Avogadro constant on:
[Wikipedia]
[Google]
[Amazon]
The Avogadro constant, commonly denoted or , is an SI defining constant with an exact value of when expressed in reciprocal moles.
It defines the ratio of the number of constituent particles to the
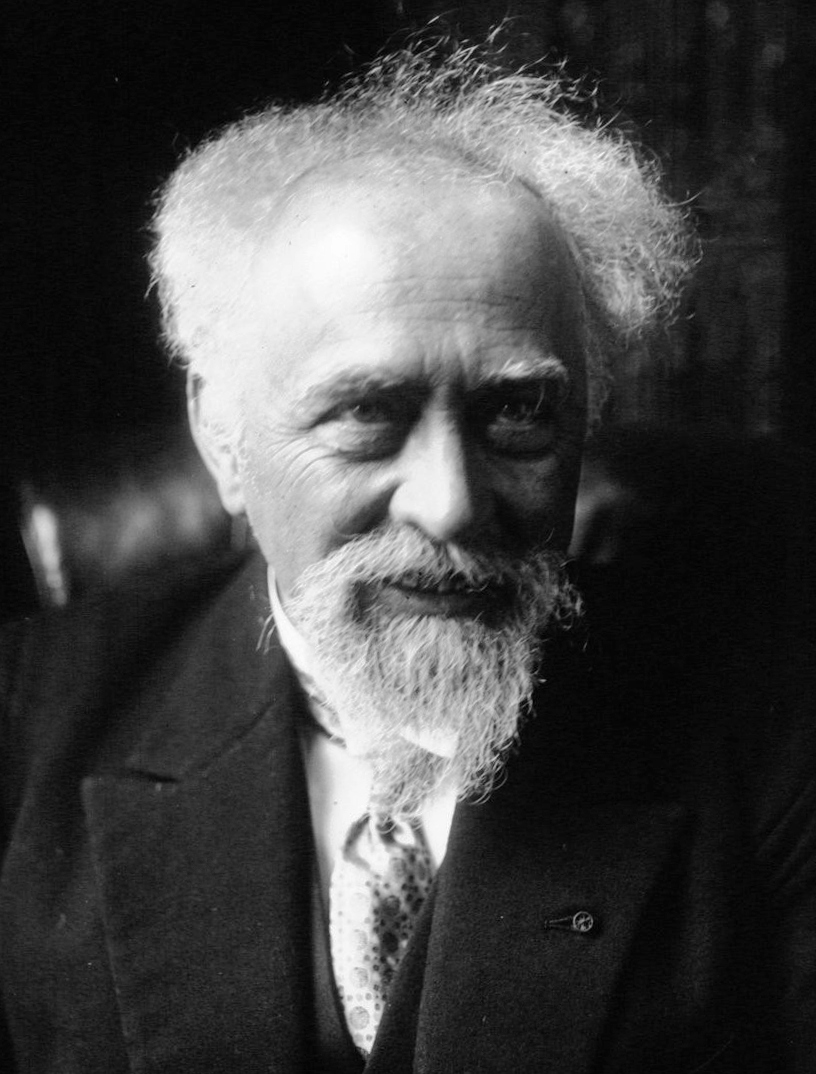 The Avogadro constant is named after the Italian scientist Amedeo Avogadro (1776–1856), who, in 1811, first proposed that the volume of a gas (at a given pressure and temperature) is proportional to the number of
The Avogadro constant is named after the Italian scientist Amedeo Avogadro (1776–1856), who, in 1811, first proposed that the volume of a gas (at a given pressure and temperature) is proportional to the number of
 The value of Avogadro's number (not yet known by that name) was first obtained indirectly by Josef Loschmidt in 1865, by estimating the number of particles in a given volume of gas. This value, the number density of particles in an
The value of Avogadro's number (not yet known by that name) was first obtained indirectly by Josef Loschmidt in 1865, by estimating the number of particles in a given volume of gas. This value, the number density of particles in an
1996 definition of the Avogadro constant
from the
Some Notes on Avogadro's Number,
''(historical notes)''
An Exact Value for Avogadro's Number
– '' American Scientist''
Avogadro and molar Planck constants for the redefinition of the kilogram
* * Scanned version of "Two hypothesis of Avogadro", 1811 Avogadro's article, on
BibNum
' {{Authority control Amount of substance Fundamental constants
amount of substance
In chemistry, the amount of substance (symbol ) in a given sample of matter is defined as a ratio () between the particle number, number of elementary entities () and the Avogadro constant (). The unit of amount of substance in the International ...
in a sample, where the particles in question are any designated elementary entity, such as molecule
A molecule is a group of two or more atoms that are held together by Force, attractive forces known as chemical bonds; depending on context, the term may or may not include ions that satisfy this criterion. In quantum physics, organic chemi ...
s, atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
s, ions, ion pairs. The numerical value of this constant is known as the Avogadro number, commonly denoted . The Avogadro ''number'' is an exact number equal to the number of constituent particles in one mole of any substance (by definition of the mole), historically derived from the experimental determination of the number of atoms in 12 grams of carbon-12
Carbon-12 (12C) is the most abundant of the two stable isotopes of carbon ( carbon-13 being the other), amounting to 98.93% of element carbon on Earth; its abundance is due to the triple-alpha process by which it is created in stars. Carbon-1 ...
(12C) before the 2019 revision of the SI
In 2019, four of the seven SI base units specified in the International System of Quantities were redefined in terms of natural physical constants, rather than human artefacts such as the standard kilogram.
Effective 20 May 2019, the 144th ...
. Both the constant and the number are named after the Italian physicist and chemist Amedeo Avogadro.
The Avogadro constant is used as a proportionality factor in relating the ''amount of substance
In chemistry, the amount of substance (symbol ) in a given sample of matter is defined as a ratio () between the particle number, number of elementary entities () and the Avogadro constant (). The unit of amount of substance in the International ...
'' , in a sample of a substance , to the corresponding number of elementary entities :
:
The Avogadro constant is also the factor that converts the average mass
Mass is an Intrinsic and extrinsic properties, intrinsic property of a physical body, body. It was traditionally believed to be related to the physical quantity, quantity of matter in a body, until the discovery of the atom and particle physi ...
of one particle of a substance to its molar mass
In chemistry, the molar mass () (sometimes called molecular weight or formula weight, but see related quantities for usage) of a chemical substance ( element or compound) is defined as the ratio between the mass () and the amount of substance ...
. That is, . Applying this equation to 12C with an atomic mass of exactly 12 Da and a molar mass of 12 g/mol yields (after rearrangement) the following relation for the Avogadro constant: = (g/Da) mol−1, making the Avogadro number = g/Da. Historically, this was precisely true, but since the 2019 revision of the SI, the relation is now merely approximate, although equality may still be assumed with high accuracy.
The constant also relates the molar volume
In chemistry and related fields, the molar volume, symbol ''V''m, or \tilde V of a substance is the ratio of the volume (''V'') occupied by a substance to the amount of substance (''n''), usually at a given temperature and pressure. It is also eq ...
(the volume per mole) of a substance to the average volume nominally occupied by one of its particles, when both are expressed in the same units of volume. For example, since the molar volume of water in ordinary conditions is about , the volume occupied by one molecule of water is about , or about (cubic nanometres). For a crystal
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macros ...
line substance, relates the volume of a crystal with one mole worth of repeating unit cells, to the volume of a single cell (both in the same units).
Definition
The Avogadro constant was historically derived from the old definition of the mole as theamount of substance
In chemistry, the amount of substance (symbol ) in a given sample of matter is defined as a ratio () between the particle number, number of elementary entities () and the Avogadro constant (). The unit of amount of substance in the International ...
in 12 grams of carbon-12
Carbon-12 (12C) is the most abundant of the two stable isotopes of carbon ( carbon-13 being the other), amounting to 98.93% of element carbon on Earth; its abundance is due to the triple-alpha process by which it is created in stars. Carbon-1 ...
(12C). By this old definition, the numerical value of the Avogadro constant in mol−1 (the Avogadro number) was a physical constant that had to be determined experimentally.
The historical relationship of the Avogadro constant to the molar mass
In chemistry, the molar mass () (sometimes called molecular weight or formula weight, but see related quantities for usage) of a chemical substance ( element or compound) is defined as the ratio between the mass () and the amount of substance ...
of carbon-12, , and its atomic mass, , can be expressed in the following equation: Thus, , the numerical value of , was equal to the number of daltons in a gram (g/Da), where the dalton is defined as of the mass of a 12C atom.
The redefinition of the mole in 2019, as being the amount of substance consisting of exactly elementary entities, means that the mass of 1 mole of a substance is now exactly the product of the Avogadro number and the average mass of one of the entities involved. The dalton, however, is still defined as of the mass of a 12C atom, which must be determined experimentally and is known only with finite accuracy. Thus, prior experiments that aimed to determine the numerical value of the Avogadro constant when expressed in reciprocal moles—i.e. the Avogadro number (now numerically fixed)—are re-interpreted as measurements of the numerical value in grams of the dalton.
By the old definition of mole, the numerical value of the mass of one mole of a substance expressed in grams (i.e., its molar mass in g/mol or kg/kmol), was precisely equal to the average mass of one particle expressed in daltons. With the new definition, this numerical equivalence is no longer exact, as it is affected by the uncertainty in the value of the gram-to-dalton (g/Da) mass-unit ratio. However, it may still be assumed for all practical purposes. For example, the average mass of one molecule of water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known liv ...
is about 18.0153 daltons, and an amount of one mole of water has a corresponding macroscopic mass of about 18.0153 grams. Also, the Avogadro number is the approximate number of nucleon
In physics and chemistry, a nucleon is either a proton or a neutron, considered in its role as a component of an atomic nucleus. The number of nucleons in a nucleus defines the atom's mass number.
Until the 1960s, nucleons were thought to be ele ...
s (proton
A proton is a stable subatomic particle, symbol , Hydron (chemistry), H+, or 1H+ with a positive electric charge of +1 ''e'' (elementary charge). Its mass is slightly less than the mass of a neutron and approximately times the mass of an e ...
s and neutron
The neutron is a subatomic particle, symbol or , that has no electric charge, and a mass slightly greater than that of a proton. The Discovery of the neutron, neutron was discovered by James Chadwick in 1932, leading to the discovery of nucle ...
s) in one gram of ordinary matter
In classical physics and general chemistry, matter is any substance that has mass and takes up space by having volume. All everyday objects that can be touched are ultimately composed of atoms, which are made up of interacting subatomic pa ...
.
An amount of substance consisting of just a single elementary entity might be thought of as an "elementary amount", analogous to the elementary charge
The elementary charge, usually denoted by , is a fundamental physical constant, defined as the electric charge carried by a single proton (+1 ''e'') or, equivalently, the magnitude of the negative electric charge carried by a single electron, ...
, . Letting denote this elementary amount, then , with the mole defined such that , which can be rearranged as . Thus, , the reciprocal of the Avogadro constant. The fundamental definition of the Avogadro constant itself is therefore one per elementary amount (), independent of any macroscopic base unit chosen for the physical quantity. (Since there is an aggregate of an Avogadro number of elementary entities in one mole, the Avogadro constant can also be ''expressed'' (in terms of the mole) as an Avogadro number per mole—but this is ''not'' its "definition".) The Avogadro constant, a well-defined quantity value (with dimension 1/N), independent of the mole, is therefore a ''bona fide'' defining constant for the 2019 redefinition of the mole.
Introducing in place of , means that —amount of substance is an aggregate of elementary entities—which is easier to comprehend than "reciprocal Avogadro constants". Also the molar mass is then —the entity mass per entity, which is self-evident.
In older literature, the Avogadro number was also denoted , although that conflicts with the symbol for number of particles
In thermodynamics, the particle number (symbol ) of a thermodynamic system is the number of constituent particles in that system. The particle number is a fundamental thermodynamic property which is conjugate to the chemical potential. Unlike m ...
in statistical mechanics
In physics, statistical mechanics is a mathematical framework that applies statistical methods and probability theory to large assemblies of microscopic entities. Sometimes called statistical physics or statistical thermodynamics, its applicati ...
.
History
Origin of the concept
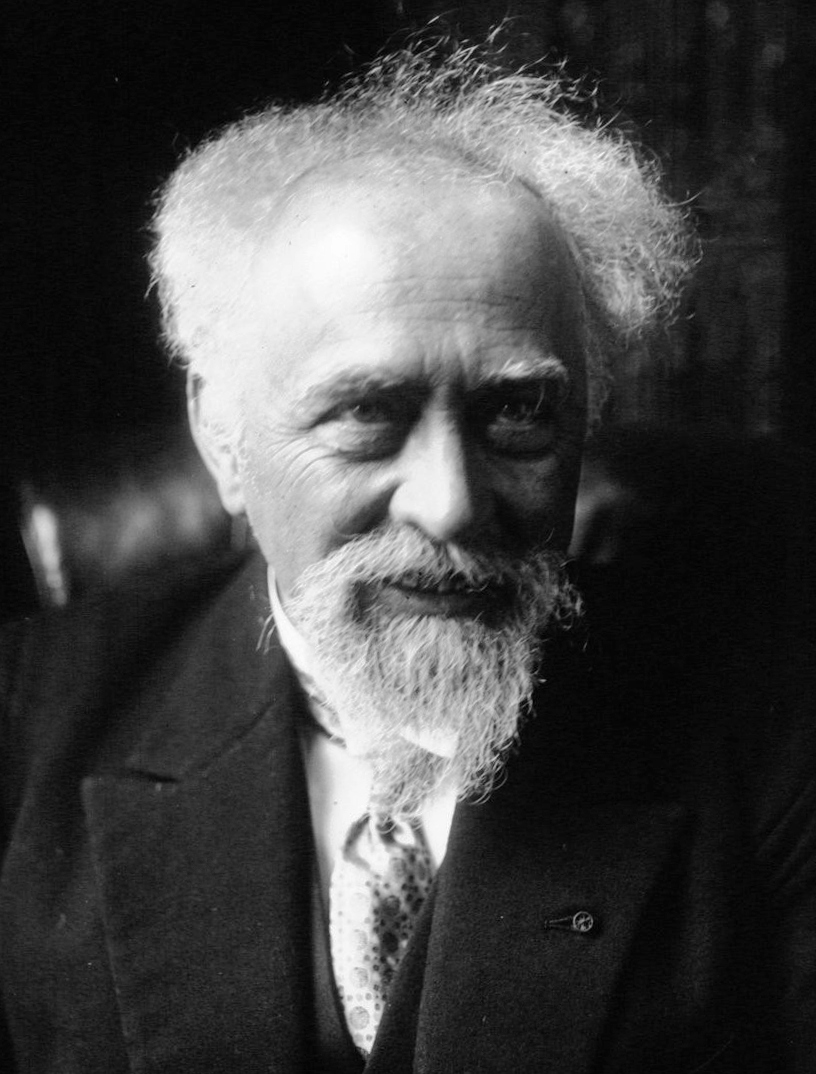 The Avogadro constant is named after the Italian scientist Amedeo Avogadro (1776–1856), who, in 1811, first proposed that the volume of a gas (at a given pressure and temperature) is proportional to the number of
The Avogadro constant is named after the Italian scientist Amedeo Avogadro (1776–1856), who, in 1811, first proposed that the volume of a gas (at a given pressure and temperature) is proportional to the number of atom
Atoms are the basic particles of the chemical elements. An atom consists of a atomic nucleus, nucleus of protons and generally neutrons, surrounded by an electromagnetically bound swarm of electrons. The chemical elements are distinguished fr ...
s or molecule
A molecule is a group of two or more atoms that are held together by Force, attractive forces known as chemical bonds; depending on context, the term may or may not include ions that satisfy this criterion. In quantum physics, organic chemi ...
s regardless of the nature of the gas.
Avogadro's hypothesis was popularized four years after his death by Stanislao Cannizzaro, who advocated Avogadro's work at the Karlsruhe Congress in 1860.
The name ''Avogadro's number'' was coined in 1909 by the physicist Jean Perrin, who defined it as the number of molecules in exactly 32 grams of oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
gas. The goal of this definition was to make the mass of a mole of a substance, in grams, be numerically equal to the mass of one molecule relative to the mass of the hydrogen atom; which, because of the law of definite proportions
In chemistry, the law of definite proportions, sometimes called Proust's law or the law of constant composition, states that a given
chemical compound contains its constituent elements in a fixed ratio (by mass) and does not depend on its source ...
, was the natural unit of atomic mass, and was assumed to be of the atomic mass of oxygen.
First measurements
 The value of Avogadro's number (not yet known by that name) was first obtained indirectly by Josef Loschmidt in 1865, by estimating the number of particles in a given volume of gas. This value, the number density of particles in an
The value of Avogadro's number (not yet known by that name) was first obtained indirectly by Josef Loschmidt in 1865, by estimating the number of particles in a given volume of gas. This value, the number density of particles in an ideal gas
An ideal gas is a theoretical gas composed of many randomly moving point particles that are not subject to interparticle interactions. The ideal gas concept is useful because it obeys the ideal gas law, a simplified equation of state, and is ...
, is now called the Loschmidt constant in his honor, and is related to the Avogadro constant, , by
:
where is the pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and eve ...
, is the gas constant, and is the absolute temperature
Thermodynamic temperature, also known as absolute temperature, is a physical quantity which measures temperature starting from absolute zero, the point at which particles have minimal thermal motion.
Thermodynamic temperature is typically expres ...
. Because of this work, the symbol is sometimes used for the Avogadro constant, and, in German literature, that name may be used for both constants, distinguished only by the units of measurement
A unit of measurement, or unit of measure, is a definite magnitude (mathematics), magnitude of a quantity, defined and adopted by convention or by law, that is used as a standard for measurement of the same kind of quantity. Any other qua ...
. (However, should not be confused with the entirely different Loschmidt constant in English-language literature.)
Perrin himself determined the Avogadro number, which he called "Avogadro's constant" (constante d'Avogadro), by several different experimental methods. He was awarded the 1926 Nobel Prize in Physics
The Nobel Prize in Physics () is an annual award given by the Royal Swedish Academy of Sciences for those who have made the most outstanding contributions to mankind in the field of physics. It is one of the five Nobel Prizes established by the ...
, largely for this work.
The electric charge per mole of electrons is a constant called the Faraday constant
In physical chemistry, the Faraday constant (symbol , sometimes stylized as ℱ) is a physical constant defined as the quotient of the total electric charge () by the amount () of elementary charge carriers in any given sample of matter: it ...
and has been known since 1834, when Michael Faraday
Michael Faraday (; 22 September 1791 – 25 August 1867) was an English chemist and physicist who contributed to the study of electrochemistry and electromagnetism. His main discoveries include the principles underlying electromagnetic inducti ...
published his works on electrolysis. In 1910, Robert Millikan with the help of Harvey Fletcher obtained the first measurement of the charge on an electron. Dividing the charge on a mole of electrons by the charge on a single electron provided a more accurate estimate of the Avogadro number.
SI definition of 1971
In 1971, in its 14th conference, theInternational Bureau of Weights and Measures
The International Bureau of Weights and Measures (, BIPM) is an List of intergovernmental organizations, intergovernmental organisation, through which its 64 member-states act on measurement standards in areas including chemistry, ionising radi ...
(BIPM) decided to regard the amount of substance
In chemistry, the amount of substance (symbol ) in a given sample of matter is defined as a ratio () between the particle number, number of elementary entities () and the Avogadro constant (). The unit of amount of substance in the International ...
as an independent dimension of measurement, with the mole as its base unit in the International System of Units
The International System of Units, internationally known by the abbreviation SI (from French ), is the modern form of the metric system and the world's most widely used system of measurement. It is the only system of measurement with official s ...
(SI). Specifically, the mole was defined as the amount of a substance that contains as many elementary entities as there are atoms in () of carbon-12
Carbon-12 (12C) is the most abundant of the two stable isotopes of carbon ( carbon-13 being the other), amounting to 98.93% of element carbon on Earth; its abundance is due to the triple-alpha process by which it is created in stars. Carbon-1 ...
(12C). Thus, in particular, an amount of one mole of carbon 12 had a corresponding mass that was ''exactly'' of that element.
By this definition, one mole of any substance contained exactly as many elementary entities as one mole of any other substance. However, this number was a physical constant that had to be experimentally determined since it depended on the mass (in grams) of one atom of 12C, and therefore, it was known only to a limited number of decimal digits. The common rule of thumb that "one gram of matter contains nucleons" was exact for carbon-12, but slightly inexact for other elements and isotopes.
In the same conference, the BIPM also named (the factor that related the amount of a substance to the corresponding number of particles) the "Avogadro ''constant''". However, the term "Avogadro number" continued to be used, especially in introductory works. As a consequence of this definition, was not a pure number, but had the quantity dimension of reciprocal of amount of substance (N−1).
SI redefinition of 2019
In its 26th Conference, the BIPM adopted a different approach: effective 20 May 2019, it defined the Avogadro constant as the exact value , thus redefining the mole as exactly constituent particles of the substance under consideration. One consequence of this change is that the mass of a mole of 12C atoms is no longer exactly 0.012 kg. On the other hand, the dalton, Da ( unified atomic mass unit, u), remains unchanged as of the mass of 12C. Thus, the molar mass constant remains very close to but no longer exactly equal to 1 g/mol, although the difference ( in relative terms, as of March 2019) is insignificant for all practical purposes.Connection to other constants
The Avogadro constant is related to other physical constants and properties. * It relates the molar gas constant and theBoltzmann constant
The Boltzmann constant ( or ) is the proportionality factor that relates the average relative thermal energy of particles in a ideal gas, gas with the thermodynamic temperature of the gas. It occurs in the definitions of the kelvin (K) and the ...
, which in the SI is defined to be exactly :
*:
* It relates the Faraday constant
In physical chemistry, the Faraday constant (symbol , sometimes stylized as ℱ) is a physical constant defined as the quotient of the total electric charge () by the amount () of elementary charge carriers in any given sample of matter: it ...
and the elementary charge
The elementary charge, usually denoted by , is a fundamental physical constant, defined as the electric charge carried by a single proton (+1 ''e'') or, equivalently, the magnitude of the negative electric charge carried by a single electron, ...
, which in the SI is defined as exactly :
*:
* It relates the molar mass constant and the atomic mass constant currently
*:
See also
* Committee on Data of the International Science Council * List of scientists whose names are used in physical constants * Mole DayReferences
External links
1996 definition of the Avogadro constant
from the
IUPAC
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...
'' Compendium of Chemical Terminology'' ("''Gold Book''")
Some Notes on Avogadro's Number,
''(historical notes)''
An Exact Value for Avogadro's Number
– '' American Scientist''
Avogadro and molar Planck constants for the redefinition of the kilogram
* * Scanned version of "Two hypothesis of Avogadro", 1811 Avogadro's article, on
BibNum
' {{Authority control Amount of substance Fundamental constants