Botrytis cinerea on:
[Wikipedia]
[Google]
[Amazon]
''Botrytis cinerea'' is a necrotrophic
Biocontrol of ''Botrytis Cinerea'' on Strawberry Fruit by Plant Growth Promoting Bacteria
''The Journal of Animal & Plant Sciences'', 21(4), 2011: pp. 758-763, ISSN 1018-7081.
 In greenhouse horticulture, ''Botrytis cinerea'' is well known as a cause of considerable damage in
In greenhouse horticulture, ''Botrytis cinerea'' is well known as a cause of considerable damage in
 ''Botrytis cinerea'' not only infects plants, it also hosts several mycoviruses itself (see the table/image).
A range of phenotypic alterations due to the mycoviral infection have been observed from symptomless to mild impact, or more severe phenotypic changes including reduction in pathogenicity, growth/suppression of mycelia, sporulation and sclerotia production, formation of abnormal colony sectors (Wu et al., 2010) and virulence.
''Botrytis cinerea'' not only infects plants, it also hosts several mycoviruses itself (see the table/image).
A range of phenotypic alterations due to the mycoviral infection have been observed from symptomless to mild impact, or more severe phenotypic changes including reduction in pathogenicity, growth/suppression of mycelia, sporulation and sclerotia production, formation of abnormal colony sectors (Wu et al., 2010) and virulence.
Genome information for ''Botrytis cinerea''
Genome analysis of ''Botrytis cinerea''
*
TheWineDoctor.com
* * * {{Authority control Sclerotiniaceae Fungal plant pathogens and diseases Small fruit diseases Fungal strawberry diseases Fungal citrus diseases Fungal grape diseases Oenology Fungi described in 1794
fungus
A fungus ( : fungi or funguses) is any member of the group of eukaryotic organisms that includes microorganisms such as yeasts and molds, as well as the more familiar mushrooms. These organisms are classified as a kingdom, separately fr ...
that affects many plant species, although its most notable hosts may be wine grapes
This list of grape varieties includes cultivated grapes, whether used for wine, or eating as a table grape, fresh or dried (raisin, currant, sultana). For a complete list of all grape species including those unimportant to agriculture, see Vit ...
. In viticulture
Viticulture (from the Latin word for '' vine'') or winegrowing (wine growing) is the cultivation and harvesting of grapes. It is a branch of the science of horticulture. While the native territory of '' Vitis vinifera'', the common grape vine, r ...
, it is commonly known as "botrytis bunch rot"; in horticulture
Horticulture is the branch of agriculture that deals with the art, science, technology, and business of plant cultivation. It includes the cultivation of fruits, vegetables, nuts, seeds, herbs, sprouts, mushrooms, algae, flowers, seaweeds and no ...
, it is usually called "grey mould" or "gray mold".
The fungus gives rise to two different kinds of infections on grapes. The first, grey rot, is the result of consistently wet or humid conditions, and typically results in the loss of the affected bunches. The second, noble rot
Noble rot (french: pourriture noble; german: Edelfäule; it, Muffa nobile; hu, Aszúsodás) is the beneficial form of a grey fungus, ''Botrytis cinerea'', affecting wine grapes. Infestation by ''Botrytis'' requires moist conditions. If the we ...
, occurs when drier conditions follow wetter, and can result in distinctive sweet dessert wine
Dessert wines, sometimes called pudding wines in the United Kingdom, are sweet wines typically served with dessert.
There is no simple definition of a dessert wine. In the UK, a dessert wine is considered to be any sweet wine drunk with a meal ...
s, such as Sauternes or the Aszú of Tokaji
Tokaji ( hu, of Tokaj ) or Tokay is the name of the wines from the Tokaj wine region (also ''Tokaj-Hegyalja wine region'' or ''Tokaj-Hegyalja'') in Hungary or the adjoining Tokaj wine region in Slovakia. This region is noted for its sweet wines ...
/Grasă de Cotnari
''Grasă de Cotnari'' () is a Romanian wine variety associated with the Cotnari wine region, in Iași County (historical region of Moldavia), where it has been grown ever since the rule of Prince Stephen the Great (1457–1504).
It is also ...
. The species name ''Botrytis cinerea'' is derived from the Latin
Latin (, or , ) is a classical language belonging to the Italic languages, Italic branch of the Indo-European languages. Latin was originally a dialect spoken in the lower Tiber area (then known as Latium) around present-day Rome, but through ...
for "grapes like ashes"; although poetic, the "grapes" refers to the bunching of the fungal spores
In biology, a spore is a unit of sexual or asexual reproduction that may be adapted for dispersal and for survival, often for extended periods of time, in unfavourable conditions. Spores form part of the life cycles of many plants, algae, ...
on their conidiophores
A conidium ( ; ), sometimes termed an asexual chlamydospore or chlamydoconidium (), is an asexual, non-motile spore of a fungus. The word ''conidium'' comes from the Ancient Greek word for dust, ('). They are also called mitospores due to ...
, and "ashes" just refers to the greyish colour of the spores ''en masse''. The fungus is usually referred to by its anamorph
In mycology, the terms teleomorph, anamorph, and holomorph apply to portions of the life cycles of fungi in the phyla Ascomycota and Basidiomycota:
*Teleomorph: the sexual reproductive stage (morph), typically a fruiting body.
*Anamorph: an a ...
(asexual form) name, because the sexual phase is rarely observed. The teleomorph
In mycology, the terms teleomorph, anamorph, and holomorph apply to portions of the life cycles of fungi in the phyla Ascomycota and Basidiomycota:
*Teleomorph: the sexual reproductive stage (morph), typically a fruiting body.
*Anamorph: an asex ...
(sexual form) is an ascomycete
Ascomycota is a phylum of the kingdom Fungi that, together with the Basidiomycota, forms the subkingdom Dikarya. Its members are commonly known as the sac fungi or ascomycetes. It is the largest phylum of Fungi, with over 64,000 species. The defi ...
, ''Botryotinia fuckeliana'', also known as ''Botryotinia cinerea'' (see taxonomy box).
Etymology
"Botrytis Botrytis may refer to:
* ''Botrytis'' (fungus), the anamorphs of fungi of the genus '' Botryotinia''
**''Botrytis cinerea'', a mold important in wine making
*Botrytis, the cauliflower cultivar group of ''Brassica oleracea
''Brassica oleracea'' is ...
" is derived from the Ancient Greek
Ancient Greek includes the forms of the Greek language used in ancient Greece and the ancient world from around 1500 BC to 300 BC. It is often roughly divided into the following periods: Mycenaean Greek (), Dark Ages (), the Archaic pe ...
''botrys ''(βότρυς) meaning "grapes", combined with the New Latin
New Latin (also called Neo-Latin or Modern Latin) is the revival of Literary Latin used in original, scholarly, and scientific works since about 1500. Modern scholarly and technical nomenclature, such as in zoological and botanical taxonomy ...
suffix ''-itis'' for disease. ''Botryotinia fuckeliana'' was named by mycologist Heinrich Anton de Bary
Heinrich Anton de Bary (26 January 183119 January 1888) was a German surgeon, botanist, microbiologist, and mycologist (fungal systematics and physiology).
He is considered a founding father of plant pathology (phytopathology) as well as the fou ...
in honor of another mycologist, Karl Wilhelm Gottlieb Leopold Fuckel
Karl Wilhelm Gottlieb Leopold Fuckel (3 February 1821 – 8 May 1876) was a German botanist who worked largely on fungi.
He worked as an apothecary from 1836 to 1852, afterwards deriving income from a vineyard he owned in Oestrich im Rheingau. ...
. Synonyms for the sexual stage are:
* ''Botrytis fuckeliana'' N.F. Buchw., (1949)
* ''Botrytis gemella'' (Bonord.) Sacc., (1881)
* ''Botrytis grisea'' (Schwein.) Fr., (1832)
* ''Botrytis vulgaris'' (Pers.) Fr., (1832)
* ''Haplaria grisea'' Link, (1809)
* ''fuckeliana'' de Bary
* ''Phymatotrichum gemellum'' Bonord., (1851)
* ''Polyactis vulgaris'' Pers., (1809)
* ''Sclerotinia fuckeliana'' (de Bary) Fuckel, (1870)
Hosts and symptoms
Hosts
The disease, gray mold, affects more than 200dicotyledon
The dicotyledons, also known as dicots (or, more rarely, dicotyls), are one of the two groups into which all the flowering plants (angiosperms) were formerly divided. The name refers to one of the typical characteristics of the group: namely, t ...
ous plant species and a few monocotyledon
Monocotyledons (), commonly referred to as monocots, ( Lilianae '' sensu'' Chase & Reveal) are grass and grass-like flowering plants (angiosperms), the seeds of which typically contain only one embryonic leaf, or cotyledon. They constitute one ...
ous plants found in temperate and subtropical regions, and potentially over a thousand species. Serious economic losses can be a result of this disease to both field and greenhouse grown crops. The causal agent, ''Botrytis cinerea'' can infect mature or senescent tissues, plants prior to harvest, or seedlings. There is a wide variety of hosts infected by this pathogen including protein crops, fiber crops, oil crops, and horticultural crops. Horticultural crops include vegetables (examples are chickpeas, lettuce, broccoli, and beans) and small fruit crops (examples are grape, strawberry, raspberry, and blackberry), these are most severely affected and devastated by gray mold. Plant organs affected include fruits, flowers, leaves, storage organs, and shoots.
Symptoms and signs
Symptoms vary across plant organs and tissues. ''B. cinerea'' is a soft rot that will have a collapsed and water soaked appearance on soft fruit and leaves. Brown lesions may develop slowly on undeveloped fruit. Twigs infected with gray mold will die back. Blossoms will cause fruit drop and injury, such as ridging on developing and mature fruit. Symptoms are visible at wound sites where the fungus begins to rot the plant. Gray masses with a velvety appearance are conidia on the plant tissues are a sign of plant pathogen. These conidia are asexual spores that will continue to infect the plant and surrounding hosts throughout the growing season making this a polycyclic disease. Plants can produce localized lesions when a pathogen attacks. An oxidative burst causes hypersensitive cell death called ahypersensitive response
Hypersensitive response (HR) is a mechanism used by plants to prevent the spread of infection by microbial pathogens. HR is characterized by the rapid death of cells in the local region surrounding an infection and it serves to restrict the g ...
(HR). This soft rot can trigger HR to assist in colonization. ''Botrytis cinerea'', as a necrotrophic pathogen, exploits the dead tissue for its pathogenicity or its ability to cause disease. Susceptible plants cannot use the HR to protect against ''B. cinerea''.
See:
* List of potato diseases
This is a list of diseases and disorders found in potatoes.
Bacterial diseases
Fungal diseases
Protistan diseases
Viral and viroid diseases
Nematode parasitic
Phytoplasmal diseases
Miscellaneous diseases and disorders
References
Com ...
* List of canola diseases
This article is a list of diseases of rapeseed and canola (''Brassica napus'' and ''B. rapa'' or ''B. campestris'').
Bacterial diseases
Fungal diseases
Viral diseases
Phytoplasmal diseases
Miscellaneous diseases and disorders
{, cl ...
* List of maize diseases Bacterial diseases
Fungal diseases
Nematodes, Parasitic
Virus and virus-like diseases
References
Common Names of Diseases, The American Phytopathological Society
{{corn
*
Maize
Maize ( ; ''Zea mays'' subsp. ''mays'', from ...
* List of alfalfa diseases
This article is a list of diseases of alfalfa (''Medicago sativa'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
Phytoplasmal and spiroplasmal diseases
See also
* Alfalfa pests, pests named for alfalfa
R ...
* List of African daisy diseases
This is a list of diseases of the African daisy (''Gerbera jamesonii
''Gerbera jamesonii'' is a species of flowering plant in the genus ''Gerbera'' belonging to the basal Mutisieae Tribe (biology), tribe within the large Asteraceae (or Composita ...
* List of African violet diseases
This article is a list of diseases of African violets (''Saintpaulia ionantha'').
Bacterial diseases
Fungal diseases
Fulginacillis Follicle Growth Sistementaris Gland
Viral diseases
Nematodes, parasitic
Miscellaneous di ...
* List of pea diseases
This article is a list of diseases of peas (''Pisum sativum'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
{, class="wikitable" style="clear"
! colspan=2, Viral diseases
, -
, Cucumber mosaic virus
, , gen ...
* List of lentil diseases
* List of anemone diseases
* List of almond diseases
* List of apple diseases
This article is a list of diseases of apples (''Malus domestica'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
Viroid diseases
Suspected viral- and viroid-like diseases
Phytoplasmal diseases
Miscel ...
* List of apricot diseases
This article is a list of diseases of apricots (Prunus armeniaca).
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
includes uncharacterized graft-transmissible pathogens TP
Graft-transmissible pathogens TP/h1>
...
* List of asparagus diseases
* List of avocado diseases
* List of azalea diseases
* List of beet diseases
This article is a list of diseases of beets (''Beta vulgaris''), a plant grown for its edible taproot and leaves.
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
Phytoplasmal and sprioplasmal diseases
Miscell ...
* List of bellflower diseases
This article is a list of diseases of bellflowers (''Campanula carpatica'').
Bacterial diseases
Fungal diseases
Viral and viroid diseases
References
{{reflistCommon Names of Diseases, The American Phytopathological Society
Campanula
Be ...
* List of bleeding heart diseases
A ''list'' is any set of items in a row. List or lists may also refer to:
People
* List (surname)
Organizations
* List College, an undergraduate division of the Jewish Theological Seminary of America
* SC Germania List, German rugby union ...
* List of butterfly flower diseases
This is a list of diseases of Butterfly Flower (''Schizanthus × wisetonensis'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral and viroid diseases
{, class="wikitable" style="clear:left"
! colspan=2, Viral and viroid ...
* List of caneberries diseases
This article is a list of diseases of caneberries (''Rubus
''Rubus'' is a large and diverse genus of flowering plants in the rose family, Rosaceae, subfamily Rosoideae, with over 1,350 species.
Raspberries, blackberries, and dewberries are co ...
* List of carrot diseases
This is a list of diseases of carrots (''Daucus carota'' subsp. ''sativus'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
, Cyst nematode
, ,
'' Heterodera carotae''
, -
, Lance nematode
, ,
'' Hoplolaimus uniformis''
, -
...
* List of tea diseases
Many of the diseases, pathogens and pests that affect the tea plant (''Camellia sinensis'') may affect other members of the plant genus ''Camellia''.
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Lepidoptera (butterflies and moth) ...
* List of tobacco diseases
This is a list of diseases of tobacco (''Nicotiana tabacum'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral and phytoplasma diseases
Miscellaneous diseases and disorders
{, class="wikitable" style="clear ...
* List of tomato diseases
This article is a list of diseases of tomatoes (''Solanum lycopersicum'').
Bacterial diseases
Fungal diseases
Lepidoptera larvae
Nematodes
Viral and viroid
Miscellaneous diseases and disorders
{, class="wikitable" style="clear"
, ...
* List of verbena diseases
This article is a list of diseases of verbena (''Verbena × hybrida'').
Fungal diseases
Viral diseases
{, class="wikitable" style="clear"
, Bidens mottle
, , genus Potyvirus, Bidens mottle virus (BiMoV)
, -
, Impatiens necrotic spot
, , gen ...
* List of sweet potato diseases
* List of sunflower diseases
This article is a list of diseases of sunflowers (''Helianthus annuus'') and jerusalem artichoke (''H. tuberosus'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Phytoplasma and Viral diseases
References
{{reflist
Common ...
* List of strawberry diseases
* List of sapphire flower diseases
* List of safflower diseases
This article is a list of diseases of safflowers (''Carthamus tinctorius'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral and Phytoplasma
{, class="wikitable" style="clear"
! colspan=2, Viral and mycoplasmalike organ ...
* List of rose diseases
Roses (''Rosa'' species) are susceptible to a number of Pest (organism), pests, plant disease, diseases and disorders. Many of the problems affecting roses are seasonal and climatic.Ross, D.,''Rose-growing for Pleasure'', Lothian Publis ...
* List of primula diseases
* List of poinsettia diseases
This article is a list of diseases of poinsettia (''Euphorbia pulcherrima'').
Bacterial diseases
Fungal diseases
This also includes oomycetes
Nematodes, parasitic
Viral and viroid diseases
Miscellaneous diseases and disorders
Reference ...
* List of pocketbook plant diseases
* List of pistachio diseases
* List of pigeonpea diseases
* List of Persian violet diseases
This article is a list of diseases of Persian violets (''Exacum affine
''Exacum affine''''Exacum affine''.< ...
* List of Capsicum diseases
This article is a list of diseases of ''Capsicum'' species.
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
Post-harvest diseases
Abiotic diseases
{, class="wikitable" style="clear"
! colspan=2, Abiotic disease ...
* List of pear diseases
* List of peanut diseases
This article is a list of diseases of peanuts (''Arachis hypogaea'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Phytoplasma, Virus and viruslike diseases
Miscellaneous and diseases or disorders
References
{{reflist
...
* List of peach and nectarine diseases
This article is a list of diseases of peaches and nectarines (Peach: ''Prunus persica''; Nectarine: ''P. persica'' var. ''nucipersica'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral and viroid diseases
(Also uncharact ...
* List of mimulus, monkey-flower diseases
* List of mango diseases
This article is a list of diseases of mangos (''Mangifera indica'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Miscellaneous diseases and disorders
ReferencesCommon Names of Diseases, The American Phytopathological Societ ...
* List of lettuce diseases
This article is a list of diseases of lettuce (''Lactuca sativa'').
Bacterial diseases
Fungal diseases
Miscellaneous diseases and disorders
Nematodes, parasitic
Phytoplasma, Viral and viroid diseases
ReferencesCommon Names of Diseas ...
* List of kalanchoe diseases
* List of Jerusalem cherry diseases
This article is a list of diseases of the Jerusalem cherry (''Solanum pseudocapsicum'').
Fungal diseases
Nematodes, parasitic
Viral and viroid diseases
References
{{reflist External linksCommon Names of Diseases, The American Phytopath ...
* List of impatiens diseases
This article is a list of diseases of impatiens, such as Busy Lizzie (''I. walleriana'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral and viroid diseases
{, class="wikitable" style="clear"
! colspan=2, v diseases
, ...
* List of hop diseases
This article is a list of diseases of hops (''Humulus lupulus'').
Bacterial diseases
Fungal diseases
Miscellaneous diseases and disorders
Nematodes, parasitic
Virus and viroid diseases
{, class="wikitable" style="clear"
! colspan=2, ...
* List of hemp diseases
This is a list of diseases of hemp (''Cannabis sativa'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
Phytoplasmal diseases
Miscellaneous diseases and disorders
References
Common Names of Diseases, The Am ...
* List of grape diseases
This is a list of diseases of grapes (''Vitis'' spp.).
Bacterial diseases
Fungal diseases
Miscellaneous diseases and disorders
Nematodes, parasitic
Phytoplasma, virus and viruslike diseases
See also
*'' Ampeloglypter ater''
*'' A ...
* List of geranium diseases
This article is a list of diseases of geraniums (''Pelargonium'').
Bacterial diseases
Fungal diseases
Virus diseases
Nematodes, parasitic
Miscellaneous diseases and disorders
{, class="wikitable" style="clear"
! colspan=2, Miscella ...
* List of fuchsia diseases
This article is a list of diseases of fuchsias (''Fuchsia × hybrida'').
Bacterial diseases
Fungal diseases
Viral and viroid diseases
{, class="wikitable" style="clear"
! colspan=2, Viral and viroid diseases
, -
, Impatiens necrotic spot ...
* List of cyclamen diseases
* List of cucurbit diseases
This article is a list of diseases of cucurbits (''Citrullus'' spp., ''Cucumis'' spp., ''Cucurbita'' spp., and others).
Bacterial diseases
Fungal diseases
Miscellaneous diseases and disorders
Nematodes, parasitic
{, class="wikitable" ...
* List of crucifer diseases
* List of citrus diseases
A ''list'' is any set of items in a row. List or lists may also refer to:
People
* List (surname)
Organizations
* List College, an undergraduate division of the Jewish Theological Seminary of America
* SC Germania List, German rugby union ...
* List of cineraria diseases
This is a list of diseases of cineraria (''Pericallis × hybrida'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral and viroid diseases
Phytoplasmal diseases
{, class="wikitable" style="clear"
! colspan=2, Phytoplasm ...
* List of chickpea diseases
This is a list of diseases of chickpeas (''Cicer arietinum'')
Nematodes, parasitic
Viral diseases
Phytoplasmal diseases
{, class="wikitable" style="clear"
! colspan=2, Phytoplasmal diseases
, -
, Phyllody
, , Phytoplasma
, -
Refer ...
* List of cattleya diseases
* List of carnation diseases
This article is a list of diseases of carnations (''Dianthus caryophylium'').
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
References
{{reflist
Common Names of Diseases, The American Phytopathological Soci ...
* List of Douglas-fir diseases
This article is a list of diseases of Douglas-fir (''Pseudotsuga menziezii'').
Fungal diseases
{, class="wikitable" style="clear"
! colspan=2, Fungal diseases
, -
, Annosus root disease
, , '' Heterobasidion annosum''
'' Spiniger meineckellu ...
* List of dahlia diseases
* List of foliage plant diseases (Araceae)
This is a list of diseases of foliage plants belonging to the Araceae
The Araceae are a family of monocotyledonous flowering plants in which flowers are borne on a type of inflorescence called a spadix. The spadix is usually accompanied by, and ...
* List of foliage plant diseases (Acanthaceae)
This is a list of diseases of foliage plants belonging to the Acanthaceae.
Plant species
Bacterial diseases
Fungal diseases
Viral diseases
ReferencesCommon Names of Diseases, The American Phytopathological Society
{{DEFAULTSORT:Lis ...
* List of foliage plant diseases (Agavaceae)
* List of foliage plant diseases (Araliaceae)
* {{DEFAULTSORT:Lis ...
List of foliage plant diseases (Asclepiadaceae)
This is a list of diseases of foliage plants belonging to the family Asclepiadaceae.
Plant Species
Fungal diseases
ReferencesCommon Names of Diseases, The American Phytopathological Society
{{DEFAULTSORT:List Of Foliage Plant Diseases (Ascle ...
* List of foliage plant diseases (Gesneriaceae)
This is a list of diseases of foliage plants belonging to the family Gesneriaceae
Gesneriaceae, the gesneriad family, is a family of flowering plants consisting of about 152 genera and ca. 3,540 species in the tropics and subtropics of the Ol ...
* List of Ficus diseases
* List of foliage plant diseases (Polypodiaceae)
This is a list of diseases of foliage plants belonging to the family Polypodiaceae
Polypodiaceae is a family of ferns. In the Pteridophyte Phylogeny Group classification of 2016 (PPG I), the family includes around 65 genera and an estimated 1,65 ...
* List of foliage plant diseases (Vitaceae)
This is a list of diseases of foliage plants belonging to the family Vitaceae.
Plant Species
Fungal diseases
{, class="wikitable" style="clear"
! colspan=3, Fungal diseases
, -
, Common name
, , Scientific name
, , Plants affected
, -
, An ...
* List of rhododendron diseases
This article is a list of diseases of rhododendron (''Rhododendron'' spp.).
Bacterial diseases
Fungal diseases
Nematodes, parasitic
Viral diseases
References
{{reflist
Common Names of Diseases, The American Phytopathological Society ...
Biology
''Botrytis cinerea'' is characterized by abundant hyalineconidia
A conidium ( ; ), sometimes termed an asexual chlamydospore or chlamydoconidium (), is an asexual, non- motile spore of a fungus. The word ''conidium'' comes from the Ancient Greek word for dust, ('). They are also called mitospores due to t ...
(asexual spores) borne on grey, branching tree-like conidiophore
A conidium ( ; ), sometimes termed an asexual chlamydospore or chlamydoconidium (), is an asexual, non-motile spore of a fungus. The word ''conidium'' comes from the Ancient Greek word for dust, ('). They are also called mitospores due to the ...
s. The fungus also produces highly resistant sclerotia
A sclerotium (; (), is a compact mass of hardened fungal mycelium containing food reserves. One role of sclerotia is to survive environmental extremes. In some higher fungi such as ergot, sclerotia become detached and remain dormant until favor ...
as survival structures in older cultures. It overwinters as sclerotia or intact mycelia
Mycelium (plural mycelia) is a root-like structure of a fungus consisting of a mass of branching, thread-like hyphae. Fungal colonies composed of mycelium are found in and on soil and many other substrates. A typical single spore germinates in ...
, both of which germinate in spring to produce conidiophores. The conidia, dispersed by wind and by rain-water, cause new infections. ''B. cinerea'' performs an asexual cycle over the summer season.
Different strains show considerable genetic variability.
''Gliocladium roseum
''Clonostachys rosea f. rosea'', also known as ''Gliocladium roseum'', is a species of fungus in the family Bionectriaceae. It colonizes living plants as an endophyte, digests material in soil as a saprophyte and is also known as a parasite of ...
'' is a fungal parasite of ''B. cinerea''.
The hypothetical protein BcKMO was shown to positively regulate growth and development. It showed a great similarity to the kynurenine 3-monooxygenase encoding gene in eukaryotes.
Overexpression of the gene ' produces altered versions of the transcription factor '' mrr1'', which in turn confer a multiple fungicide resistance
Fungicides are biocidal chemical compounds or biological organisms used to kill parasitic fungi or their spores. A fungistatic inhibits their growth. Fungi can cause serious damage in agriculture, resulting in critical losses of yield, quality ...
phenotype known as . An even higher overexpression yields ''mrr1'' composed partly of , yielding MDR1h phenotypes with even more anilinopyramidine- and phenylpyrrole- resistance.
Environment
Gray mold favors moist, humid, and warm environmental conditions between . Temperature, relative humidity, and wetness duration produce a conducive environment that is favorable for inoculation ofmycelium
Mycelium (plural mycelia) is a root-like structure of a fungus consisting of a mass of branching, thread-like hyphae. Fungal colonies composed of mycelium are found in and on soil and many other substrates. A typical single spore germinates ...
or conidia
A conidium ( ; ), sometimes termed an asexual chlamydospore or chlamydoconidium (), is an asexual, non- motile spore of a fungus. The word ''conidium'' comes from the Ancient Greek word for dust, ('). They are also called mitospores due to t ...
. Controlled environments, such as crop production greenhouses, provide the moisture and high temperatures that favor the spreading and development of the pathogen ''B. cinerea.''
Standing water on plant leaf surfaces provides a place for spores to germinate. Humid conditions can result from improper irrigation practice, plants placed too close together, or the structure of the greenhouse not allowing for efficient ventilation and air flow. Ventilation at night significantly reduces the incidence of gray mold.
Melanized sclerotium allows ''B. cinerea'' to survive for years in the soil. Sclerotia and the asexual conidia spores contribute to the widespread infection of the pathogen.
A low pH is preferred by the gray mold to perform well. ''B. cinerea'' can acidify its environment by secreting organic acid
An organic acid is an organic compound with acidic properties. The most common organic acids are the carboxylic acids, whose acidity is associated with their carboxyl group –COOH. Sulfonic acids, containing the group –SO2OH, are ...
s, like oxalic acid. By acidifying its surroundings, cell wall degrading enzymes (CWDEs) are enhanced, plant-protection enzymes are inhibited, stoma
In botany, a stoma (from Greek ''στόμα'', "mouth", plural "stomata"), also called a stomate (plural "stomates"), is a pore found in the epidermis of leaves, stems, and other organs, that controls the rate of gas exchange. The pore is bo ...
tal closure is deregulated, and pH signaling is mediated to facilitate its pathogenesis
Pathogenesis is the process by which a disease or disorder develops. It can include factors which contribute not only to the onset of the disease or disorder, but also to its progression and maintenance. The word comes from Greek πάθος ''pat ...
.
Viticulture
In the ''Botrytis'' infection known as "noble rot" ('' pourriture noble'' inFrench
French (french: français(e), link=no) may refer to:
* Something of, from, or related to France
** French language, which originated in France, and its various dialects and accents
** French people, a nation and ethnic group identified with Franc ...
, or '' Edelfäule'' in German
German(s) may refer to:
* Germany (of or related to)
**Germania (historical use)
* Germans, citizens of Germany, people of German ancestry, or native speakers of the German language
** For citizens of Germany, see also German nationality law
**Ge ...
), the fungus removes water from the grapes, leaving behind a higher percent of solids, such as sugars, fruit acids and minerals. This results in a more intense, concentrated final product. The wine is often said to have an aroma of honeysuckle
Honeysuckles are arching shrubs or twining vines in the genus ''Lonicera'' () of the family Caprifoliaceae, native to northern latitudes in North America and Eurasia. Approximately 180 species of honeysuckle have been identified in both con ...
and a bitter finish on the palate.
A distinct fermentation process initially caused by nature, the combination of geology, climate and specific weather led to the particular balance of beneficial fungus while leaving enough of the grape intact for harvesting. The Chateau d'Yquem is the only Premier Cru Supérieur, largely due to the vineyard's susceptibility to noble rot.
''Botrytis'' complicates winemaking by making fermentation
Fermentation is a metabolic process that produces chemical changes in organic substrates through the action of enzymes. In biochemistry, it is narrowly defined as the extraction of energy from carbohydrates in the absence of oxygen. In food p ...
more complex. ''Botrytis'' produces an anti-fungal compound that kills yeast
Yeasts are eukaryotic, single-celled microorganisms classified as members of the fungus kingdom. The first yeast originated hundreds of millions of years ago, and at least 1,500 species are currently recognized. They are estimated to constit ...
and often results in the fermentation stopping before the wine has accumulated sufficient levels of alcohol.
Botrytis bunch rot is another condition of grapes caused by ''B. cinerea'' that causes great losses for the wine industry. It is always present on the fruitset, however, it requires a wound to start a bunch rot infection. Wounds can come from insects, wind, accidental damage, etc. To control botrytis bunch rot there are a number of fungicide
Fungicides are biocidal chemical compounds or biological organisms used to kill parasitic fungi or their spores. A fungistatic inhibits their growth. Fungi can cause serious damage in agriculture, resulting in critical losses of yield, quality ...
s available on the market. Generally, these should be applied at bloom, bunch closure and veraison (the most important being the bloom application). Some winemakers are known to use the German method of fermentation and prefer having a 5% bunch rot rate in their grapes and will usually hold the grapes on the vine a week longer than normal.
Horticulture
''Botrytis cinerea'' affects many other plants.Strawberries
It is economically important on soft fruits such asstrawberries
The garden strawberry (or simply strawberry; ''Fragaria × ananassa'') is a widely grown hybrid species of the genus '' Fragaria'', collectively known as the strawberries, which are cultivated worldwide for their fruit. The fruit is widely ap ...
and bulb crops. Unlike wine grapes, the affected strawberries are not edible and are discarded. To minimize infection in strawberry fields, good ventilation around the berries is important to prevent moisture being trapped among leaves and berries. A number of bacteria have been proven to act as natural antagonists to ''B. cinerea'' in controlled studies.Donmez, M. F.; Esitken, A.; Yildiz, H.; Ercisli, SBiocontrol of ''Botrytis Cinerea'' on Strawberry Fruit by Plant Growth Promoting Bacteria
''The Journal of Animal & Plant Sciences'', 21(4), 2011: pp. 758-763, ISSN 1018-7081.
Other plants
 In greenhouse horticulture, ''Botrytis cinerea'' is well known as a cause of considerable damage in
In greenhouse horticulture, ''Botrytis cinerea'' is well known as a cause of considerable damage in tomato
The tomato is the edible berry of the plant ''Solanum lycopersicum'', commonly known as the tomato plant. The species originated in western South America, Mexico, and Central America. The Mexican Nahuatl word gave rise to the Spanish word ...
es.
The infection also affects rhubarb
Rhubarb is the fleshy, edible stalks ( petioles) of species and hybrids (culinary rhubarb) of '' Rheum'' in the family Polygonaceae, which are cooked and used for food. The whole plant – a herbaceous perennial growing from short, thick rhi ...
, snowdrop
''Galanthus'' (from Ancient Greek , (, "milk") + (, "flower")), or snowdrop, is a small genus of approximately 20 species of bulbous perennial herbaceous plants in the family Amaryllidaceae. The plants have two linear leaves and a single sm ...
s, white meadowfoam
''Limnanthes alba'' is a species of flowering plant in the meadowfoam family known by the common name white meadowfoam. It is native to California and Oregon, where it grows in wet grassy habitat, such as vernal pools and moist spots in woodlan ...
, western hemlock
''Tsuga heterophylla'', the western hemlock or western hemlock-spruce, is a species of hemlock native to the west coast of North America, with its northwestern limit on the Kenai Peninsula, Alaska, and its southeastern limit in northern Sonoma ...
, Douglas-fir
The Douglas fir (''Pseudotsuga menziesii'') is an evergreen conifer species in the pine family, Pinaceae. It is native to western North America and is also known as Douglas-fir, Douglas spruce, Oregon pine, and Columbian pine. There are three va ...
cannabis
''Cannabis'' () is a genus of flowering plants in the family Cannabaceae. The number of species within the genus is disputed. Three species may be recognized: '' Cannabis sativa'', '' C. indica'', and '' C. ruderalis''. Alternative ...
, and '' Lactuca sativa''. UV-C treatment against ''B. cinerea'' was investigated by Vàsquez ''et al.'', 2017. They find it increases phenylalanine ammonia-lyase
The enzyme phenylalanine ammonia lyase (EC 4.3.1.24) catalyzes the conversion of L-phenylalanine to ammonia and ''trans''-cinnamic acid.:
:L-phenylalanine = ''trans''-cinnamate + NH3
Phenylalanine ammonia lyase (PAL) is the first and committed ...
activity and production of phenolics. This in turn decreases ''L. sativa''s susceptibility. Potassium bicarbonate-based fungicide may be used.
Human disease
''Botrytis cinerea'' mold on grapes may cause "winegrower's lung", a rare form ofhypersensitivity pneumonitis
Hypersensitivity pneumonitis (HP) or extrinsic allergic alveolitis (EAA) is a syndrome caused by the repetitive inhalation of antigens from the environment in susceptible or sensitized people. Common antigens include molds, bacteria, bird dropping ...
(a respiratory allergic reaction in predisposed individuals).
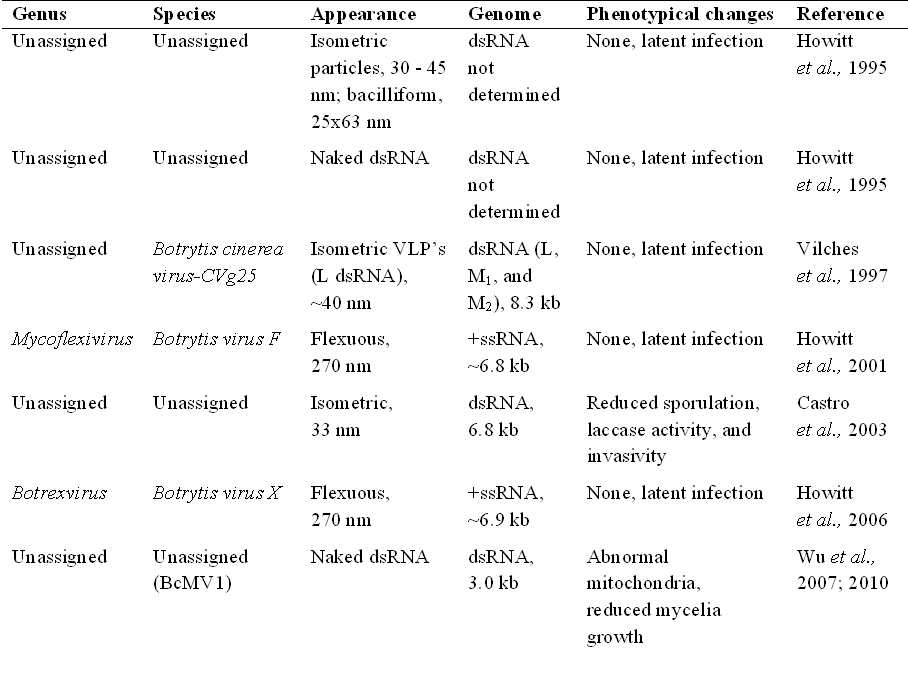
Mycoviruses of ''Botrytis cinerea''
 ''Botrytis cinerea'' not only infects plants, it also hosts several mycoviruses itself (see the table/image).
A range of phenotypic alterations due to the mycoviral infection have been observed from symptomless to mild impact, or more severe phenotypic changes including reduction in pathogenicity, growth/suppression of mycelia, sporulation and sclerotia production, formation of abnormal colony sectors (Wu et al., 2010) and virulence.
''Botrytis cinerea'' not only infects plants, it also hosts several mycoviruses itself (see the table/image).
A range of phenotypic alterations due to the mycoviral infection have been observed from symptomless to mild impact, or more severe phenotypic changes including reduction in pathogenicity, growth/suppression of mycelia, sporulation and sclerotia production, formation of abnormal colony sectors (Wu et al., 2010) and virulence.
Management
''Botrytis cinerea'' can be managed through cultural, chemical, and biological practices. There are no resistant species to the gray mold rot. Gray mold can be culturally controlled by monitoring the amount and timing of fertilizer applications to reduce the amount of fruit rot. Excessive application of nitrogen will increase the incidence of disease while not improving yields. Not plantingcultivar
A cultivar is a type of cultivated plant that people have selected for desired traits and when propagated retain those traits. Methods used to propagate cultivars include: division, root and stem cuttings, offsets, grafting, tissue culture ...
s that have an upright or dense growth habit can reduce disease as these limit airflow and are favorable for the pathogen. Spacing of plants so they are not touching will increase airflow allowing the area to dry out and reduce the spread of disease. Pruning
Pruning is a horticultural, arboricultural, and silvicultural practice involving the selective removal of certain parts of a plant, such as branches, buds, or roots.
The practice entails the ''targeted'' removal of diseased, damaged, dead, ...
or purposeful removal of diseased, dead, or overgrown limbs on a regular schedule can also help to improve air movement.
Sanitation
Sanitation refers to public health conditions related to clean drinking water and treatment and disposal of human excreta and sewage. Preventing human contact with feces is part of sanitation, as is hand washing with soap. Sanitation syste ...
by removing dead or dying plant tissue in the fall will decrease inoculum levels as there is no debris for the sclerotium or mycelia to overwinter. Removing debris in the spring will remove inoculum from the site. Disposal of berries during harvest that have signs and symptoms of gray mold will reduce inoculum for the following year.
Biochar
Biochar is the lightweight black residue, made of carbon and
ashes, remaining after the pyrolysis of biomass. Biochar is defined by the International Biochar Initiative as "the solid material obtained from the thermochemical conversion of ...
, a form of charcoal, can be applied as a soil amendment
A soil conditioner is a product which is added to soil to improve the soil’s physical qualities, usually its fertility (ability to provide nutrition for plants) and sometimes its mechanics. In general usage, the term "soil conditioner" is often ...
to strawberry plants to reduce the severity of the fungal disease by stimulating defense pathways within the plant.
Gray mold can be chemically controlled with well-timed fungicide applications starting during the first bloom. Timing can reduce the chance of resistance and will save on costs.
Biological controls or microbial antagonists used for disease suppression, have been successfully used in Europe and Brazil in the form of fungi-like ''Trichoderma harzianum
''Trichoderma harzianum'' is a fungus that is also used as a fungicide. It is used for foliar application, seed treatment and soil treatment for suppression of fungal pathogens causing various fungal plant diseases. Commercial biotechnological p ...
'' Rifai and ''Clonostachys rosea'' f. ''rosea'' Bainier (syn. ''Gliocladium roseum''). ''Trichoderma'' species especially, have been shown to control gray mold.
Multiple fungicide resistance
Fungicides are biocidal chemical compounds or biological organisms used to kill parasitic fungi or their spores. A fungistatic inhibits their growth. Fungi can cause serious damage in agriculture, resulting in critical losses of yield, quality ...
is a problem in many production areas.
See also
* BotrydialReferences
External links
Genome information for ''Botrytis cinerea''
Genome analysis of ''Botrytis cinerea''
*
TheWineDoctor.com
* * * {{Authority control Sclerotiniaceae Fungal plant pathogens and diseases Small fruit diseases Fungal strawberry diseases Fungal citrus diseases Fungal grape diseases Oenology Fungi described in 1794