Β-sitosterol on:
[Wikipedia]
[Google]
[Amazon]
β-sitosterol (beta-sitosterol) is one of several
 The regulation of the biosynthesis of both sterols and some specific lipids occurs during membrane biogenesis. Through 13C-labeling patterns, it has been determined that both the mevalonate and deoxyxylulose pathways are involved in the formation of β-sitosterol. The precise mechanism of β-sitosterol formation varies according to the organism, but is generally found to come from cycloartenol.Dewick, P. M. Medicinal Natural Products: A Biosynthetic Approach. 3 ed.; John Wiley & Sons Ltd.: United Kingdom cyclization, 2009; p 539.
The biosynthesis of cycloartenol begins as one molecule of isopentenyl diphosphate (IPP) and two molecules of dimethylallyl diphosphate (DMAPP) form farnesyl diphosphate (FPP). Two molecules of FPP are then joined tail-to-tail to yield
The regulation of the biosynthesis of both sterols and some specific lipids occurs during membrane biogenesis. Through 13C-labeling patterns, it has been determined that both the mevalonate and deoxyxylulose pathways are involved in the formation of β-sitosterol. The precise mechanism of β-sitosterol formation varies according to the organism, but is generally found to come from cycloartenol.Dewick, P. M. Medicinal Natural Products: A Biosynthetic Approach. 3 ed.; John Wiley & Sons Ltd.: United Kingdom cyclization, 2009; p 539.
The biosynthesis of cycloartenol begins as one molecule of isopentenyl diphosphate (IPP) and two molecules of dimethylallyl diphosphate (DMAPP) form farnesyl diphosphate (FPP). Two molecules of FPP are then joined tail-to-tail to yield
phytosterol
Phytosterols are phytosteroids, similar to cholesterol, that serve as structural components of biological membranes of plants. They encompass plant sterols and stanols. More than 250 sterols and related compounds have been identified. Free phy ...
s (plant sterols) with chemical structure
A chemical structure determination includes a chemist's specifying the molecular geometry and, when feasible and necessary, the electronic structure of the target molecule or other solid. Molecular geometry refers to the spatial arrangement of ...
s similar to that of cholesterol
Cholesterol is any of a class of certain organic molecules called lipids. It is a sterol (or modified steroid), a type of lipid. Cholesterol is biosynthesized by all animal cells and is an essential structural component of animal cell mem ...
. It is a white, waxy powder with a characteristic odor, and is one of the components of the food additive E499
Stigmasterol-rich plant sterols is a food additive. It is a mixture derived from soybeans that consists of the plant sterols stigmasterol, β-sitosterol, campesterol, and brassicasterol, with stigmasterol representing >85% of the mixture. Its E ...
. Phytosterols are hydrophobic and soluble in alcohols.
Natural occurrences and food
β-sitosterol is widely distributed in theplant kingdom
Plants are predominantly photosynthetic eukaryotes of the kingdom Plantae. Historically, the plant kingdom encompassed all living things that were not animals, and included algae and fungi; however, all current definitions of Plantae exclude ...
. It is found in vegetable oil
Vegetable oils, or vegetable fats, are oils extracted from seeds or from other parts of fruits. Like animal fats, vegetable fats are ''mixtures'' of triglycerides. Soybean oil, grape seed oil, and cocoa butter are examples of seed oils, or f ...
, nuts, avocado
The avocado (''Persea americana'') is a medium-sized, evergreen tree in the laurel family ( Lauraceae). It is native to the Americas and was first domesticated by Mesoamerican tribes more than 5,000 years ago. Then as now it was prized for ...
s, and derived prepared foods such as salad dressing
A salad dressing is a sauce for salads. Used on virtually all '' leafy salads'', dressings may also be used in making salads of beans (such as three bean salad), noodle or pasta salads and antipasti, and forms of potato salad.
Salad dressing ...
s.
Human research
β-sitosterol is being studied for its potential to reducebenign prostatic hyperplasia
Benign prostatic hyperplasia (BPH), also called prostate enlargement, is a noncancerous increase in size of the prostate gland. Symptoms may include frequent urination, trouble starting to urinate, weak stream, inability to urinate, or loss o ...
(BPH) and blood cholesterol
Cholesterol is any of a class of certain organic molecules called lipids. It is a sterol (or modified steroid), a type of lipid. Cholesterol is biosynthesized by all animal cells and is an essential structural component of animal cell mem ...
levels.
Genetic disorder
While plant sterols are usually beneficial, there is a rare autosomal recessive genetic disorder phytosterolemia which causes over-absorption of phytosterols.Precursor of anabolic steroid boldenone
Being a steroid, β-sitosterol is a precursor of anabolic steroidboldenone
Boldenone (developmental code name RU-18761), is a naturally occurring anabolic–androgenic steroid (AAS) and the 1(2)- dehydrogenated analogue of testosterone. Boldenone itself has never been marketed; as a pharmaceutical drug, it is used as ...
. Boldenone undecylenate
Boldenone undecylenate, or boldenone undecenoate, sold under the brand names Equipoise and Parenabol among others, is an androgen and anabolic steroid (AAS) medication which is used in veterinary medicine, mainly in horses. It was formerly used i ...
is commonly used in veterinary medicine to induce growth in cattle but it is also one of the most commonly abused anabolic steroids in sports. This led to suspicion that some athletes testing positive on boldenone undecylenate didn't actually abuse the hormone itself but consumed food rich in β-sitosterol.
Chemistry
Chemical engineering
The use of sitosterol as a chemical intermediate was for many years limited due to the lack of a chemical point of attack on the side-chain that would permit its removal. Extensive efforts on the part of many laboratories eventually led to the discovery of apseudomonas
''Pseudomonas'' is a genus of Gram-negative, Gammaproteobacteria, belonging to the family Pseudomonadaceae and containing 191 described species. The members of the genus demonstrate a great deal of metabolic diversity and consequently are able t ...
microbe that efficiently effected that transformation. Fermentation digests the entire aliphatic side-chain at carbon 17 to afford a mixture of 17-keto products including dehydroepiandrosterone
Dehydroepiandrosterone (DHEA), also known as androstenolone, is an endogenous steroid hormone precursor. It is one of the most abundant circulating steroids in humans. DHEA is produced in the adrenal glands, the gonads, and the brain. It fun ...
.
Synthesis
Total synthesis of β-sitosterol has not been achieved. However, β-sitosterol has been synthesized from stigmasterol 1, which involves a specific hydrogenation of the side-chain of stigmasterol. The first step in the synthesis forms stigmasterol tosylate 2 from stigmasterol 1 (95% purity) using p-TsCl, DMAP, and pyridine (90% yield). The tosylate 2 then undergoes solvolysis as it is treated with pyridine and anhydrous MeOH to give a 5:1 ratio of i-stigmasterol methyl ether 3 (74% yield) to stigmasterol methyl ether 4, which is subsequently removed by chromatography. The hydrogenation step of a previously proposed synthesis involved the catalyst Pd/C and the solvent ethyl acetate. However, due to isomerisation during hydrolysis, other catalysts, such as PtO2, and solvents, such as ethanol, were tested. There was little change with the use of a different catalyst. Ethanol, however, prevented isomerisation and the formation of the unidentified impurity to give compound 5. The last step of the synthesis is deprotection of the β-ring double bond of 5 with p-TsOH, aqueous dioxane, and heat (80 °C) to yield β-sitosterol 6. The cumulative yield for the final two steps was 55%, and the total yield for the synthesis was 37%.Biosynthesis
 The regulation of the biosynthesis of both sterols and some specific lipids occurs during membrane biogenesis. Through 13C-labeling patterns, it has been determined that both the mevalonate and deoxyxylulose pathways are involved in the formation of β-sitosterol. The precise mechanism of β-sitosterol formation varies according to the organism, but is generally found to come from cycloartenol.Dewick, P. M. Medicinal Natural Products: A Biosynthetic Approach. 3 ed.; John Wiley & Sons Ltd.: United Kingdom cyclization, 2009; p 539.
The biosynthesis of cycloartenol begins as one molecule of isopentenyl diphosphate (IPP) and two molecules of dimethylallyl diphosphate (DMAPP) form farnesyl diphosphate (FPP). Two molecules of FPP are then joined tail-to-tail to yield
The regulation of the biosynthesis of both sterols and some specific lipids occurs during membrane biogenesis. Through 13C-labeling patterns, it has been determined that both the mevalonate and deoxyxylulose pathways are involved in the formation of β-sitosterol. The precise mechanism of β-sitosterol formation varies according to the organism, but is generally found to come from cycloartenol.Dewick, P. M. Medicinal Natural Products: A Biosynthetic Approach. 3 ed.; John Wiley & Sons Ltd.: United Kingdom cyclization, 2009; p 539.
The biosynthesis of cycloartenol begins as one molecule of isopentenyl diphosphate (IPP) and two molecules of dimethylallyl diphosphate (DMAPP) form farnesyl diphosphate (FPP). Two molecules of FPP are then joined tail-to-tail to yield squalene
Squalene is an organic compound. It is a triterpenoid with the formula C30H50. It is a colourless oil, although impure samples appear yellow. It was originally obtained from shark liver oil (hence its name, as ''Squalus'' is a genus of sharks). A ...
, a triterpene
Triterpenes are a class of chemical compounds composed of three terpene units with the molecular formula C30H48; they may also be thought of as consisting of six isoprene units. Animals, plants and fungi all produce triterpenes, including squa ...
. Squalene, through a cyclization reaction with 2,3-oxidosqualene 6 as an intermediate forms cycloartenol.
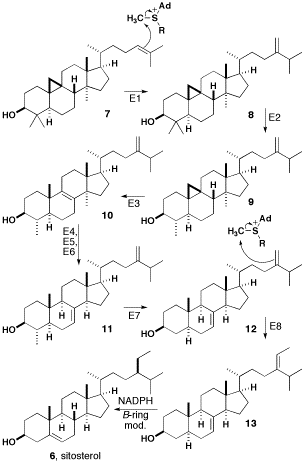
The double bond of cycloartenol (compound 7 in diagram) is methylated by SAM to give a carbocation that undergoes a hydride shift and loses a proton to yield a compound with a methylene side-chain. Both of these steps are catalyzed by sterol C-24 methyltransferase (Step E1 in diagram). Compound 8 is then catalyzed by sterol C-4 demethylase (E2) and loses a methyl group to produce cycloeucalenol. Subsequent to this, the cyclopropane ring is opened with cycloeucalenol cycloisomerase (E3) to form 10. Compound 10 loses a methyl group and undergoes an allylic isomerization to form gramisterol 11. This step is catalyzed by sterol C-14 demethylase (E4), sterol Δ14-reductase (E5), and sterol Δ8-Δ7-isomerase (E6). The last methyl group is removed by sterol demethylase (E7) to form episterol 12. Episterol 12 is methylated by SAM to produce a second carbocation, which loses a proton to yield 13. This step is catalyzed by 24-methylenesterol C-methyltransferase (E8). Compound 13 now undergoes reduction by NADPH and modifications in the β-ring to form β-sitosterol.
See also
*Charantin Charantin is a chemical substance obtained from the Asian bitter melon (''Momordica charantia''), reputed to be responsible for the hypoglycaemic properties of those plants. It was identified by Lolitkar and Rao in 1960. It was also found in the ...
, a β-sitosteryl glucoside found in the bitter melon
''Momordica charantia'' (commonly called bitter melon; Goya; bitter apple; bitter gourd; bitter squash; balsam-pear; with many more names listed below) is a tropical and subtropical vine of the family Cucurbitaceae, widely grown in Asia, Afr ...
plant.
References
{{DEFAULTSORT:Sitosterol, beta- 5α-Reductase inhibitors Phytoestrogens Phytosterols