Electrophilic on:
[Wikipedia]
[Google]
[Amazon]
In
 In contrast, phenylpropyne reacts by the AdE2ip ("addition, electrophilic, second-order, ion pair") mechanism to give predominantly the ''syn'' product (~10:1 ''syn'':''anti''). In this case, the intermediate vinyl cation is formed by addition of HCl because it is resonance-stabilized by the phenyl group. Nevertheless, the lifetime of this high energy species is short, and the resulting vinyl cation-chloride anion ion pair immediately collapses, before the chloride ion has a chance to leave the solvent shell, to give the vinyl chloride. The proximity of the anion to the side of the vinyl cation where the proton was added is used to rationalize the observed predominance of ''syn'' addition.
In contrast, phenylpropyne reacts by the AdE2ip ("addition, electrophilic, second-order, ion pair") mechanism to give predominantly the ''syn'' product (~10:1 ''syn'':''anti''). In this case, the intermediate vinyl cation is formed by addition of HCl because it is resonance-stabilized by the phenyl group. Nevertheless, the lifetime of this high energy species is short, and the resulting vinyl cation-chloride anion ion pair immediately collapses, before the chloride ion has a chance to leave the solvent shell, to give the vinyl chloride. The proximity of the anion to the side of the vinyl cation where the proton was added is used to rationalize the observed predominance of ''syn'' addition.
 Oxaziridines such as chiral N-sulfonyloxaziridines effect enantioselective ketone alpha oxidation en route to the AB-ring segments of various natural products, including γ-rhodomycionone and α-citromycinone.
Polymer-bound chiral selenium electrophiles effect asymmetric selenenylation reactions. The reagents are aryl selenenyl bromides, and they were first developed for solution phase chemistry and then modified for solid phase bead attachment via an aryloxy moiety. The solid-phase reagents were applied toward the selenenylation of various alkenes with good enantioselectivities. The products can be cleaved from the solid support using organotin hydride reducing agents. Solid-supported reagents offers advantages over solution phase chemistry due to the ease of workup and purification.
Oxaziridines such as chiral N-sulfonyloxaziridines effect enantioselective ketone alpha oxidation en route to the AB-ring segments of various natural products, including γ-rhodomycionone and α-citromycinone.
Polymer-bound chiral selenium electrophiles effect asymmetric selenenylation reactions. The reagents are aryl selenenyl bromides, and they were first developed for solution phase chemistry and then modified for solid phase bead attachment via an aryloxy moiety. The solid-phase reagents were applied toward the selenenylation of various alkenes with good enantioselectivities. The products can be cleaved from the solid support using organotin hydride reducing agents. Solid-supported reagents offers advantages over solution phase chemistry due to the ease of workup and purification.
chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a natural science that covers the elements that make up matter to the compounds made of atoms, molecules and ions: their composition, structure, proper ...
, an electrophile is a chemical species
A chemical species is a chemical substance or ensemble composed of chemically identical molecular entities that can explore the same set of molecular energy levels on a characteristic or delineated time scale. These energy levels determine the wa ...
that forms bonds with nucleophiles by accepting an electron pair
In chemistry, an electron pair or Lewis pair consists of two electrons that occupy the same molecular orbital but have opposite spins. Gilbert N. Lewis introduced the concepts of both the electron pair and the covalent bond in a landmark paper he ...
. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons.
Electrophiles mainly interact with nucleophiles through addition and substitution
Substitution may refer to:
Arts and media
*Chord substitution, in music, swapping one chord for a related one within a chord progression
*Substitution (poetry), a variation in poetic scansion
* "Substitution" (song), a 2009 song by Silversun Pic ...
reactions. Frequently seen electrophiles in organic syntheses
''Organic Syntheses'' is a peer-reviewed scientific journal that was established in 1921. It publishes detailed and checked procedures for the synthesis of organic compounds. A unique feature of the review process is that all of the data and exp ...
include cations such as H+ and NO+, polarized neutral molecules such as HCl HCL may refer to:
Science and medicine
* Hairy cell leukemia, an uncommon and slowly progressing B cell leukemia
* Harvard Cyclotron Laboratory, from 1961 to 2002, a proton accelerator used for research and development
* Hollow-cathode lamp, a s ...
, alkyl halides, acyl halide
In organic chemistry, an acyl halide (also known as an acid halide) is a chemical compound derived from an oxoacid by replacing a hydroxyl group () with a halide group (, where X is a halogen).
If the acid is a carboxylic acid (), the compoun ...
s, and carbonyl compounds, polarizable neutral molecules such as Cl2 and Br2, oxidizing agent
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
s such as organic peracids, chemical species that do not satisfy the octet rule such as carbene
In organic chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is or where the R represents substituents or hydrogen atoms.
The term "carbene" m ...
s and radicals
Radical may refer to:
Politics and ideology Politics
*Radical politics, the political intent of fundamental societal change
*Radicalism (historical), the Radical Movement that began in late 18th century Britain and spread to continental Europe and ...
, and some Lewis acids such as BH3 and DIBAL.
Organic chemistry
Addition of halogens
These occur between alkenes and electrophiles, often halogens as in halogen addition reactions. Common reactions include use of bromine water totitrate
Titration (also known as titrimetry and volumetric analysis) is a common laboratory method of quantitative chemical analysis to determine the concentration of an identified analyte (a substance to be analyzed). A reagent, termed the ''titrant' ...
against a sample to deduce the number of double bonds present. For example, ethene + bromine
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest element in group 17 of the periodic table ( halogens) and is a volatile red-brown liquid at room temperature that evaporates readily to form a simi ...
→ 1,2-dibromoethane:
:C2H4 + Br2 → BrCH2CH2Br
This takes the form of 3 main steps shown below;
:
#Forming of a π-complex
#:The electrophilic Br-Br molecule interacts with electron-rich alkene molecule to form a π-complex 1.
#Forming of a three-membered bromonium ion
#:The alkene is working as an electron donor and bromine as an electrophile. The three-membered bromonium ion 2 consisted of two carbon atoms and a bromine atom forms with a release of Br−.
#Attacking of bromide ion
#:The bromonium ion is opened by the attack of Br− from the back side. This yields the vicinal dibromide with an antiperiplanar configuration. When other nucleophiles such as water or alcohol are existing, these may attack 2 to give an alcohol or an ether.
This process is called AdE2 mechanism ("addition, electrophilic, second-order"). Iodine (I2), chlorine
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine i ...
(Cl2), sulfenyl
In chemistry, a sulfenic acid is an organosulfur compound and oxoacid with the general formula . It is the first member of the family of organosulfur oxoacids, which also include sulfinic acids () and sulfonic acids (), respectively. The base me ...
ion (RS+), mercury cation (Hg2+), and dichlorocarbene (:CCl2) also react through similar pathways. The direct conversion of 1 to 3 will appear when the Br− is large excess in the reaction medium. A β-bromo carbenium ion
A carbenium ion is a positive ion with the structure RR′R″C+, that is, a chemical species with a trivalent carbon that bears a +1 formal charge.
In older literature the name carbonium ion was used for this class, but now it refers exclusivel ...
intermediate may be predominant instead of 3 if the alkene has a cation-stabilizing substituent like phenyl group. There is an example of the isolation of the bromonium ion 2.
Addition of hydrogen halides
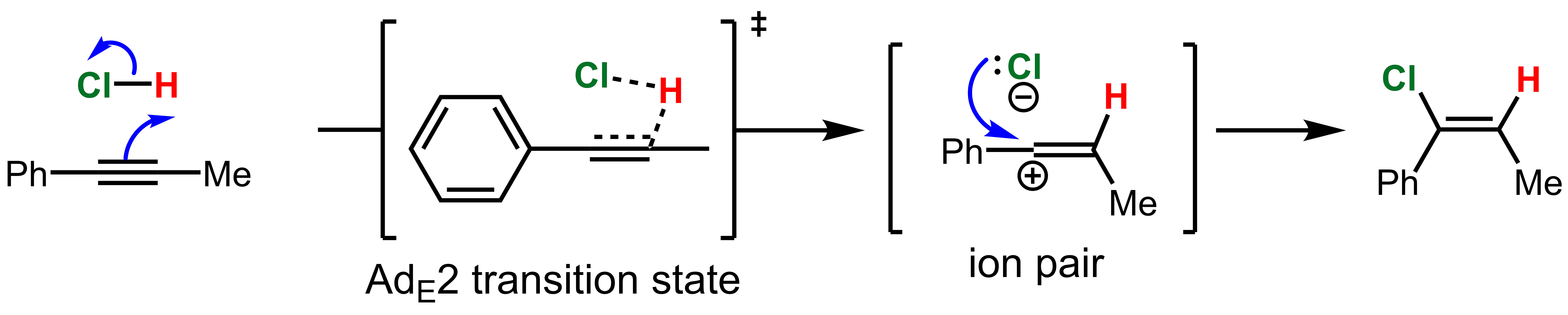
Hydrogen halides such as hydrogen chloride (HCl) adds to alkenes to give alkyl halides in hydrohalogenation. For example, the reaction of HCl with ethylene furnishes chloroethane. The reaction proceeds with a cation intermediate, being different from the above halogen addition. An example is shown below: : #Proton (H+) adds (by working as an electrophile) to one of the carbon atoms on the alkene to form cation 1. #Chloride ion (Cl−) combines with the cation 1 to form the adducts 2 and 3. In this manner, the stereoselectivity of the product, that is, from which side Cl− will attack relies on the types of alkenes applied and conditions of the reaction. At least, which of the two carbon atoms will be attacked by H+ is usually decided by Markovnikov's rule. Thus, H+ attacks the carbon atom that carries fewer substituents so as the more stabilized carbocation (with the more stabilizing substituents) will form. This is another example of an AdE2 mechanism. Hydrogen fluoride (HF) and hydrogen iodide (HI) react with alkenes in a similar manner, and Markovnikov-type products will be given. Hydrogen bromide (HBr) also takes this pathway, but sometimes a radical process competes and a mixture of isomers may form. Although introductory textbooks seldom mentions this alternative, the AdE2 mechanism is generally competitive with the AdE3 mechanism (described in more detail for alkynes, below), in which transfer of the proton and nucleophilic addition occur in a concerted manner. The extent to which each pathway contributes depends on the several factors like the nature of the solvent (e.g., polarity), nucleophilicity of the halide ion, stability of the carbocation, and steric effects. As brief examples, the formation of a sterically unencumbered, stabilized carbocation favors the AdE2 pathway, while a more nucleophilic bromide ion favors the AdE3 pathway to a greater extent compared to reactions involving the chloride ion. In the case of dialkyl-substituted alkynes (e.g., 3-hexyne), the intermediate vinyl cation that would result from this process is highly unstable. In such cases, the simultaneous protonation (by HCl) and attack of the alkyne by the nucleophile (Cl−) is believed to take place. This mechanistic pathway is known by the Ingold label AdE3 ("addition, electrophilic, third-order"). Because the simultaneous collision of three chemical species in a reactive orientation is improbable, the termolecular transition state is believed to be reached when the nucleophile attacks a reversibly-formed weak association of the alkyne and HCl. Such a mechanism is consistent with the predominantly ''anti'' addition (>15:1 ''anti'':''syn'' for the example shown) of the hydrochlorination product and the termolecular rate law, Rate = ''k'' lkyneHCl]2. In support of the proposed alkyne-HCl association, a T-shaped complex of an alkyne and HCl has been characterized crystallographically. In contrast, phenylpropyne reacts by the AdE2ip ("addition, electrophilic, second-order, ion pair") mechanism to give predominantly the ''syn'' product (~10:1 ''syn'':''anti''). In this case, the intermediate vinyl cation is formed by addition of HCl because it is resonance-stabilized by the phenyl group. Nevertheless, the lifetime of this high energy species is short, and the resulting vinyl cation-chloride anion ion pair immediately collapses, before the chloride ion has a chance to leave the solvent shell, to give the vinyl chloride. The proximity of the anion to the side of the vinyl cation where the proton was added is used to rationalize the observed predominance of ''syn'' addition.
In contrast, phenylpropyne reacts by the AdE2ip ("addition, electrophilic, second-order, ion pair") mechanism to give predominantly the ''syn'' product (~10:1 ''syn'':''anti''). In this case, the intermediate vinyl cation is formed by addition of HCl because it is resonance-stabilized by the phenyl group. Nevertheless, the lifetime of this high energy species is short, and the resulting vinyl cation-chloride anion ion pair immediately collapses, before the chloride ion has a chance to leave the solvent shell, to give the vinyl chloride. The proximity of the anion to the side of the vinyl cation where the proton was added is used to rationalize the observed predominance of ''syn'' addition.

Hydration
One of the more complex hydration reactions utilisessulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid ( Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen and hydrogen, with the molecular fo ...
as a catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
. This reaction occurs in a similar way to the addition reaction but has an extra step in which the OSO3H group is replaced by an OH group, forming an alcohol:
:C2H4 + H2O → C2H5OH
As can be seen, the H2SO4 does take part in the overall reaction, however it remains unchanged so is classified as a catalyst.
This is the reaction in more detail:
:
#The H–OSO3H molecule has a δ+ charge on the initial H atom. This is attracted to and reacts with the double bond in the same way as before.
#The remaining (negatively charged) −OSO3H ion then attaches to the carbocation, forming ethyl hydrogensulphate (upper way on the above scheme).
#When water (H2O) is added and the mixture heated, ethanol (C2H5OH) is produced. The "spare" hydrogen atom from the water goes into "replacing" the "lost" hydrogen and, thus, reproduces sulfuric acid. Another pathway in which water molecule combines directly to the intermediate carbocation (lower way) is also possible. This pathway become predominant when aqueous sulfuric acid is used.
Overall, this process adds a molecule of water to a molecule of ethene.
This is an important reaction in industry, as it produces ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
, whose purposes include fuels and starting material for other chemicals.
Chiral derivatives
Many electrophiles are chiral and optically stable. Typically chiral electrophiles are also optically pure. One such reagent is the fructose-derived organocatalyst used in the Shi epoxidation. The catalyst can accomplish highly enantioselective epoxidations of ''trans''-disubstituted and trisubstitutedalkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic ...
s. The Shi catalyst, a ketone, is oxidized by stoichiometric oxone
Potassium peroxymonosulfate is widely used as an oxidizing agent. It is the potassium salt of peroxymonosulfuric acid. Usually potassium peroxymonosulfate refers to the triple salt known as oxone.
The standard electrode potential for potassium ...
to the active dioxirane form before proceeding in the catalytic cycle.
 Oxaziridines such as chiral N-sulfonyloxaziridines effect enantioselective ketone alpha oxidation en route to the AB-ring segments of various natural products, including γ-rhodomycionone and α-citromycinone.
Polymer-bound chiral selenium electrophiles effect asymmetric selenenylation reactions. The reagents are aryl selenenyl bromides, and they were first developed for solution phase chemistry and then modified for solid phase bead attachment via an aryloxy moiety. The solid-phase reagents were applied toward the selenenylation of various alkenes with good enantioselectivities. The products can be cleaved from the solid support using organotin hydride reducing agents. Solid-supported reagents offers advantages over solution phase chemistry due to the ease of workup and purification.
Oxaziridines such as chiral N-sulfonyloxaziridines effect enantioselective ketone alpha oxidation en route to the AB-ring segments of various natural products, including γ-rhodomycionone and α-citromycinone.
Polymer-bound chiral selenium electrophiles effect asymmetric selenenylation reactions. The reagents are aryl selenenyl bromides, and they were first developed for solution phase chemistry and then modified for solid phase bead attachment via an aryloxy moiety. The solid-phase reagents were applied toward the selenenylation of various alkenes with good enantioselectivities. The products can be cleaved from the solid support using organotin hydride reducing agents. Solid-supported reagents offers advantages over solution phase chemistry due to the ease of workup and purification.
Electrophilicity scale
Several methods exist to rank electrophiles in order of reactivity and one of them is devised byRobert Parr
Robert Ghormley Parr (September 22, 1921 – March 27, 2017) was an American theoretical chemist who was a Professor of Chemistry at the University of North Carolina at Chapel Hill.
Career
Parr received an A. B. degree ''magna cum laude'' ...
with the electrophilicity index ω given as:
:
with the electronegativity and chemical hardness. This equation is related to the classical equation for electrical power
Electric power is the rate at which electrical energy is transferred by an electric circuit. The SI unit of power is the watt, one joule per second. Standard prefixes apply to watts as with other SI units: thousands, millions and billion ...
:
:
where is the resistance (Ohm
Ohm (symbol Ω) is a unit of electrical resistance named after Georg Ohm.
Ohm or OHM may also refer to:
People
* Georg Ohm (1789–1854), German physicist and namesake of the term ''ohm''
* Germán Ohm (born 1936), Mexican boxer
* Jörg Ohm (bor ...
or Ω) and is voltage
Voltage, also known as electric pressure, electric tension, or (electric) potential difference, is the difference in electric potential between two points. In a static electric field, it corresponds to the work needed per unit of charge to ...
. In this sense the electrophilicity index is a kind of electrophilic power. Correlations have been found between electrophilicity of various chemical compounds and reaction rates in biochemical systems and such phenomena as allergic contact dermititis.
An electrophilicity index also exists for free radicals. Strongly electrophilic radicals such as the halogens react with electron-rich reaction sites, and strongly nucleophilic radicals such as the 2-hydroxypropyl-2-yl and tert-butyl radical react with a preference for electron-poor reaction sites.
Superelectrophiles
Superelectrophiles are defined as cationic electrophilic reagents with greatly enhanced reactivities in the presence of superacids. These compounds were first described by George A. Olah. Superelectrophiles form as a doubly electron deficient superelectrophile by protosolvation of a cationic electrophile. As observed by Olah, a mixture of acetic acid and boron trifluoride is able to remove a hydride ion fromisobutane
Isobutane, also known as ''i''-butane, 2-methylpropane or methylpropane, is a chemical compound with molecular formula HC(CH3)3. It is an isomer of butane. Isobutane is a colourless, odourless gas.
It is the simplest alkane with a tertiary carbo ...
when combined with hydrofluoric acid via the formation of a superacid from BF3 and HF. The responsible reactive intermediate is the H3CO2H3sup>2+ dication. Likewise, methane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane ...
can be nitrated to nitromethane with nitronium tetrafluoroborate NOBF only in presence of a strong acid like fluorosulfuric acid via the protonated nitronium dication.
In gitionic (gitonic) superelectrophiles, charged centers are separated by no more than one atom, for example, the protonitronium ion O=N+=O+—H (a protonated nitronium ion). And, in distonic superelectrophiles, they are separated by 2 or more atoms, for example, in the fluorination reagent F-TEDA-BF4.
See also
*Nucleophile
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they ar ...
* TRPA1, the sensory neural target for electrophilic irritants in mammals.
References
{{reflist, 30em Physical organic chemistry