Conjugated system on:
[Wikipedia]
[Google]
[Amazon]


 In
In
 Conjugation is possible by means of alternating single and
Conjugation is possible by means of alternating single and  A common model for the treatment of conjugated molecules is a composite valence bond / Hückel molecular orbital theory (VB/HMOT) treatment, in which the σ framework of the molecule is separated from the π system (or systems) of the molecule (''see the article on the
A common model for the treatment of conjugated molecules is a composite valence bond / Hückel molecular orbital theory (VB/HMOT) treatment, in which the σ framework of the molecule is separated from the π system (or systems) of the molecule (''see the article on the  This simple model for chemical bonding is successful for the description of most normal-valence molecules consisting of only s- and p-block elements, although systems that involve electron-deficient bonding, including nonclassical carbocations, lithium and boron clusters, and hypervalent centers require significant modifications in which σ bonds are also allowed to delocalize and are perhaps better treated with canonical molecular orbitals that are delocalized over the entire molecule. Likewise, d- and f-block organometallics are also inadequately described by this simple model. Bonds in strained small rings (such as cyclopropane or epoxide) are not well-described by strict σ/π separation, as bonding between atoms in the ring consists of "
This simple model for chemical bonding is successful for the description of most normal-valence molecules consisting of only s- and p-block elements, although systems that involve electron-deficient bonding, including nonclassical carbocations, lithium and boron clusters, and hypervalent centers require significant modifications in which σ bonds are also allowed to delocalize and are perhaps better treated with canonical molecular orbitals that are delocalized over the entire molecule. Likewise, d- and f-block organometallics are also inadequately described by this simple model. Bonds in strained small rings (such as cyclopropane or epoxide) are not well-described by strict σ/π separation, as bonding between atoms in the ring consists of "
 There are also other types of interactions that generalize the idea of interacting p orbitals in a conjugated system. The concept of ''hyperconjugation'' holds that certain σ bonds can also delocalize into a low-lying unoccupied orbital of a π system or an unoccupied p orbital. Hyperconjugation is commonly invoked to explain the stability of alkyl substituted radicals and carbocations. Hyperconjugation is less important for species in which all atoms satisfy the octet rule, but a recent computational study supports hyperconjugation as the origin of the increased stability of alkenes with a higher degree of substitution (
There are also other types of interactions that generalize the idea of interacting p orbitals in a conjugated system. The concept of ''hyperconjugation'' holds that certain σ bonds can also delocalize into a low-lying unoccupied orbital of a π system or an unoccupied p orbital. Hyperconjugation is commonly invoked to explain the stability of alkyl substituted radicals and carbocations. Hyperconjugation is less important for species in which all atoms satisfy the octet rule, but a recent computational study supports hyperconjugation as the origin of the increased stability of alkenes with a higher degree of substitution (

 Not all compounds with alternating double and single bonds are aromatic.
Not all compounds with alternating double and single bonds are aromatic.

 Conjugated chromophores are found in many
Conjugated chromophores are found in many  Conjugated polymer nanoparticles (PDots) are assembled from hydrophobic fluorescent conjugated polymers, along with
Conjugated polymer nanoparticles (PDots) are assembled from hydrophobic fluorescent conjugated polymers, along with
 In
In theoretical chemistry
Theoretical chemistry is the branch of chemistry which develops theoretical generalizations that are part of the theoretical arsenal of modern chemistry: for example, the concepts of chemical bonding, chemical reaction, valence, the surface o ...
, a conjugated system is a system of connected p-orbital
In atomic theory and quantum mechanics, an atomic orbital is a function describing the location and wave-like behavior of an electron in an atom. This function can be used to calculate the probability of finding any electron of an atom in any ...
s with delocalized electron
In chemistry, delocalized electrons are electrons in a molecule, ion or solid metal that are not associated with a single atom or a covalent bond.IUPAC Gold Boo''delocalization''/ref>
The term delocalization is general and can have slightly dif ...
s in a molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and b ...
, which in general lowers the overall energy of the molecule and increases stability
Stability may refer to:
Mathematics
*Stability theory, the study of the stability of solutions to differential equations and dynamical systems
** Asymptotic stability
** Linear stability
** Lyapunov stability
** Orbital stability
** Structural sta ...
. It is conventionally represented as having alternating single and multiple bonds. Lone pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC '' Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. L ...
s, radicals
Radical may refer to:
Politics and ideology Politics
*Radical politics, the political intent of fundamental societal change
*Radicalism (historical), the Radical Movement that began in late 18th century Britain and spread to continental Europe and ...
or carbenium ion
A carbenium ion is a positive ion with the structure RR′R″C+, that is, a chemical species with a trivalent carbon that bears a +1 formal charge.
In older literature the name carbonium ion was used for this class, but now it refers exclusivel ...
s may be part of the system, which may be cyclic
Cycle, cycles, or cyclic may refer to:
Anthropology and social sciences
* Cyclic history, a theory of history
* Cyclical theory, a theory of American political history associated with Arthur Schlesinger, Sr.
* Social cycle, various cycles in so ...
, acyclic, linear
Linearity is the property of a mathematical relationship ('' function'') that can be graphically represented as a straight line. Linearity is closely related to '' proportionality''. Examples in physics include rectilinear motion, the linear ...
or mixed. The term "conjugated" was coined in 1899 by the German chemist Johannes Thiele Johannes Thiele may refer to:
*Johannes Thiele (zoologist)
*Johannes Thiele (chemist)
Friedrich Karl Johannes Thiele (May 13, 1865 – April 17, 1918) was a German chemist and a prominent professor at several universities, including those in ...
.
Conjugation is the overlap
Overlap may refer to:
* In set theory, an overlap of elements shared between sets is called an intersection, as in a Venn diagram.
* In music theory, overlap is a synonym for reinterpretation of a chord at the boundary of two musical phrases
* O ...
of one p-orbital with another across an adjacent σ bond (in transition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that can ...
s, d-orbitals can be involved).
A conjugated system has a region of overlapping p-orbitals, bridging the interjacent locations that simple diagrams illustrate as not having a π bond. They allow a delocalization of π electrons across all the adjacent aligned p-orbitals.
The π electrons do not belong to a single bond or atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, a ...
, but rather to a group of atoms.
Molecules containing conjugated systems of orbitals and electrons are called conjugated molecules, which have overlapping p orbitals on three or more atoms. Some simple organic
Organic may refer to:
* Organic, of or relating to an organism, a living entity
* Organic, of or relating to an anatomical organ
Chemistry
* Organic matter, matter that has come from a once-living organism, is capable of decay or is the product ...
conjugated molecules are 1,3-butadiene, benzene, and allylic
In organic chemistry, an allyl group is a substituent with the structural formula , where R is the rest of the molecule. It consists of a methylene bridge () attached to a vinyl group (). The name is derived from the scientific name for garlic, . ...
carbocations. The largest conjugated systems are found in graphene
Graphene () is an allotrope of carbon consisting of a Single-layer materials, single layer of atoms arranged in a hexagonal lattice nanostructure.
, graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on la ...
, conductive polymer
Conductive polymers or, more precisely, intrinsically conducting polymers (ICPs) are organic polymers that conduct electricity. Such compounds may have metallic conductivity or can be semiconductors. The biggest advantage of conductive polymers ...
s and carbon nanotube
A scanning tunneling microscopy image of a single-walled carbon nanotube
Rotating single-walled zigzag carbon nanotube
A carbon nanotube (CNT) is a tube made of carbon with diameters typically measured in nanometers.
''Single-wall carbon na ...
s.
Chemical bonding in conjugated systems
 Conjugation is possible by means of alternating single and
Conjugation is possible by means of alternating single and double bond
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist betwee ...
s in which each atom supplies a p orbital perpendicular to the plane of the molecule. However, that is not the only way for conjugation to take place. As long as each contiguous atom in a chain has an available p orbital, the system can be considered conjugated. For example, furan
Furan is a heterocyclic organic compound, consisting of a five-membered aromatic ring with four carbon atoms and one oxygen atom. Chemical compounds containing such rings are also referred to as furans.
Furan is a colorless, flammable, highl ...
is a five-membered ring with two alternating double bonds flanking an oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements ...
. The oxygen has two lone pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC '' Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. L ...
s, one of which occupies a p orbital perpendicular to the ring on that position, thereby maintaining the conjugation of that five-membered ring by overlap with the perpendicular p orbital on each of the adjacent carbon atoms. The other lone pair remains in plane and does not participate in conjugation.
In general, any sp2 or sp-hybridized carbon or heteroatom
In chemistry, a heteroatom () is, strictly, any atom that is not carbon or hydrogen.
Organic chemistry
In practice, the term is usually used more specifically to indicate that non-carbon atoms have replaced carbon in the backbone of the molecula ...
, including ones bearing an empty orbital or lone pair orbital, can participate in conjugated systems, though lone pairs do not always participate in a conjugated system. For example, in pyridine, the nitrogen atom already participates in the conjugated system through a formal double bond with an adjacent carbon, so the lone pair remains in the plane of the ring in an sp2 hybrid orbital and does not participate in the conjugation. A requirement for conjugation is orbital overlap; thus, the conjugated system must be planar (or nearly so). As a consequence, lone pairs which do participate in conjugated systems will occupy orbitals of pure p character instead of sp''n'' hybrid orbitals typical for nonconjugated lone pairs.
 A common model for the treatment of conjugated molecules is a composite valence bond / Hückel molecular orbital theory (VB/HMOT) treatment, in which the σ framework of the molecule is separated from the π system (or systems) of the molecule (''see the article on the
A common model for the treatment of conjugated molecules is a composite valence bond / Hückel molecular orbital theory (VB/HMOT) treatment, in which the σ framework of the molecule is separated from the π system (or systems) of the molecule (''see the article on the sigma-pi and equivalent-orbital models The σ-π model and equivalent-orbital model refer to two possible representations of molecules in valence bond theory. The σ-π model differentiates bonds and lone pairs of σ symmetry from those of π symmetry, while the equivalent-orbital model ...
for this model and an alternative treatment''). Although σ bonding can be treated using a delocalized approach as well, it is generally the π bonding that is being considered when delocalized bonding is invoked in the context of simple organic molecules.
''Sigma (σ) framework'': The σ framework is described by a strictly localized bonding scheme and consists of σ bonds formed from the interactions between sp3-, sp2-, and sp- hybridized atomic orbitals on the main group elements (and 1s atomic orbitals on hydrogen), together with localized lone pairs derived from filled, nonbonding hybrid orbitals. The interaction that results in σ bonding takes the form of head-to-head overlap of the larger lobe of each hybrid orbital (or the single spherical lobe of a hydrogen 1s orbital). Each atomic orbital contributes one electron when the orbitals overlap pairwise to form two-electron σ bonds, or two electrons when the orbital constitutes a lone pair. These localized orbitals (bonding and non-bonding) are all located in the plane of the molecule, with σ bonds mainly localized between nuclei along the internuclear axis.
''Pi (π) system or systems'': Orthogonal to the σ framework described above, π bonding occurs above and below the plane of the molecule where σ bonding takes place. The π system(s) of the molecule are formed by the interaction of unhybridized p atomic orbitals on atoms employing sp2- and sp-hybridization. The interaction that results in π bonding takes place between p orbitals that are adjacent by virtue of a σ bond joining the atoms and takes the form of side-to-side overlap of the two equally large lobes that make up each p orbital. Atoms that are sp3-hybridized do not have an unhybridized p orbital available for participation in π bonding and their presence necessarily terminates a π system or separates two π systems. A basis p orbital that takes part in a π system can contribute one electron (which corresponds to half of a formal "double bond"), two electrons (which corresponds to a delocalized "lone pair"), or zero electrons (which corresponds to a formally "empty" orbital). Bonding for π systems formed from the overlap of more than two p orbitals is handled using the Hückel approach to obtain a zeroth order (qualitative) approximation of the π symmetry molecular orbitals that result from delocalized π bonding.
 This simple model for chemical bonding is successful for the description of most normal-valence molecules consisting of only s- and p-block elements, although systems that involve electron-deficient bonding, including nonclassical carbocations, lithium and boron clusters, and hypervalent centers require significant modifications in which σ bonds are also allowed to delocalize and are perhaps better treated with canonical molecular orbitals that are delocalized over the entire molecule. Likewise, d- and f-block organometallics are also inadequately described by this simple model. Bonds in strained small rings (such as cyclopropane or epoxide) are not well-described by strict σ/π separation, as bonding between atoms in the ring consists of "
This simple model for chemical bonding is successful for the description of most normal-valence molecules consisting of only s- and p-block elements, although systems that involve electron-deficient bonding, including nonclassical carbocations, lithium and boron clusters, and hypervalent centers require significant modifications in which σ bonds are also allowed to delocalize and are perhaps better treated with canonical molecular orbitals that are delocalized over the entire molecule. Likewise, d- and f-block organometallics are also inadequately described by this simple model. Bonds in strained small rings (such as cyclopropane or epoxide) are not well-described by strict σ/π separation, as bonding between atoms in the ring consists of "bent bond
In organic chemistry, a bent bond, also known as a banana bond, is a type of covalent chemical bond with a geometry somewhat reminiscent of a banana. The term itself is a general representation of electron density or configuration resembling a ...
s" or "banana bonds" that are bowed outward and are intermediate in nature between σ and π bonds. Nevertheless, organic chemists frequently use the language of this model to rationalize the structure and reactivity of typical organic compounds.
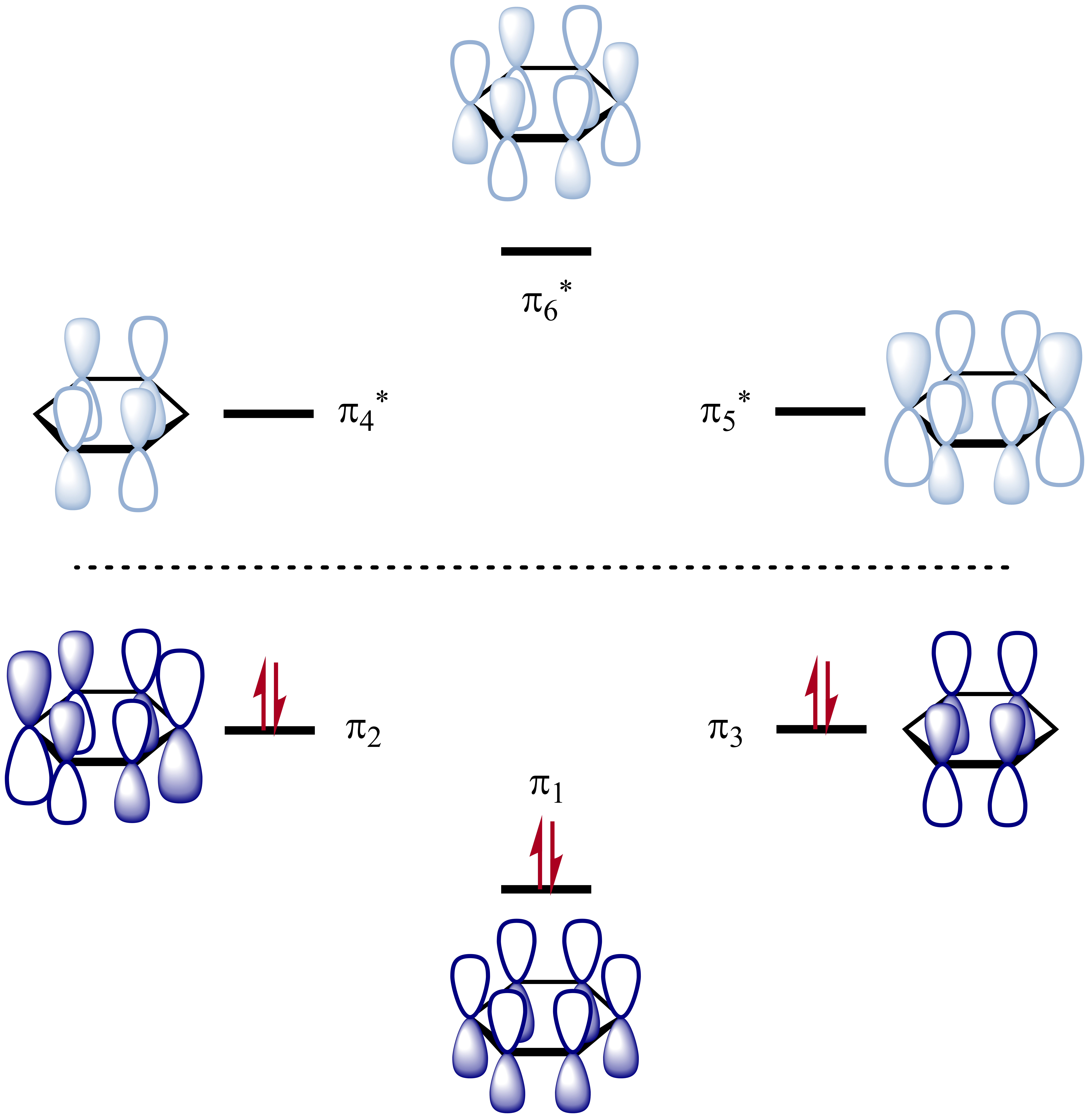
Electrons in conjugated π systems are shared by all adjacent sp2- and sp-hybridized atoms that contribute overlapping, parallel p atomic orbitals. As such, the atoms and π-electrons involved behave as one large bonded system. These systems are often referred to n''-center ''k-''electron π-bonds,' compactly denoted by the symbol Π, to emphasize this behavior. For example, the delocalized π electrons in acetate anion and benzene are said to be involved in Π and Π systems, respectively (''see the article on three-center four-electron bonding''). It is important to recognize that, generally speaking, these multi-center bonds correspond to the occupation of several molecular orbitals (MOs) with varying degrees of bonding or non-bonding character (filling of orbitals with antibonding character is uncommon). Each one is occupied by one or two electrons in accordance with the aufbau principle
The aufbau principle , from the German ''Aufbauprinzip'' (building-up principle), also called the aufbau rule, states that in the ground state of an atom or ion, electrons fill subshells of the lowest available energy, then they fill subshells ...
and Hund's rule. Cartoons showing overlapping p orbitals, like the one for benzene below, show the basis p atomic orbitals ''before'' they are combined to form molecular orbitals. In compliance with the Pauli exclusion principle
In quantum mechanics, the Pauli exclusion principle states that two or more identical particles with half-integer spins (i.e. fermions) cannot occupy the same quantum state within a quantum system simultaneously. This principle was formula ...
, overlapping p orbitals ''do not'' result in the formation of one large MO containing more than two electrons.
Hückel MO theory is commonly used approach to obtain a zeroth order picture of delocalized π molecular orbitals, including the mathematical sign of the wavefunction at various parts of the molecule and the locations of nodal planes. It is particularly easy to apply for conjugated hydrocarbons and provides a reasonable approximation as long as the molecule is assumed to be planar with good overlap of p orbitals.
Stabilization energy
The quantitative estimation of stabilization from conjugation is notoriously contentious and depends on the implicit assumptions that are made when comparing reference systems or reactions. The energy of stabilization is known as the resonance energy when formally defined as the difference in energy between the real chemical species and the hypothetical species featuring localized π bonding that corresponds to the most stableresonance form
In chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures (or ''forms'', also variously known as ''resonance structures'' or ' ...
. This energy cannot be measured, and a precise definition accepted by most chemists will probably remain elusive. Nevertheless, some broad statements can be made. In general, stabilization is more significant for cationic systems than neutral ones. For buta-1,3-diene
1,3-Butadiene () is the organic compound with the formula (CH2=CH)2. It is a colorless gas that is easily condensed to a liquid. It is important industrially as a precursor to synthetic rubber. The molecule can be viewed as the union of two viny ...
, a crude measure of stabilization is the activation energy
In chemistry and physics, activation energy is the minimum amount of energy that must be provided for compounds to result in a chemical reaction. The activation energy (''E''a) of a reaction is measured in joules per mole (J/mol), kilojoules p ...
for rotation of the C2-C3 bond. This places the resonance stabilization at around 6 kcal/mol. Comparison of heats of hydrogenation of 1,4-pentadiene and 1,3-pentadiene estimates a slightly more modest value of 3.5 kcal/mol. For comparison, allyl cation has a gas-phase rotation barrier of around 38 kcal/mol, a much greater penalty for loss of conjugation. Comparison of hydride ion affinities of propyl cation and allyl cation, corrected for inductive effects, results in a considerably lower estimate of the resonance energy at 20–22 kcal/mol. Nevertheless, it is clear that conjugation stabilizes allyl cation to a much greater extent than buta-1,3-diene. In contrast to the usually minor effect of neutral conjugation, aromatic stabilization can be considerable. Estimates for the resonance energy of benzene range from around 36–73 kcal/mol.
Generalizations and related concepts
 There are also other types of interactions that generalize the idea of interacting p orbitals in a conjugated system. The concept of ''hyperconjugation'' holds that certain σ bonds can also delocalize into a low-lying unoccupied orbital of a π system or an unoccupied p orbital. Hyperconjugation is commonly invoked to explain the stability of alkyl substituted radicals and carbocations. Hyperconjugation is less important for species in which all atoms satisfy the octet rule, but a recent computational study supports hyperconjugation as the origin of the increased stability of alkenes with a higher degree of substitution (
There are also other types of interactions that generalize the idea of interacting p orbitals in a conjugated system. The concept of ''hyperconjugation'' holds that certain σ bonds can also delocalize into a low-lying unoccupied orbital of a π system or an unoccupied p orbital. Hyperconjugation is commonly invoked to explain the stability of alkyl substituted radicals and carbocations. Hyperconjugation is less important for species in which all atoms satisfy the octet rule, but a recent computational study supports hyperconjugation as the origin of the increased stability of alkenes with a higher degree of substitution (Zaitsev's rule
In organic chemistry, Zaitsev's rule (or Saytzeff's rule, Saytzev's rule) is an empirical rule for predicting the favored alkene product(s) in elimination reactions. While at the University of Kazan, Russian chemist Alexander Zaitsev studied a v ...
).
''Homoconjugation'' is an overlap of two π-systems separated by a non-conjugating group, such as CH2. Unambiguous examples are comparatively rare in neutral systems, due to a comparatively minor energetic benefit that is easily overridden by a variety of other factors; however, they are common in cationic systems in which a large energetic benefit can be derived from delocalization of positive charge (''see the article on homoaromaticity
Homoaromaticity, in organic chemistry, refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thoug ...
for details.''). Neutral systems generally require constrained geometries favoring interaction to produce significant degrees of homoconjugation. In the example below, the carbonyl stretching frequencies of the IR spectra of the respective compounds demonstrate homoconjugation, or lack thereof, in the neutral ground state molecules.
Due to the partial π character of formally σ bonds in a cyclopropane ring, evidence for transmission of "conjugation" through cyclopropanes has also been obtained.
Two appropriately aligned π systems whose ends meet at right angles can engage in '' spiroconjugation''.
Conjugated cyclic compounds

Cyclic compound
A cyclic compound (or ring compound) is a term for a compound in the field of chemistry in which one or more series of atoms in the compound is connected to form a ring. Rings may vary in size from three to many atoms, and include examples where ...
s can be partly or completely conjugated. Annulenes
Annulenes are monocyclic hydrocarbons that contain the maximum number of non-cumulated or conjugated double bonds (' mancude'). They have the general formula CnHn (when ''n'' is an even number) or C''n''H''n''+1 (when ''n'' is an odd number). The ...
, completely conjugated monocyclic hydrocarbons, may be aromatic, nonaromatic or antiaromatic.
Aromatic compounds
Compounds that have a monocyclic, planar conjugated system containing (4''n'' + 2) π-electrons for whole numbers ''n'' arearomatic
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to satur ...
and exhibit an unusual stability. The classic example benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms ...
has a system of six π electrons, which, together with the planar ring of C–C σ bonds containing 12 electrons and radial C–H σ bonds containing six electrons, forms the thermodynamically and kinetically stable '' benzene ring'', the common core of the benzenoid aromatic compounds. For benzene itself, there are two equivalent conjugated contributing Lewis structures (the so-called Kekulé structures) that predominate. The true electronic structure is therefore a quantum-mechanical combination (resonance hybrid) of these contributors, which results in the experimentally observed C–C bonds which are intermediate between single and double bonds and of equal strength and length. In the molecular orbital picture, the six p atomic orbitals of benzene combine to give six molecular orbitals. Three of these orbitals, which lie at lower energies than the isolated p orbital and are therefore net bonding in character (one molecular orbital is strongly bonding, while the other two are equal in energy but bonding to a lesser extent) are occupied by six electrons, while three destabilized orbitals of overall antibonding character remain unoccupied. The result is strong thermodynamic and kinetic aromatic stabilization. Both models describe rings of π electron density above and below the framework of C–C σ bonds.
Nonaromatic and antiaromatic compounds
Cyclooctatetraene
1,3,5,7-Cyclooctatetraene (COT) is an unsaturated derivative of cyclooctane, with the formula C8H8. It is also known as nnulene. This polyunsaturated hydrocarbon is a colorless to light yellow flammable liquid at room temperature. Because of ...
, for example, possesses alternating single and double bonds. The molecule typically adopts a "tub" conformation. Because the p orbitals of the molecule do not align themselves well in this non-planar molecule, the π bonds are essentially isolated and not conjugated. The lack of conjugation allows the 8 π electron molecule to avoid antiaromaticity
Antiaromaticity is a chemical property of a cyclic molecule with a π electron system that has higher energy, i.e., it is less stable due to the presence of 4n delocalised (π or lone pair) electrons in it, as opposed to aromaticity. Unlike aroma ...
, a destabilizing effect associated with cyclic, conjugated systems containing 4''n'' π (''n'' = 0, 1, 2, ...) electrons. This effect is due to the placement of two electrons into two degenerate nonbonding (or nearly nonbonding) orbitals of the molecule, which, in addition to drastically reducing the thermodynamic stabilization of delocalization, would either force the molecule to take on triplet diradical character, or cause it to undergo Jahn-Teller distortion to relieve the degeneracy. This has the effect of greatly increasing the kinetic reactivity of the molecule. Because of the lack of long-range interactions, cyclooctatetraene takes on a nonplanar conformation and is nonaromatic in character, behaving as a typical alkene. In contrast, derivatives of the cyclooctatetraene dication and dianion have been found to be planar experimentally, in accord with the prediction that they are stabilized aromatic systems with 6 and 10 π electrons, respectively. Because antiaromaticity is a property that molecules try to avoid whenever possible, only a few experimentally observed species are believed to be antiaromatic. Cyclobutadiene
Cyclobutadiene is an organic compound with the formula . It is very reactive owing to its tendency to dimerize. Although the parent compound has not been isolated, some substituted derivatives are robust and a single molecule of cyclobutadiene is ...
and cyclopentadienyl cation are commonly cited as examples of antiaromatic systems.
In pigments
In a conjugated pi-system, electrons are able to capture certain photons as the electrons resonate along a certain distance of p-orbitals - similar to how a radio antenna detects photons along its length. Typically, the more conjugated (longer) the pi-system is, the longer the wavelength of photon can be captured. Compounds whose molecules contain a sufficient number of conjugated bonds can absorb light in the visible region, and therefore appear colorful to the eye, usually appearing yellow or red. Manydye
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution and ...
s make use of conjugated electron systems to absorb visible light
Light or visible light is electromagnetic radiation that can be perceived by the human eye. Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequencies of 750–420 t ...
, giving rise to strong colors. For example, the long conjugated hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ...
chain in beta-carotene leads to its strong orange color. When an electron in the system absorbs a photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they alwa ...
of light of the right wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, tr ...
, it can be promoted to a higher energy level. A simple model of the energy levels is provided by the quantum-mechanical
Quantum mechanics is a fundamental theory in physics that provides a description of the physical properties of nature at the scale of atoms and subatomic particles. It is the foundation of all quantum physics including quantum chemistry, qu ...
problem of a one-dimensional particle in a box of length L, representing the movement of a π electron along a long conjugated chain of carbon atoms. In this model the lowest possible absorption energy corresponds to the energy difference between the highest occupied molecular orbital (HOMO
''Homo'' () is the genus that emerged in the (otherwise extinct) genus '' Australopithecus'' that encompasses the extant species ''Homo sapiens'' ( modern humans), plus several extinct species classified as either ancestral to or closely rela ...
) and the lowest unoccupied molecular orbital (LUMO). For a chain of ''n'' C=C bonds or 2''n'' carbon atoms in the molecular ground state
The ground state of a quantum-mechanical system is its stationary state of lowest energy; the energy of the ground state is known as the zero-point energy of the system. An excited state is any state with energy greater than the ground state. ...
, there are 2''n'' π electrons occupying ''n'' molecular orbitals, so that the energy gap is
:
Since the box length ''L'' increases approximately linearly with the number of C=C bonds ''n'', this means that the energy Δ''E'' of a photon absorbed in the HOMO–LUMO transition is approximately proportional to 1/''n''. The photon wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, tr ...
λ = ''hc''/Δ''E'' is then approximately proportional to ''n''. Although this model is very approximate, λ does in general increase with ''n'' (or ''L'') for similar molecules. For example, the HOMO–LUMO absorption wavelengths for conjugated butadiene
1,3-Butadiene () is the organic compound with the formula (CH2=CH)2. It is a colorless gas that is easily condensed to a liquid. It is important industrially as a precursor to synthetic rubber. The molecule can be viewed as the union of two v ...
, hexatriene and octatetraene are 217 nm, 252 nm and 304 nm respectively. However, for good numerical agreement of the particle in a box model with experiment, the single-bond/double-bond bond length alternations of the polyenes must be taken into account. Alternatively, one can use the Hückel method which is also designed to model the electronic structure of conjugated systems.
Many electronic transitions in conjugated π-systems are from a predominantly bonding molecular orbital
In chemistry, a molecular orbital is a mathematical function describing the location and wave-like behavior of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of find ...
(MO) to a predominantly antibonding
In chemical bonding theory, an antibonding orbital is a type of molecular orbital that weakens the chemical bond between two atoms and helps to raise the energy of the molecule relative to the separated atoms. Such an orbital has one or more no ...
MO (π to π*), but electrons from non-bonding lone pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC '' Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. L ...
s can also be promoted to a π-system MO (n to π*) as often happens in charge-transfer complex
In chemistry, a charge-transfer (CT) complex or electron-donor-acceptor complex describes a type of supramolecular assembly of two or more molecules or ions. The assembly consists of two molecules that self-attract through electrostatic forc ...
es. A HOMO to LUMO transition is made by an electron if it is allowed by the selection rules
In physics and chemistry, a selection rule, or transition rule, formally constrains the possible transitions of a system from one quantum state to another. Selection rules have been derived for electromagnetic transitions in molecules, in atoms, i ...
for electromagnetic transitions. Conjugated systems of fewer than eight conjugated double bonds absorb only in the ultraviolet region and are colorless to the human eye. With every double bond added, the system absorbs photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they alwa ...
s of longer wavelength (and lower energy), and the compound ranges from yellow to red in color. Compounds that are blue or green typically do not rely on conjugated double bonds alone.
This absorption of light in the ultraviolet to visible spectrum can be quantified using ultraviolet–visible spectroscopy
UV spectroscopy or UV–visible spectrophotometry (UV–Vis or UV/Vis) refers to absorption spectroscopy or reflectance spectroscopy in part of the ultraviolet and the full, adjacent visible regions of the electromagnetic spectrum. Being relativ ...
, and forms the basis for the entire field of photochemistry
Photochemistry is the branch of chemistry concerned with the chemical effects of light. Generally, this term is used to describe a chemical reaction caused by absorption of ultraviolet ( wavelength from 100 to 400 nm), visible light (400� ...
.
Conjugated systems that are widely used for synthetic pigment
A pigment is a colored material that is completely or nearly insoluble in water. In contrast, dyes are typically soluble, at least at some stage in their use. Generally dyes are often organic compounds whereas pigments are often inorganic compou ...
s and dye
A dye is a colored substance that chemically bonds to the substrate to which it is being applied. This distinguishes dyes from pigments which do not chemically bind to the material they color. Dye is generally applied in an aqueous solution and ...
s are diazo
The diazo group is an organic moiety consisting of two linked nitrogen atoms ( azo) at the terminal position. Overall charge neutral organic compounds containing the diazo group bound to a carbon atom are called diazo compounds or diazoalkanes ...
and azo compounds and phthalocyanine compounds.
Phthalocyanine compounds
Conjugated systems not only have low energy excitations in the visible spectral region but they also accept or donate electrons easily.Phthalocyanine
Phthalocyanine () is a large, aromatic, macrocyclic, organic compound with the formula and is of theoretical or specialized interest in chemical dyes and photoelectricity.
It is composed of four isoindole units linked by a ring of nitrogen ato ...
s, which, like Phthalocyanine Blue BN
Copper phthalocyanine (CuPc), also called phthalocyanine blue, phthalo blue and many other names, is a bright, crystalline, synthetic blue pigment from the group of phthalocyanine dyes. Its brilliant blue is frequently used in paints and dyes ...
and Phthalocyanine Green G
Phthalocyanine green G, which has many commercial names, is a synthetic green pigment from the group of phthalocyanine dyes, a complex of copper(II) with chlorinated phthalocyanine. It is a soft green powder, which is insoluble in water.. It is ...
, often contain a transition metal ion, exchange an electron with the complexed transition metal ion that easily changes its oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
. Pigments and dyes like these are charge-transfer complex
In chemistry, a charge-transfer (CT) complex or electron-donor-acceptor complex describes a type of supramolecular assembly of two or more molecules or ions. The assembly consists of two molecules that self-attract through electrostatic forc ...
es.
Porphyrins and similar compounds
Porphyrin
Porphyrins ( ) are a group of heterocyclic macrocycle organic compounds, composed of four modified pyrrole subunits interconnected at their α carbon atoms via methine bridges (=CH−). The parent of porphyrin is porphine, a rare chemical com ...
s have conjugated molecular ring systems (macrocycle
Macrocycles are often described as molecules and ions containing a ring of twelve or more atoms. Classical examples include the crown ethers, calixarenes, porphyrins, and cyclodextrins. Macrocycles describe a large, mature area of chemistry.
...
s) that appear in many enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
s of biological systems. As a ligand
In coordination chemistry, a ligand is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's elect ...
, porphyrin forms numerous complexes with metallic ions like iron
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in ...
in hemoglobin
Hemoglobin (haemoglobin BrE) (from the Greek word αἷμα, ''haîma'' 'blood' + Latin ''globus'' 'ball, sphere' + ''-in'') (), abbreviated Hb or Hgb, is the iron-containing oxygen-transport metalloprotein present in red blood cells (erythroc ...
that colors blood red. Hemoglobin transports oxygen to the cells of our bodies. Porphyrin–metal complexes often have strong colors. A similar molecular structural ring unit called chlorin is similarly complexed with magnesium
Magnesium is a chemical element with the symbol Mg and atomic number 12. It is a shiny gray metal having a low density, low melting point and high chemical reactivity. Like the other alkaline earth metals (group 2 of the periodic ...
instead of iron when forming part of the most common forms of chlorophyll
Chlorophyll (also chlorophyl) is any of several related green pigments found in cyanobacteria and in the chloroplasts of algae and plants. Its name is derived from the Greek words , ("pale green") and , ("leaf"). Chlorophyll allow plants to ...
molecules, giving them a green color. Another similar macrocycle unit is corrin
Corrin is a heterocyclic compound. It is the parent macrocycle related to the substituted derivative that is found in vitamin B12. Its name reflects that it is the "core" of vitamin B12 (cobalamins).Nelson, D. L.; Cox, M. M. "Lehninger, Princi ...
, which complexes with cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, p ...
when forming part of cobalamin
Vitamin B12, also known as cobalamin, is a water-soluble vitamin involved in metabolism. It is one of eight B vitamins. It is required by animals, which use it as a cofactor in DNA synthesis, in both fatty acid and amino acid metabolism. ...
molecules, constituting Vitamin B12
Vitamin B12, also known as cobalamin, is a water-soluble vitamin involved in metabolism. It is one of eight B vitamins. It is required by animals, which use it as a cofactor in DNA synthesis, in both fatty acid and amino acid metabolism. ...
, which is intensely red. The corrin unit has six conjugated double bonds but is not conjugated all the way around its macrocycle ring.
Chromophores
Conjugated systems form the basis ofchromophore
A chromophore is the part of a molecule responsible for its color.
The color that is seen by our eyes is the one not absorbed by the reflecting object within a certain wavelength spectrum of visible light. The chromophore is a region in the mo ...
s, which are light-absorbing parts of a molecule that can cause a compound to be colored. Such chromophores are often present in various organic compounds and sometimes present in polymer
A polymer (; Greek '' poly-'', "many" + '' -mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
s that are colored or glow in the dark. Chromophores often consist of a series of conjugated bonds and/or ring systems, commonly aromatic, which can include C–C, C=C, C=O, or N=N bonds.
organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. Th ...
s including azo dyes
Azo dyes are organic compounds bearing the functional group R−N=N−R′, in which R and R′ are usually aryl and substituted aryl groups. They are a commercially important family of azo compounds, i.e. compounds containing the C-N=N ...
(also artificial food additives), compounds in fruits and vegetables (lycopene
Lycopene is an organic compound classified as a tetraterpene and a carotene. Lycopene (from the neo-Latin '' Lycopersicum'', the tomato species) is a bright red carotenoid hydrocarbon found in tomatoes and other red fruits and vegetables.
Occu ...
and anthocyanidin
Anthocyanidins are common plant pigments, the sugar-free counterparts of anthocyanins. They are based on the flavylium cation, an oxonium ion, with various groups substituted for its hydrogen atoms. They generally change color from red through ...
s), photoreceptors of the eye, and some pharmaceutical compounds such as the following:
amphiphilic
An amphiphile (from the Greek αμφις amphis, both, and φιλíα philia, love, friendship), or amphipath, is a chemical compound possessing both hydrophilic (''water-loving'', polar) and lipophilic (''fat-loving'') properties. Such a compo ...
polymers to provide water solubility. Pdots are important labels for single-molecule
A single-molecule experiment is an experiment that investigates the properties of individual molecules. Single-molecule studies may be contrasted with measurements on an ensemble or bulk collection of molecules, where the individual behavior of mo ...
fluorescence microscopy
A fluorescence microscope is an optical microscope that uses fluorescence instead of, or in addition to, scattering, reflection, and attenuation or absorption, to study the properties of organic or inorganic substances. "Fluorescence microscop ...
, based on high brightness, lack of blinking or dark fraction, and slow photobleaching
In optics, photobleaching (sometimes termed fading) is the photochemical alteration of a dye or a fluorophore molecule such that it is permanently unable to fluoresce. This is caused by cleaving of covalent bonds or non-specific reactions between ...
.
See also
*Resonance
Resonance describes the phenomenon of increased amplitude that occurs when the frequency of an applied periodic force (or a Fourier component of it) is equal or close to a natural frequency of the system on which it acts. When an oscil ...
* Hyperconjugation
*Cross-conjugation
Cross-conjugation is a special type of conjugation in a molecule, when in a set of three pi bonds only two pi bonds interact with each other by conjugation, while the third one is excluded from interaction. Whereas a normal conjugated system such a ...
*Polyene
In organic chemistry, polyenes are poly- unsaturated, organic compounds that contain at least three alternating double () and single () carbon–carbon bonds. These carbon–carbon double bonds interact in a process known as conjugation, result ...
*Conjugated microporous polymer Conjugated microporous polymers (CMPs) are a sub-class of porous materials that are related to structures such as zeolites, metal-organic frameworks, and covalent organic frameworks, but are amorphous in nature, rather than crystalline. CMPs are a ...
* List of conjugated polymers
*Metallic bond
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It may be des ...
Notes
References
{{chemical bonds Physical organic chemistry *