|
Perilla Ketone
Perilla ketone is a natural terpenoid that consists of a furan ring with a six-carbon side chain containing a ketone functional group. It is a colorless oil that is sensitive to oxygen, becoming colored upon standing. The ketone was identified in 1943 by Sebe as the main component of the essential oil of Perilla frutescens. Perilla ketone is present in the leaves and seeds of purple mint (''Perilla frutescens''), which is toxic to some animals. When cattle and horses consume purple mint when grazing in fields in which it grows, the perilla ketone causes pulmonary edema leading to a condition sometimes called perilla mint toxicosis. Synthesis Perilla ketone was synthesized in 1957 by Matsuura from 3-furoyl chloride and an organocadmium compound similar to the Gilman reagent made from an isoamyl Grignard reagent and cadmium chloride. Perilla ketone has also been prepared in 74% yield via the Stille reaction from a 3-furyl-organotin compound and isocaproyl chloride in tetrahydr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Terpenoid
The terpenoids, also known as isoprenoids, are a class of naturally occurring organic compound, organic chemicals derived from the 5-carbon compound isoprene and its derivatives called terpenes, diterpenes, etc. While sometimes used interchangeably with "terpenes", terpenoids contain additional functional groups, usually containing oxygen. When combined with the hydrocarbon terpenes, terpenoids comprise about 80,000 compounds. They are the largest class of plant secondary metabolites, representing about 60% of known Natural Products, natural products. Many terpenoids have substantial pharmacological bioactivity and are therefore of interest to medicinal chemists. Plant terpenoids are used for their aromatic qualities and play a role in traditional herbal remedies. Terpenoids contribute to the scent of eucalyptus, the flavors of cinnamon, cloves, and ginger, the yellow color in sunflowers, and the red color in tomatoes. Well-known terpenoids include citral, menthol, camphor, salvin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Furan
Furan is a Heterocyclic compound, heterocyclic organic compound, consisting of a five-membered aromatic Ring (chemistry), ring with four carbon Atom, atoms and one oxygen atom. Chemical compounds containing such rings are also referred to as furans. Furan is a colorless, flammable, highly Volatility (chemistry), volatile liquid with a boiling point close to room temperature. It is soluble in common organic Solvent, solvents, including ethanol, alcohol, diethyl ether, ether, and acetone, and is slightly soluble in water. Its odor is "strong, ethereal; chloroform-like". It is toxic and may be carcinogenic in humans. Furan is used as a starting point for other speciality chemicals. History The name "furan" comes from the Latin ''furfur'', which means bran (furfural is produced from bran). The first furan derivative to be described was 2-furoic acid, by Carl Wilhelm Scheele in 1780. Another important derivative, furfural, was reported by Johann Wolfgang Döbereiner in 1831 an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Side Chain
In organic chemistry and biochemistry, a side chain is a substituent, chemical group that is attached to a core part of the molecule called the "main chain" or backbone chain, backbone. The side chain is a hydrocarbon branching element of a molecule that is attached to a larger hydrocarbon backbone. It is one factor in determining a molecule's properties and reactivity. A side chain is also known as a pendant chain, but a pendant group (side group) has a different definition. Conventions The placeholder R is often used as a generic placeholder for alkyl (saturated hydrocarbon) group side chains in structural formulae. To indicate other non-carbon groups in structure diagrams, X, Y, or Z are often used. History The ''R'' symbol was introduced by 19th-century French chemist Charles Frédéric Gerhardt, who advocated its adoption on the grounds that it would be widely recognizable and intelligible given its correspondence in multiple European languages to the initial letter of "r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Ketone
In organic chemistry, a ketone is an organic compound with the structure , where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' are methyl), with the formula . Many ketones are of great importance in biology and industry. Examples include many sugars (ketoses), many steroids, ''e.g.'', testosterone, and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considered retained IUPAC names, although some introdu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Perilla
''Perilla'' is a genus consisting of one major Asiatic crop species '' Perilla frutescens'' and a few wild species in nature belonging to the mint family, Lamiaceae. The genus encompasses several distinct varieties of Asian herb, seed, and vegetable crop, including '' P. frutescens'' (deulkkae) and ''P. frutescens'' var. ''crispa'' (shiso). The genus name ''Perilla'' is also a frequently employed common name ("perilla"), applicable to all varieties. Perilla varieties are cross-fertile and intra-specific hybridization occurs naturally. Some varieties are considered invasive. Taxa and synonyms The classification of ''Perilla'' is confused, partly because botanists struggled with distinguishing the two distinct cultigens as different species or variations. Until a few decades ago, ''P. frutescens'' var. ''crispa'' was regarded as a species in its own right, distinct from ''P. frutescens'', although it was well established that these types readily cross-pollinate. An early e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Pulmonary Edema
Pulmonary edema (British English: oedema), also known as pulmonary congestion, is excessive fluid accumulation in the tissue or air spaces (usually alveoli) of the lungs. This leads to impaired gas exchange, most often leading to shortness of breath ( dyspnea) which can progress to hypoxemia and respiratory failure. Pulmonary edema has multiple causes and is traditionally classified as cardiogenic (caused by the heart) or noncardiogenic (all other types not caused by the heart). Various laboratory tests ( CBC, troponin, BNP, etc.) and imaging studies (chest x-ray, CT scan, ultrasound) are often used to diagnose and classify the cause of pulmonary edema. Treatment is focused on three aspects: * improving respiratory function, * treating the underlying cause, and * preventing further damage and allow full recovery to the lung. Pulmonary edema can cause permanent organ damage, and when sudden (acute), can lead to respiratory failure or cardiac arrest due to hypoxia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Organocadmium Compound
Organocadmium chemistry describes the physical properties, synthesis, reactions, and use of organocadmium compounds, which are organometallic compounds containing a carbon to cadmium chemical bond. Cadmium shares group 12 with zinc and mercury and their corresponding chemistries have much in common. The synthetic utility of organocadmium compounds is limited. The simplest organocadmium compound is dimethylcadmium. It is a linear molecule with a C-Cd bond length of 213 pm. Organocadmium compounds are typically sensitive to air, light, and moisture. Synthesis Dimethylcadmium and diethylcadmium were reported in 1917 by Erich Krause. In general, they are prepared by transmetalation or by an exchange reaction between an alkylating agent and a cadmium salt. According to one procedure, diethylcadmium is produced by the reaction of cadmium bromide with two equivalents of the Grignard reagent ethylmagnesium bromide in diethyl ether. Diethylcadmium is a colorless oil with melting ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Gilman Reagent
A Gilman reagent is a diorganocopper compound with the formula Li uR2 where R is an alkyl or aryl. They are colorless solids. Use in organic chemistry These reagents are useful because, unlike related Grignard reagents and organolithium reagents, they react with organic halides to replace the halide group with an R group (the Corey–House reaction). Such displacement reactions allow for the synthesis of complex products from simple building blocks. Lewis acids can be used to modify the reagent. History These reagents were discovered by Henry Gilman and coworkers. Lithium dimethylcopper (CH3)2CuLi can be prepared by adding copper(I) iodide to methyllithium in tetrahydrofuran at −78 °C. In the reaction depicted below, the Gilman reagent is a methylating reagent reacting with an alkyne in a conjugate addition, and the ester group forms a cyclic enone. : Structure Lithium dimethylcuprate exists as a dimer in diethyl ether Diethyl ether, or simply ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Cadmium Chloride
Cadmium chloride is a white crystalline compound of cadmium and chloride, with the formula CdCl2. This salt is a hygroscopic solid that is highly soluble in water and slightly soluble in alcohol. The crystal structure of cadmium chloride (described below), is a reference for describing other crystal structures. Also known are CdCl2•H2O and the hemipentahydrate CdCl2•2.5H2O. Structure Anhydrous Anhydrous cadmium chloride forms a layered structure consisting of octahedral Cd2+ centers linked with chloride ligands. Cadmium iodide, CdI2, has a similar structure, but the iodide ions are arranged in a HCP lattice, whereas in CdCl2 the chloride ions are arranged in a CCP lattice.N. N. Greenwood, A. Earnshaw, ''Chemistry of the Elements'', 2nd ed., Butterworth-Heinemann, Oxford, UK, 1997. Hydrates The anhydrous form absorbs moisture from the air to form various hydrates. Three of these hydrates have been examined by X-ray crystallography. Chemical properties Cadmium chloride di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Bulletin Of The Chemical Society Of Japan
is a scientific journal, which was founded in 1926 by the Chemical Society of Japan. It publishes accounts, articles, and short articles in the fields of theoretical and physical chemistry, analytical and inorganic chemistry, organic and biological chemistry, and applied and materials chemistry. Due to World War II World War II or the Second World War (1 September 1939 – 2 September 1945) was a World war, global conflict between two coalitions: the Allies of World War II, Allies and the Axis powers. World War II by country, Nearly all of the wo ... publication was suspended between 1945 and 1946. It is published in both a print edition and an online edition. External links Homepage of the Chemical Society of Japan References Chemistry journals Academic journals established in 1926 Academic journals published by learned and professional societies Chemical Society of Japan {{chem-journal-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
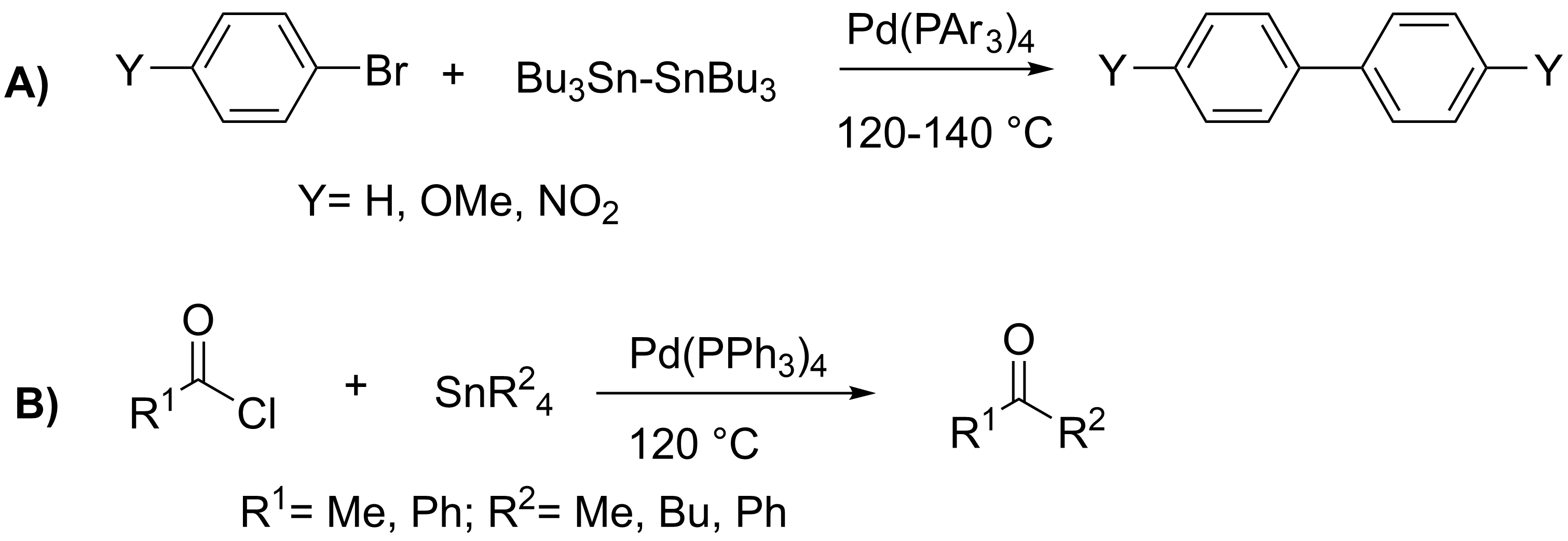
Stille Reaction
The Stille reaction is a chemical reaction widely used in organic synthesis. The reaction involves the coupling of two organic groups, one of which is carried as an organotin chemistry, organotin compound (also known as organostannanes). A variety of organic electrophiles provide the other coupling partner. The Stille reaction is one of many Cross-coupling reaction, palladium-catalyzed coupling reactions.Hartwig, J. F. ''Organotransition Metal Chemistry, from Bonding to Catalysis''; University Science Books: New York, 2010. Stille, J. K. ''Angew. Chem. Int. Ed. Engl.'' 1986, ''25'', 508–524.ReviewFarina, V.; Krishnamurthy, V.; Scott, W. J. ''Org. React.'' 1998, ''50'', 1–652.Review : + \ \ce \ \overbrace^ + \!-\! :*\!,\ : Allyl, alkenyl, aryl, benzyl, acyl :*: halides (Cl, Br, I), pseudohalides (OTf, OPO(OR)2), OAc The R1 group attached to the trialkyltin is normally sp2-hybridized, including vinyl group, vinyl, and aryl groups. These organostannanes are also stable to bo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Tetrahydrofuran
Tetrahydrofuran (THF), or oxolane, is an organic compound with the formula (CH2)4O. The compound is classified as heterocyclic compound, specifically a cyclic ether. It is a colorless, water- miscible organic liquid with low viscosity. It is mainly used as a precursor to polymers. Being polar and having a wide liquid range, THF is a versatile solvent. It is an isomer of another solvent, butanone. Production About 200,000 tonnes of tetrahydrofuran are produced annually. The most widely used industrial process involves the acid-catalyzed dehydration of 1,4-Butanediol, 1,4-butanediol. Ashland Inc., Ashland/ISP is one of the biggest producers of this chemical route. The method is similar to the production of diethyl ether from ethanol. The butanediol is derived from Condensation reaction, condensation of acetylene with formaldehyde followed by hydrogenation. DuPont developed a process for producing THF by oxidizing Butane#Isomers, ''n''-butane to crude maleic anhydride, follow ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |