolution into crossover products. An MSH4 hypomorphic (partially functional) mutant of ''S. cerevisiae'' showed a 30% genome wide reduction in crossover numbers, and a large number of meioses with non exchange chromosomes. Nevertheless, this mutant gave rise to spore viability patterns suggesting that segregation Segregation may refer to:
Separation of people
* Geographical segregation, rates of two or more populations which are not homogenous throughout a defined space
* School segregation
* Housing segregation
* Racial segregation, separation of humans ...
of non-exchange chromosomes occurred efficiently. Thus in ''S. cerevisiae'' proper segregation apparently does not entirely depend on crossovers between homologous pairs.
Chiasma
The grasshopper '' Melanoplus femur-rubrum'' was exposed to an acute dose ofX-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
s during each individual stage of meiosis
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately r ...
, and chiasma frequency was measured. Irradiation during the leptotene- zygotene stages of meiosis
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately r ...
(that is, prior to the pachytene
Meiosis (; , since it is a reductional division) is a special type of cell division of germ cells in sexually-reproducing organisms that produces the gametes, such as sperm or egg cells. It involves two rounds of division that ultimately res ...
period in which crossover recombination occurs) was found to increase subsequent chiasma frequency. Similarly, in the grasshopper '' Chorthippus brunneus'', exposure to X-irradiation during the zygotene-early pachytene stages caused a significant increase in mean cell chiasma frequency. Chiasma frequency was scored at the later diplotene-diakinesis stages of meiosis. These results suggest that X-rays induce DNA damages that are repaired by a crossover pathway leading to chiasma formation.
Class I and class II crossovers
Double strand breaks (DSBs) are repaired by two pathways to generate crossovers in eukaryotes. The majority of them are repaired by MutL homologs MLH1 and MLH3, which defines the class I crossovers. The remaining are the result of the class II pathway, which is regulated by MUS81 endonuclease. There are interconnections between these two pathways—class I crossovers can compensate for the loss of class II pathway. In MUS81 knockout mice, class I crossovers are elevated, while total crossover counts at chiasmata are normal. However, the mechanisms underlining this crosstalk are not well understood. A recent study suggests that a scaffold protein called SLX4 may participate in this regulation. Specifically, SLX4 knockout mice largely phenocopies the MUS81 knockout—once again, an elevated class I crossovers while normal chiasmata count.Consequences
In most eukaryotes, acell
Cell most often refers to:
* Cell (biology), the functional basic unit of life
Cell may also refer to:
Locations
* Monastic cell, a small room, hut, or cave in which a religious recluse lives, alternatively the small precursor of a monastery ...
carries two versions of each gene
In biology, the word gene (from , ; "... Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a b ...
, each referred to as an allele
An allele (, ; ; modern formation from Greek ἄλλος ''állos'', "other") is a variation of the same sequence of nucleotides at the same place on a long DNA molecule, as described in leading textbooks on genetics and evolution.
::"The chro ...
. Each parent passes on one allele to each offspring. An individual gamete
A gamete (; , ultimately ) is a haploid cell that fuses with another haploid cell during fertilization in organisms that reproduce sexually. Gametes are an organism's reproductive cells, also referred to as sex cells. In species that produce ...
inherits a complete haploid complement of alleles on chromosomes that are independently selected from each pair of chromatids lined up on the metaphase plate. Without recombination, all alleles for those genes linked together on the same chromosome would be inherited together. Meiotic recombination allows a more independent segregation between the two alleles that occupy the positions of single genes, as recombination shuffles the allele content between homologous chromosomes.
Recombination results in a new arrangement of maternal and paternal alleles on the same chromosome. Although the same genes appear in the same order, some alleles are different. In this way, it is theoretically possible to have any combination of parental alleles in an offspring, and the fact that two alleles appear together in one offspring does not have any influence on the statistical probability that another offspring will have the same combination. This principle of " independent assortment" of genes is fundamental to genetic inheritance.
However, the frequency of recombination is actually not the same for all gene combinations. This leads to the notion of "genetic distance
Genetic distance is a measure of the genetic divergence between species or between populations within a species, whether the distance measures time from common ancestor or degree of differentiation. Populations with many similar alleles have s ...
", which is a measure of recombination frequency averaged over a (suitably large) sample of pedigrees. Loosely speaking, one may say that this is because recombination is greatly influenced by the proximity of one gene to another. If two genes are located close together on a chromosome, the likelihood that a recombination event will separate these two genes is less than if they were farther apart. Genetic linkage describes the tendency of genes to be inherited together as a result of their location on the same chromosome. Linkage disequilibrium
In population genetics, linkage disequilibrium (LD) is the non-random association of alleles at different loci in a given population. Loci are said to be in linkage disequilibrium when the frequency of association of their different alleles is h ...
describes a situation in which some combinations of genes or genetic markers occur more or less frequently in a population than would be expected from their distances apart. This concept is applied when searching for a gene that may cause a particular disease
A disease is a particular abnormal condition that negatively affects the structure or function of all or part of an organism, and that is not immediately due to any external injury. Diseases are often known to be medical conditions that a ...
. This is done by comparing the occurrence of a specific DNA sequence with the appearance of a disease. When a high correlation between the two is found, it is likely that the appropriate gene sequence is really closer.Genetic Recombination
Non-homologous crossover
Crossovers typically occur between homologous regions of matchingchromosome
A chromosome is a long DNA molecule with part or all of the genetic material of an organism. In most chromosomes the very long thin DNA fibers are coated with packaging proteins; in eukaryotic cells the most important of these proteins are ...
s, but similarities in sequence and other factors can result in mismatched alignments. Most DNA is composed of base pair sequences repeated very large numbers of times. These repetitious segments, often referred to as satellites, are fairly homogeneous among a species. During DNA replication, each strand of DNA is used as a template for the creation of new strands using a partially-conserved mechanism; proper functioning of this process results in two identical, paired chromosomes, often called sisters. Sister chromatid
A sister chromatid refers to the identical copies (chromatids) formed by the DNA replication of a chromosome, with both copies joined together by a common centromere. In other words, a sister chromatid may also be said to be 'one-half' of the dup ...
crossover events are known to occur at a rate of several crossover events per cell per division in eukaryotes. Most of these events involve an exchange of equal amounts of genetic information, but unequal exchanges may occur due to sequence mismatch. These are referred to by a variety of names, including non-homologous crossover, unequal crossover, and unbalanced recombination, and result in an insertion or deletion of genetic information into the chromosome. While rare compared to homologous crossover events, these mutations are drastic, affecting many loci at the same time. They are considered the main driver behind the generation of gene duplications and are a general source of mutation within the genome.
The specific causes of non-homologous crossover events are unknown, but several influential factors are known to increase the likelihood of an unequal crossover. One common vector leading to unbalanced recombination is the repair of double-strand breaks (DSBs). DSBs are often repaired using homology directed repair, a process which involves invasion of a template strand
Transcription is the process of copying a segment of DNA into RNA. The segments of DNA transcribed into RNA molecules that can encode proteins are said to produce messenger RNA (mRNA). Other segments of DNA are copied into RNA molecules called ...
by the DSB strand (see figure below). Nearby homologous regions of the template strand are often used for repair, which can give rise to either insertions or deletions in the genome if a non-homologous but complementary part of the template strand is used. Sequence similarity is a major player in crossover – crossover events are more likely to occur in long regions of close identity on a gene. This means that any section of the genome with long sections of repetitive DNA is prone to crossover events.
The presence of transposable elements is another influential element of non-homologous crossover. Repetitive regions of code characterize transposable elements; complementary but non-homologous regions are ubiquitous within transposons. Because chromosomal regions composed of transposons have large quantities of identical, repetitious code in a condensed space, it is thought that transposon regions undergoing a crossover event are more prone to erroneous complementary match-up; that is to say, a section of a chromosome containing a lot of identical sequences, should it undergo a crossover event, is less certain to match up with a perfectly homologous section of complementary code and more prone to binding with a section of code on a slightly different part of the chromosome. This results in unbalanced recombination, as genetic information may be either inserted or deleted into the new chromosome, depending on where the recombination occurred.
While the motivating factors behind unequal recombination remain obscure, elements of the physical mechanism have been elucidated. Mismatch repair
DNA mismatch repair (MMR) is a system for recognizing and repairing erroneous insertion, deletion, and mis-incorporation of bases that can arise during DNA replication and recombination, as well as repairing some forms of DNA damage.
Mismatch ...
(MMR) proteins, for instance, are a well-known regulatory family of proteins, responsible for regulating mismatched sequences of DNA during replication and escape regulation. The operative goal of MMRs is the restoration of the parental genotype. One class of MMR in particular, MutSβ, is known to initiate the correction of insertion-deletion mismatches of up to 16 nucleotides. Little is known about the excision process in eukaryotes, but ''E. coli'' excisions involve the cleaving of a nick on either the 5' or 3' strand, after which DNA helicase
Helicases are a class of enzymes thought to be vital to all organisms. Their main function is to unpack an organism's genetic material. Helicases are motor proteins that move directionally along a nucleic acid phosphodiester backbone, separatin ...
and DNA polymerase III bind and generate single-stranded proteins, which are digested by exonucleases and attached to the strand by ligase
In biochemistry, a ligase is an enzyme that can catalyze the joining (ligation) of two large molecules by forming a new chemical bond. This is typically via hydrolysis of a small pendant chemical group on one of the larger molecules or the enzym ...
. Multiple MMR pathways have been implicated in the maintenance of complex organism genome stability, and any of many possible malfunctions in the MMR pathway result in DNA editing and correction errors. Therefore, while it is not certain precisely what mechanisms lead to errors of non-homologous crossover, it is extremely likely that the MMR pathway is involved.
See also
*Unequal crossing over
Unequal crossing over is a type of gene duplication or deletion event that deletes a sequence in one strand and replaces it with a duplication from its sister chromatid in mitosis or from its homologous chromosome during meiosis. It is a type of ...
* Coefficient of coincidence
*Genetic distance
Genetic distance is a measure of the genetic divergence between species or between populations within a species, whether the distance measures time from common ancestor or degree of differentiation. Populations with many similar alleles have s ...
* Independent assortment
*Mitotic crossover
Mitotic recombination is a type of genetic recombination that may occur in somatic cells during their preparation for mitosis in both sexual and asexual organisms. In asexual organisms, the study of mitotic recombination is one way to understand ...
* Recombinant frequency
References
{{Authority control Cellular processes Modification of genetic information Molecular genetics