electrocyclic reaction on:
[Wikipedia]
[Google]
[Amazon]
In
 This reaction course can be explained in a simple analysis through the frontier-orbital method: the sigma bond in the reactant will open in such a way that the resulting
This reaction course can be explained in a simple analysis through the frontier-orbital method: the sigma bond in the reactant will open in such a way that the resulting 
 Correlation diagrams, which connect the molecular orbitals of the reactant to those of the product having the same symmetry, can then be constructed for the two processes.
Correlation diagrams, which connect the molecular orbitals of the reactant to those of the product having the same symmetry, can then be constructed for the two processes.
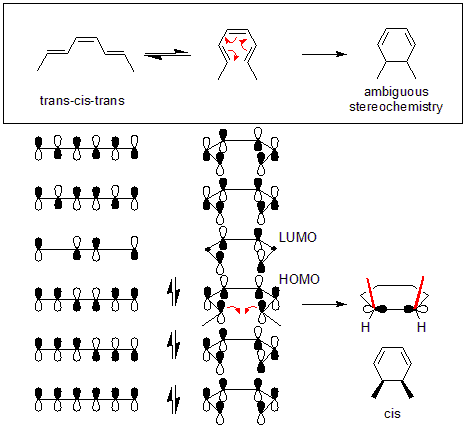
 These correlation diagrams indicate that only a conrotatory ring opening of 3,4-dimethylcyclobutene is symmetry allowed whereas only a disrotatory ring opening of 5,6-dimethylcyclohexa-1,3-diene is symmetry allowed. This is because only in these cases would maximum orbital overlap occur in the transition state. Also, the formed product would be in a ground state rather than an excited state.
These correlation diagrams indicate that only a conrotatory ring opening of 3,4-dimethylcyclobutene is symmetry allowed whereas only a disrotatory ring opening of 5,6-dimethylcyclohexa-1,3-diene is symmetry allowed. This is because only in these cases would maximum orbital overlap occur in the transition state. Also, the formed product would be in a ground state rather than an excited state.
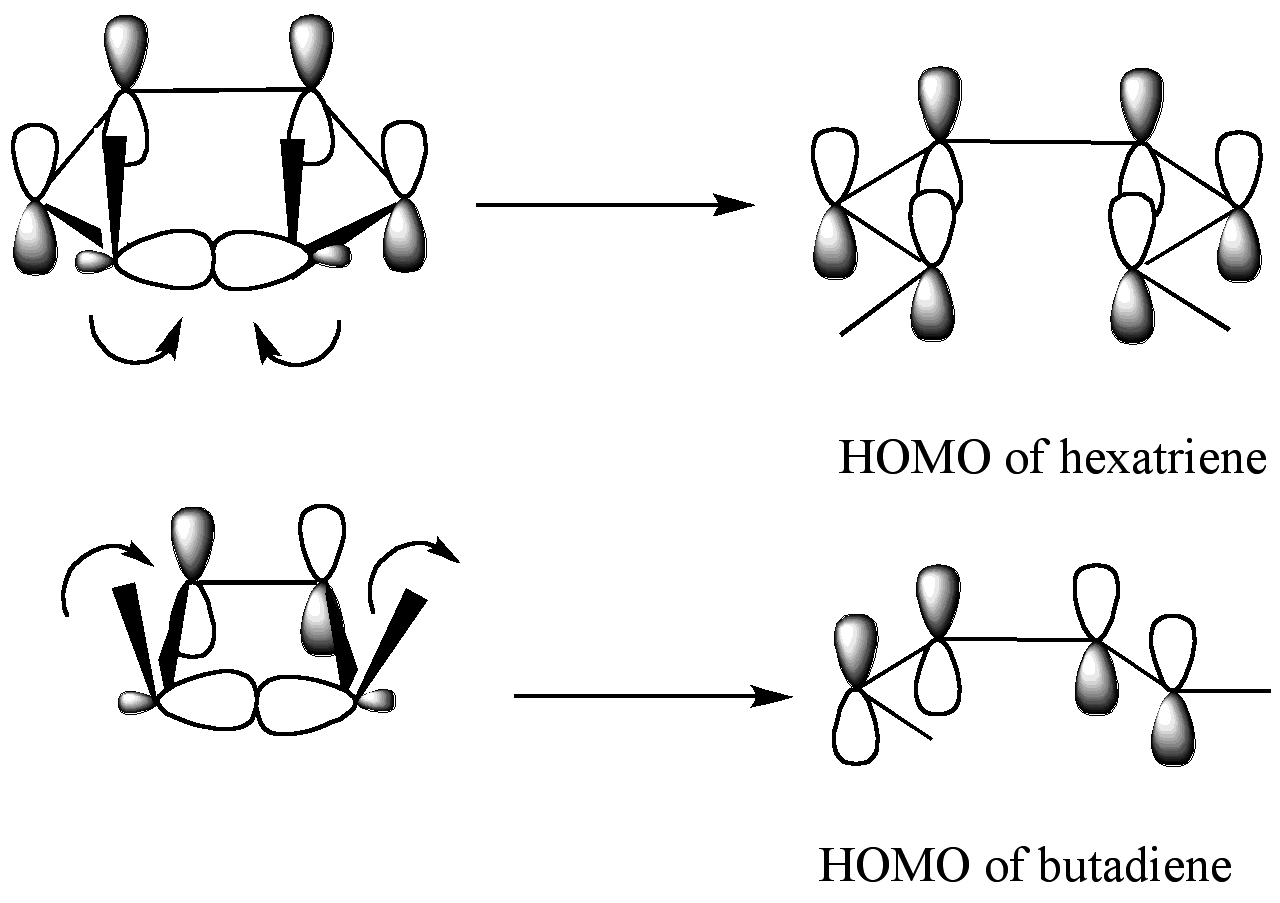 For the 5,6-dimethylcyclohexa-1,3-diene, only a disrotatory mode would result in p-orbitals having the same symmetry as the HOMO of hexatriene. For the 3,4-dimethylcyclobutene, on the other hand, only a conrotatory mode would result in p-orbitals having the same symmetry as the HOMO of butadiene.
For the 5,6-dimethylcyclohexa-1,3-diene, only a disrotatory mode would result in p-orbitals having the same symmetry as the HOMO of hexatriene. For the 3,4-dimethylcyclobutene, on the other hand, only a conrotatory mode would result in p-orbitals having the same symmetry as the HOMO of butadiene.
 Only a disrotatory mode, in which symmetry about a reflection plane is maintained throughout the reaction, would result in maximum orbital overlap in the transition state. Also, once again, this would result in the formation of a product that is in an excited state of comparable stability to the excited state of the reactant compound.
Only a disrotatory mode, in which symmetry about a reflection plane is maintained throughout the reaction, would result in maximum orbital overlap in the transition state. Also, once again, this would result in the formation of a product that is in an excited state of comparable stability to the excited state of the reactant compound.
 The first step involves a photochemically induced conrotatory ring opening of 7-dehydrocholesterol to form pre vitamin D3. A ,7hydride shift then forms vitamin D3.
Another example is in the proposed biosynthesis of aranotin, a naturally occurring oxepine, and its related compounds.
The first step involves a photochemically induced conrotatory ring opening of 7-dehydrocholesterol to form pre vitamin D3. A ,7hydride shift then forms vitamin D3.
Another example is in the proposed biosynthesis of aranotin, a naturally occurring oxepine, and its related compounds.
 Enzymatic epoxidation of phenylalanine-derived diketopiperazine forms the arene oxide, which undergoes a 6π disrotatory ring opening electrocyclization reaction to produce the uncyclized oxepine. After a second epoxidation of the ring, the nearby nucleophilic nitrogen attacks the electrophilic carbon, forming a five membered ring. The resulting ring system is a common ring system found in aranotin and its related compounds.
The benzonorcaradiene diterpenoid (A) was rearranged into the benzocycloheptatriene diterpenoid isosalvipuberlin (B) by boiling a methylene chloride solution. This transformation can be envisaged as a disrotatory electrocyclic reaction, followed by two suprafacial 1,5-sigmatropic hydrogen shifts, as shown below.
Enzymatic epoxidation of phenylalanine-derived diketopiperazine forms the arene oxide, which undergoes a 6π disrotatory ring opening electrocyclization reaction to produce the uncyclized oxepine. After a second epoxidation of the ring, the nearby nucleophilic nitrogen attacks the electrophilic carbon, forming a five membered ring. The resulting ring system is a common ring system found in aranotin and its related compounds.
The benzonorcaradiene diterpenoid (A) was rearranged into the benzocycloheptatriene diterpenoid isosalvipuberlin (B) by boiling a methylene chloride solution. This transformation can be envisaged as a disrotatory electrocyclic reaction, followed by two suprafacial 1,5-sigmatropic hydrogen shifts, as shown below.

 A
A  Asymmetric electrocyclic reactions are an emerging field in contemporary organic synthesis. The most commonly studied reactions in this field are the 4π Staudinger β-lactam synthesis and the 4π Nazarov reaction; asymmetric catalysis of both reactions have been controlled by use of a
Asymmetric electrocyclic reactions are an emerging field in contemporary organic synthesis. The most commonly studied reactions in this field are the 4π Staudinger β-lactam synthesis and the 4π Nazarov reaction; asymmetric catalysis of both reactions have been controlled by use of a
organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; ...
, an electrocyclic reaction is a type of pericyclic
In organic chemistry, a pericyclic reaction is the type of organic reaction wherein the transition state of the molecule has a cyclic geometry, the reaction progresses in a concerted fashion, and the bond orbitals involved in the reaction overlap ...
rearrangement where the net result is one pi bond
In chemistry, pi bonds (π bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. Each of these atomic orbitals ...
being converted into one sigma bond
In chemistry, sigma bonds (σ bonds) are the strongest type of covalent chemical bond. They are formed by head-on overlapping between atomic orbitals. Sigma bonding is most simply defined for diatomic molecules using the language and tools of s ...
or vice versa. These reactions are usually categorized by the following criteria:
* Reactions can be either photochemical
Photochemistry is the branch of chemistry concerned with the chemical effects of light. Generally, this term is used to describe a chemical reaction caused by absorption of ultraviolet (wavelength from 100 to 400 nm), visible light (400–7 ...
or thermal.
* Reactions can be either ring-opening or ring-closing (electrocyclization).
* Depending on the type of reaction (photochemical or thermal) and the number of pi electrons, the reaction can happen through either a conrotatory or disrotatory mechanism.
* The type of rotation determines whether the cis or trans isomer of the product will be formed.
Classical examples
TheNazarov cyclization reaction
The Nazarov cyclization reaction (often referred to as simply the Nazarov cyclization) is a chemical reaction used in organic chemistry for the synthesis of cyclopentenones. The reaction is typically divided into ''classical'' and ''modern'' var ...
is a named electrocyclic reaction converting divinylketones to cyclopentenones.
A classic example is the thermal ring-opening reaction
A cyclic compound (or ring compound) is a term for a compound in the field of chemistry in which one or more series of atoms in the compound is connected to form a ring. Rings may vary in size from three to many atoms, and include examples where a ...
of 3,4-dimethylcyclobutene. The cis isomer
Cis or cis- may refer to:
Places
* Cis, Trentino, in Italy
* In Poland:
** Cis, Świętokrzyskie Voivodeship, south-central
** Cis, Warmian-Masurian Voivodeship, north
Math, science and biology
* cis (mathematics) (cis(''θ'')), a trigonom ...
exclusively yields whereas the trans isomer gives the trans,trans diene:
p-orbital
In atomic theory and quantum mechanics, an atomic orbital is a Function (mathematics), function describing the location and wave-like behavior of an electron in an atom. This function can be used to calculate the probability of finding any electr ...
s will have the same symmetry as the HOMO
''Homo'' () is the genus that emerged in the (otherwise extinct) genus ''Australopithecus'' that encompasses the extant species ''Homo sapiens'' ( modern humans), plus several extinct species classified as either ancestral to or closely relate ...
of the product (a hexadiene
1,5-Hexadiene is the organic compound with the formula (CH)(CH=CH). It is a colorless, volatile liquid. It is used as a crosslinking agent and precursor to a variety of other compounds.
Synthesis
1,5-Hexadiene is produced commercially by the eth ...
). The only way to accomplish this is through a conrotatory
An electrocyclic reaction can either be classified as conrotatory or disrotatory based on the rotation at each end of the molecule. In conrotatory mode, both atomic orbitals of the end groups turn in the same direction (such as both atomic orbital ...
ring-opening which results in opposite signs for the terminal lobes.
Stereospecificity of electrocyclic reactions
When performing an electrocyclic reaction, it is often desirable to predict the cis/trans geometry of the reaction's product. The first step in this process is to determine whether a reaction proceeds through conrotation or disrotation. The table below shows the selectivity rules for thermal and photochemical electrocyclic reactions. For the example given below, the thermal reaction of (trans,cis,trans)-octa-2,4,6-triene will happen through a disrotatory mechanism. After determining the type of rotation, whether the product will be cis or trans can be determined by examining the starting molecule. In the example below, the disrotation causes both methyls to point upwards, causing the product to be cis-dimethylcyclohexadiene. In addition, thetorquoselectivity Torquoselectivity is a special kind of stereoselectivity observed in electrocyclic reactions in organic chemistry, defined as "the preference for inward or outward rotation of substituents in conrotatory
or disrotatory electrocyclic reactions." Torq ...
in an electrocyclic reaction refers to the direction of rotation. For example, a reaction that is conrotatory can still rotate in two directions, producing enantiomeric products. A reaction that is torquoselective restricts one of these directions of rotation (partially or completely) to produce a product in enantiomeric excess
In stereochemistry, enantiomeric excess (ee) is a measurement of purity used for chiral substances. It reflects the degree to which a sample contains one enantiomer in greater amounts than the other. A racemic mixture has an ee of 0%, while a sin ...
.

Mechanism of thermal reactions
Woodward–Hoffmann rules
 Correlation diagrams, which connect the molecular orbitals of the reactant to those of the product having the same symmetry, can then be constructed for the two processes.
Correlation diagrams, which connect the molecular orbitals of the reactant to those of the product having the same symmetry, can then be constructed for the two processes.
 These correlation diagrams indicate that only a conrotatory ring opening of 3,4-dimethylcyclobutene is symmetry allowed whereas only a disrotatory ring opening of 5,6-dimethylcyclohexa-1,3-diene is symmetry allowed. This is because only in these cases would maximum orbital overlap occur in the transition state. Also, the formed product would be in a ground state rather than an excited state.
These correlation diagrams indicate that only a conrotatory ring opening of 3,4-dimethylcyclobutene is symmetry allowed whereas only a disrotatory ring opening of 5,6-dimethylcyclohexa-1,3-diene is symmetry allowed. This is because only in these cases would maximum orbital overlap occur in the transition state. Also, the formed product would be in a ground state rather than an excited state.
Frontier molecular orbital theory
According to the frontier molecular orbital theory, the sigma bond in the ring will open in such a way that the resulting p-orbitals will have the same symmetry as the HOMO of the product. For the 5,6-dimethylcyclohexa-1,3-diene, only a disrotatory mode would result in p-orbitals having the same symmetry as the HOMO of hexatriene. For the 3,4-dimethylcyclobutene, on the other hand, only a conrotatory mode would result in p-orbitals having the same symmetry as the HOMO of butadiene.
For the 5,6-dimethylcyclohexa-1,3-diene, only a disrotatory mode would result in p-orbitals having the same symmetry as the HOMO of hexatriene. For the 3,4-dimethylcyclobutene, on the other hand, only a conrotatory mode would result in p-orbitals having the same symmetry as the HOMO of butadiene.
Mechanism of photochemical reactions
If the ring opening of 3,4-dimethylcyclobutene were carried out under photochemical conditions the resulting electrocyclization would be occur through a disrotatory mode instead of a conrotatory mode as can be seen by the correlation diagram for the allowed excited state ring opening reaction. Only a disrotatory mode, in which symmetry about a reflection plane is maintained throughout the reaction, would result in maximum orbital overlap in the transition state. Also, once again, this would result in the formation of a product that is in an excited state of comparable stability to the excited state of the reactant compound.
Only a disrotatory mode, in which symmetry about a reflection plane is maintained throughout the reaction, would result in maximum orbital overlap in the transition state. Also, once again, this would result in the formation of a product that is in an excited state of comparable stability to the excited state of the reactant compound.
Electrocyclic reactions in biological systems
Electrocyclic reactions occur frequently in nature. One of the most common such electrocyclizations is the biosynthesis of vitamin D3.
Electrocyclic reactions in organic synthesis
An often studied electrocyclic reaction is the conrotatory thermal ring-opening ofbenzocyclobutene
Benzocyclobutene (BCB) is a benzene ring fused to a cyclobutane ring. It has chemical formula .
BCB is frequently used to create photosensitive polymers. BCB-based polymer dielectrics may be spun on or applied to various substrates for use in ...
. The reaction product is a very unstable ortho-quinodimethane but this molecule can be trapped in an endo addition with a strong dienophile such as maleic anhydride
Maleic anhydride is an organic compound with the formula C2H2(CO)2O. It is the acid anhydride of maleic acid. It is a colorless or white solid with an acrid odor. It is produced industrially on a large scale for applications in coatings and poly ...
to the Diels-Alder adduct. The chemical yield
In chemistry, yield, also referred to as reaction yield, is a measure of the quantity of moles of a product formed in relation to the reactant consumed, obtained in a chemical reaction, usually expressed as a percentage. Yield is one of the pr ...
for the ring opening of the benzocyclobutane depicted in ''scheme 2'' is found to depend on the nature of the substituent
A substituent is one or a group of atoms that replaces (one or more) atoms, thereby becoming a moiety in the resultant (new) molecule. (In organic chemistry and biochemistry, the terms ''substituent'' and ''functional group'', as well as ''side ...
R. With a reaction solvent
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for ...
such as toluene
Toluene (), also known as toluol (), is a substituted aromatic hydrocarbon. It is a colorless, water-insoluble liquid with the smell associated with paint thinners. It is a mono-substituted benzene derivative, consisting of a methyl group (CH3) at ...
and a reaction temperature of 110 °C, the yield increases going from methyl
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many ...
to isobutylmethyl to (trimethylsilyl)methyl. The increased reaction rate
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per unit ...
for the trimethylsilyl
A trimethylsilyl group (abbreviated TMS) is a functional group in organic chemistry. This group consists of three methyl groups bonded to a silicon atom minus;Si(CH3)3 which is in turn bonded to the rest of a molecule. This structural group is ch ...
compound can be explained by silicon hyperconjugation as the βC-Si bond weakens the cyclobutane C-C bond by donating electrons.
 A
A biomimetic
Biomimetics or biomimicry is the emulation of the models, systems, and elements of nature for the purpose of solving complex human problems. The terms "biomimetics" and "biomimicry" are derived from grc, βίος (''bios''), life, and μίμησ ...
electrocyclic cascade reaction
A cascade reaction, also known as a domino reaction or tandem reaction, is a chemical process that comprises at least two consecutive reactions such that each subsequent reaction occurs only in virtue of the chemical functionality formed in the p ...
was discovered in relation to the isolation and synthesis of certain endiandric acids:
chiral auxiliary
In stereochemistry, a chiral auxiliary is a stereogenic group or unit that is temporarily incorporated into an organic compound in order to control the stereochemical outcome of the synthesis. The chirality present in the auxiliary can bias the ...
, and the Nazarov reaction has been performed catalytically using chiral Lewis acids
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any spe ...
, Brønsted acids and chiral amines.''Asymmetric electrocyclic reactions'', S. Thompson, A. G. Coyne, P. C. Knipe and M. D. Smith, Chem. Soc. Rev., 2011, 40, pp 4217-4231
References
{{DEFAULTSORT:Electrocyclic Reaction Rearrangement reactions Pericyclic reactions Reaction mechanisms