|
Density
Density (volumetric mass density or specific mass) is the ratio of a substance's mass to its volume. The symbol most often used for density is ''ρ'' (the lower case Greek letter rho), although the Latin letter ''D'' (or ''d'') can also be used: \rho = \frac, where ''ρ'' is the density, ''m'' is the mass, and ''V'' is the volume. In some cases (for instance, in the United States oil and gas industry), density is loosely defined as its weight per unit volume, although this is scientifically inaccurate this quantity is more specifically called specific weight. For a pure substance, the density is equal to its mass concentration. Different materials usually have different densities, and density may be relevant to buoyancy, purity and packaging. Osmium is the densest known element at standard conditions for temperature and pressure. To simplify comparisons of density across different systems of units, it is sometimes replaced by the dimensionless quantity "relative den ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Relative Density
Relative density, also called specific gravity, is a dimensionless quantity defined as the ratio of the density (mass of a unit volume) of a substance to the density of a given reference material. Specific gravity for solids and liquids is nearly always measured with respect to water at its densest (at ); for gases, the reference is air at room temperature (). The term "relative density" (abbreviated r.d. or RD) is preferred in SI, whereas the term "specific gravity" is gradually being abandoned. If a substance's relative density is less than 1 then it is less dense than the reference; if greater than 1 then it is denser than the reference. If the relative density is exactly 1 then the densities are equal; that is, equal volumes of the two substances have the same mass. If the reference material is water, then a substance with a relative density (or specific gravity) less than 1 will float in water. For example, an ice cube, with a relative density of about 0.91, will float. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Gram Per Cubic Centimetre
The gram per cubic centimetre is a unit of density in International System of Units (SI), and is commonly used in chemistry. Its official SI symbols are g/cm3, g·cm−3, or g cm−3. It is equal to the units gram per millilitre (g/mL) and kilogram per litre (kg/L). It is defined by dividing the gram, a unit of mass, by the cubic centimetre, a unit of volume. It is a coherent unit in the CGS system, but is not a coherent unit of the SI. The density of water is approximately 1 g/cm3, since the gram was originally defined as the mass of one cubic centimetre of water at its maximum density The maximum density of a substance is the highest attainable density of the substance under given conditions. Attaining maximum density Almost all known substances undergo thermal expansion in response to heating, meaning that a given mass of su ... at approximately . Conversions * 1 g/cm3 is equal to: *: = 1000 g/L (exactly) *: = 1000 kg/m3 (exactly) *: ≈ (approximatel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Buoyancy
Buoyancy (), or upthrust, is the force exerted by a fluid opposing the weight of a partially or fully immersed object (which may be also be a parcel of fluid). In a column of fluid, pressure increases with depth as a result of the weight of the overlying fluid. Thus, the pressure at the bottom of a column of fluid is greater than at the top of the column. Similarly, the pressure at the bottom of an object submerged in a fluid is greater than at the top of the object. The pressure difference results in a net upward force on the object. The magnitude of the force is proportional to the pressure difference, and (as explained by Archimedes' principle) is equivalent to the weight of the fluid that would otherwise occupy the submerged volume of the object, i.e. the Displacement (fluid), displaced fluid. For this reason, an object with average density greater than the surrounding fluid tends to sink because its weight is greater than the weight of the fluid it displaces. If the objec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Kilogram Per Cubic Metre
The kilogram per cubic metre (symbol: kg·m−3, or kg/m3) is the unit of density in the International System of Units (SI). It is defined by dividing the SI unit of mass, the kilogram, by the SI unit of volume, the cubic metre. Conversions *1 kg/m3 = 1 g/ L (exactly) *1 kg/m3 = 0.001 g/cm3 (exactly) *1 kg/m3 ≈ 0.06243 lb/ ft3 (approximately) *1 kg/m3 ≈ 0.1335 oz/ US gal (approximately) *1 kg/m3 ≈ 0.1604 oz/ imp gal (approximately) *1 g/cm3 = 1000 kg/m3 (exactly) *1 lb/ft3 ≈ 16.02 kg/m3 (approximately) *1 oz/(US gal) ≈ 7.489 kg/m3 (approximately) *1 oz/(imp gal) ≈ 6.236 kg/m3 (approximately) Relation to other measures The density of water is about 1000 kg/m3 or 1 g/cm3, because the size of the gram was originally based on the mass of a cubic centimetre of water. In chemistry, g/cm3 is more commonly used. See also * Gram per cubic centimetre The gram per cubic centimetre is a unit of density in International System ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
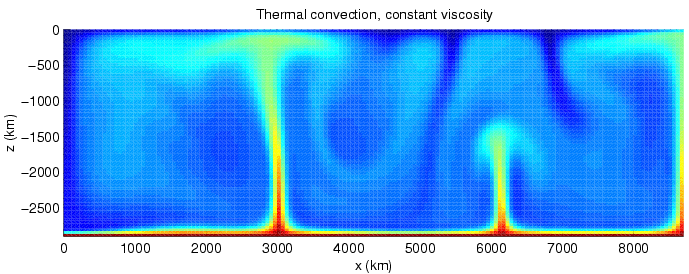
Convection
Convection is single or Multiphase flow, multiphase fluid flow that occurs Spontaneous process, spontaneously through the combined effects of material property heterogeneity and body forces on a fluid, most commonly density and gravity (see buoyancy). When the cause of the convection is unspecified, convection due to the effects of thermal expansion and buoyancy can be assumed. Convection may also take place in soft solids or mixtures where particles can flow. Convective flow may be Transient state, transient (such as when a Multiphasic liquid, multiphase mixture of oil and water separates) or steady state (see convection cell). The convection may be due to Gravity, gravitational, Electromagnetism, electromagnetic or Fictitious force, fictitious body forces. Convection (heat transfer), Heat transfer by natural convection plays a role in the structure of Earth's atmosphere, its oceans, and its Earth's mantle, mantle. Discrete convective cells in the atmosphere can be identified by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Volume
Volume is a measure of regions in three-dimensional space. It is often quantified numerically using SI derived units (such as the cubic metre and litre) or by various imperial or US customary units (such as the gallon, quart, cubic inch). The definition of length and height (cubed) is interrelated with volume. The volume of a container is generally understood to be the capacity of the container; i.e., the amount of fluid (gas or liquid) that the container could hold, rather than the amount of space the container itself displaces. By metonymy, the term "volume" sometimes is used to refer to the corresponding region (e.g., bounding volume). In ancient times, volume was measured using similar-shaped natural containers. Later on, standardized containers were used. Some simple three-dimensional shapes can have their volume easily calculated using arithmetic formulas. Volumes of more complicated shapes can be calculated with integral calculus if a formula exists for the shape ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Standard Conditions For Temperature And Pressure
Standard temperature and pressure (STP) or standard conditions for temperature and pressure are various standard sets of conditions for experimental measurements used to allow comparisons to be made between different sets of data. The most used standards are those of the International Union of Pure and Applied Chemistry (IUPAC) and the National Institute of Standards and Technology (NIST), although these are not universally accepted. Other organizations have established a variety of other definitions. In industry and commerce, the standard conditions for temperature and pressure are often necessary for expressing the volumes of gases and liquids and related quantities such as the rate of volumetric flow (the volumes of gases vary significantly with temperature and pressure): standard cubic meters per second (Sm3/s), and normal cubic meters per second (Nm3/s). Many technical publications (books, journals, advertisements for equipment and machinery) simply state "standard cond ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Specific Volume
In thermodynamics, the specific volume of a substance (symbol: , nu) is the quotient of the substance's volume () to its mass (): :\nu = \frac It is a mass-specific intrinsic property of the substance. It is the reciprocal of density (rho) and it is also related to the molar volume and molar mass: :\nu = \rho^ = \frac The standard unit of specific volume is cubic meters per kilogram (m3/kg), but other units include ft3/lb, ft3/slug, or mL/g. Specific volume for an ideal gas is related to the molar gas constant () and the gas's temperature (), pressure (), and molar mass (): : \nu = \frac It's based on the ideal gas law, PV = , and the amount of substance In chemistry, the amount of substance (symbol ) in a given sample of matter is defined as a ratio () between the particle number, number of elementary entities () and the Avogadro constant (). The unit of amount of substance in the International ..., n = m/M Applications Specific volume is commonly applied to: * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Mass Concentration (chemistry)
In chemistry, the mass concentration (or ) is defined as the mass of a constituent divided by the volume of the mixture . :\rho_i = \frac For a pure chemical the mass concentration equals its density (mass divided by volume); thus the mass concentration of a component in a mixture can be called the density of a component in a mixture. This explains the usage of (the lower case Greek letter rho), the symbol most often used for density. Definition and properties The volume in the definition refers to the volume of the solution, ''not'' the volume of the solvent. One litre of a solution usually contains either slightly more or slightly less than 1 litre of solvent because the process of dissolution causes volume of liquid to increase or decrease. Sometimes the mass concentration is called titre. Notation The notation common with mass density underlines the connection between the two quantities (the mass concentration being the mass density of a component in the solution), ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Intensive Property
Physical or chemical properties of materials and systems can often be categorized as being either intensive or extensive, according to how the property changes when the size (or extent) of the system changes. The terms "intensive and extensive quantities" were introduced into physics by German mathematician Georg Helm in 1898, and by American physicist and chemist Richard C. Tolman in 1917. According to International Union of Pure and Applied Chemistry (IUPAC), an intensive property or intensive quantity is one whose magnitude is independent of the size of the system. An intensive property is not necessarily homogeneously distributed in space; it can vary from place to place in a body of matter and radiation. Examples of intensive properties include temperature, ''T''; refractive index, ''n''; density, ''ρ''; and hardness, ''η''. By contrast, an extensive property or extensive quantity is one whose magnitude is additive for subsystems. Examples include mass, volume and Gi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |