Carbonate–silicate Cycle on:
[Wikipedia]
[Google]
[Amazon]
 The carbonate–silicate
The carbonate–silicate
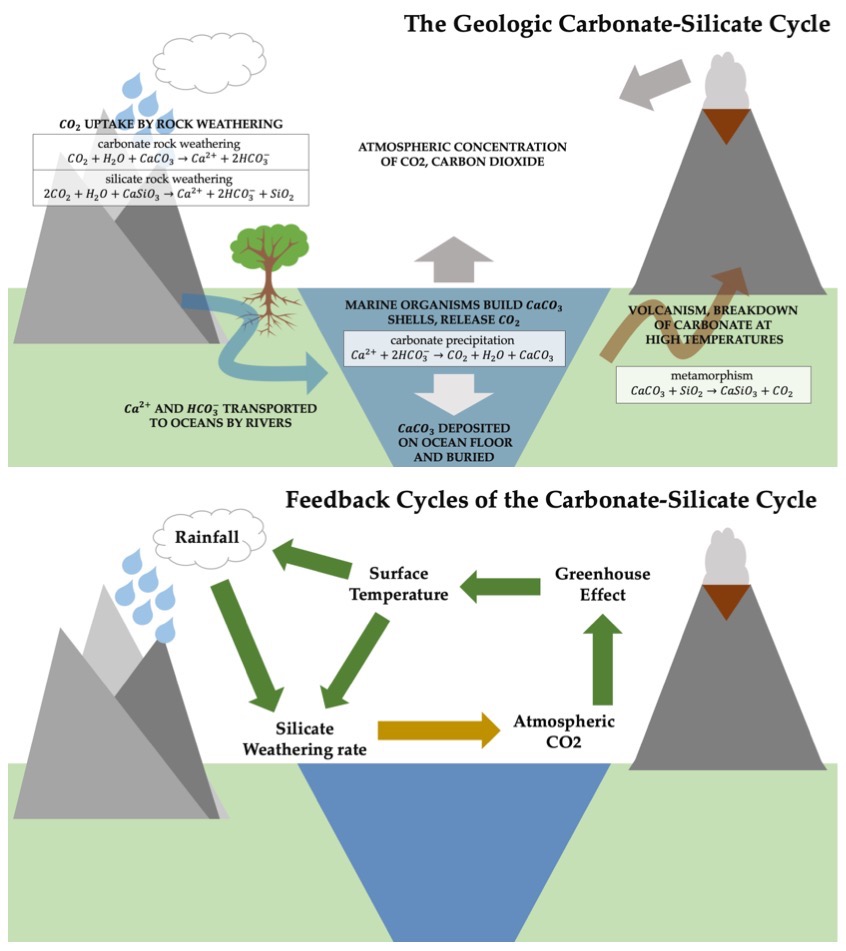 The carbonate-silicate cycle is the primary control on
The carbonate-silicate cycle is the primary control on
2CO2 + H2O + CaSiO3 -> Ca^2+ + 2HCO3- + SiO2
River runoff carries these products to the ocean, where marine calcifying organisms use Ca2+ and HCO3− to build their shells and skeletons, a process called carbonate precipitation:
: Ca^2+ + 2HCO3- -> CaCO3 + CO2 + H2O
Two molecules of CO2 are required for silicate rock weathering; marine calcification releases one molecule back to the atmosphere. The calcium carbonate (CaCO3) contained in shells and skeletons sinks after the marine organism dies and is deposited on the ocean floor.
The final stage of the process involves the movement of the seafloor. At subduction zones, the carbonate sediments are buried and forced back into the mantle. Some carbonate may be carried deep into the mantle where high pressure and temperature conditions allow it to combine metamorphically with SiO2 to form CaSiO3 and CO2, which is released from the interior into the atmosphere via volcanism, thermal vents in the ocean, or soda springs, which are natural springs that contain carbon dioxide gas or soda water:
: CaCO3 + SiO2 -> CaSiO3 + CO2
This final step returns the second CO2 molecule to the atmosphere and closes the inorganic
Understanding the long-term carbon-cycle: weathering of rocks - a vitally important carbon-sink
by John Mason, Skeptical Science {{DEFAULTSORT:Carbonate-silicate cycle Geochemistry History of climate variability and change Climate forcing
 The carbonate–silicate
The carbonate–silicate geochemical cycle In Earth science, a geochemical cycle is the pathway that chemical elements take in the surface and crust of the Earth. The term "geochemical" tells us that geological and chemical factors are all included. The migration of heated and compressed che ...
, also known as the inorganic carbon cycle, describes the long-term transformation of silicate rocks to carbonate
A carbonate is a salt of carbonic acid (H2CO3), characterized by the presence of the carbonate ion, a polyatomic ion with the formula . The word ''carbonate'' may also refer to a carbonate ester, an organic compound containing the carbonate ...
rocks by weathering
Weathering is the deterioration of rocks, soils and minerals as well as wood and artificial materials through contact with water, atmospheric gases, and biological organisms. Weathering occurs ''in situ'' (on site, with little or no movement) ...
and sedimentation
Sedimentation is the deposition of sediments. It takes place when particles in suspension settle out of the fluid in which they are entrained and come to rest against a barrier. This is due to their motion through the fluid in response to the ...
, and the transformation of carbonate rocks back into silicate rocks by metamorphism
Metamorphism is the transformation of existing rock (the protolith) to rock with a different mineral composition or texture. Metamorphism takes place at temperatures in excess of , and often also at elevated pressure or in the presence of ch ...
and volcanism
Volcanism, vulcanism or volcanicity is the phenomenon of eruption of molten rock (magma) onto the surface of the Earth or a solid-surface planet or moon, where lava, pyroclastics, and volcanic gases erupt through a break in the surface called a ...
. Urey, H. C. (1952). The planets: their origin and development. Mrs. Hepsa Ely Silliman Memorial Lectures. Carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
is removed from the atmosphere during burial of weathered minerals and returned to the atmosphere through volcanism
Volcanism, vulcanism or volcanicity is the phenomenon of eruption of molten rock (magma) onto the surface of the Earth or a solid-surface planet or moon, where lava, pyroclastics, and volcanic gases erupt through a break in the surface called a ...
. On million-year time scales, the carbonate-silicate cycle is a key factor in controlling Earth's climate because it regulates carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
levels and therefore global temperature.
However the rate of weathering is sensitive to factors that modulate how much land is exposed. These factors include sea level
Mean sea level (MSL, often shortened to sea level) is an average surface level of one or more among Earth's coastal bodies of water from which heights such as elevation may be measured. The global MSL is a type of vertical datuma standardise ...
, topography
Topography is the study of the forms and features of land surfaces. The topography of an area may refer to the land forms and features themselves, or a description or depiction in maps.
Topography is a field of geoscience and planetary sc ...
, lithology, and vegetation
Vegetation is an assemblage of plant species and the ground cover they provide. It is a general term, without specific reference to particular taxa, life forms, structure, spatial extent, or any other specific botanical or geographic characte ...
changes. Furthermore, these geomorphic and chemical changes have worked in tandem with solar forcing, whether due to orbital changes or stellar evolution, to determine the global surface temperature
In earth science, global surface temperature (GST; sometimes referred to as global mean surface temperature, GMST, or global average surface temperature) is calculated by averaging the temperature at the surface of the sea and air temperature ...
. Additionally, the carbonate-silicate cycle has been considered a possible solution to the faint young Sun paradox
The faint young Sun paradox or faint young Sun problem describes the apparent contradiction between observations of liquid water early in Earth's history and the astrophysical expectation that the Sun's output would be only 70 percent as intense ...
.
Overview of the cycle
 The carbonate-silicate cycle is the primary control on
The carbonate-silicate cycle is the primary control on carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
levels over long timescales. It can be seen as a branch of the carbon cycle
The carbon cycle is the biogeochemical cycle by which carbon is exchanged among the biosphere, pedosphere, geosphere, hydrosphere, and atmosphere of the Earth. Carbon is the main component of biological compounds as well as a major componen ...
, which also includes the organic carbon cycle, in which biological processes convert carbon dioxide and water into organic matter and oxygen via photosynthesis
Photosynthesis is a process used by plants and other organisms to convert light energy into chemical energy that, through cellular respiration, can later be released to fuel the organism's activities. Some of this chemical energy is stored i ...
.
Physical and chemical processes
The inorganic cycle begins with the production of carbonic acid (H2CO3) from rainwater and gaseous carbon dioxide. Due to this process, normal rain has a pH of around 5.6. Carbonic acid is aweak acid
Acid strength is the tendency of an acid, symbolised by the chemical formula HA, to dissociate into a proton, H+, and an anion, A-. The dissociation of a strong acid in solution is effectively complete, except in its most concentrated solutions ...
, but over long timescales, it can dissolve silicate rocks (as well as carbonate rocks). Most of the Earth's crust (and mantle) is composed of silicates. These substances break down into dissolved ions as a result. For example, calcium silicate CaSiO3, or wollastonite
Wollastonite is a calcium inosilicate mineral ( Ca Si O3) that may contain small amounts of iron, magnesium, and manganese substituting for calcium. It is usually white. It forms when impure limestone or dolomite is subjected to high temperature ...
, reacts with carbon dioxide and water to yield a calcium ion, Ca2+, a bicarbonate ion, HCO3−, and dissolved silica. This reaction structure is representative of general silicate weathering of calcium silicate minerals. The chemical pathway is as follows:
:carbon budget
A carbon budget is "the maximum amount of cumulative net global anthropogenic carbon dioxide () emissions that would result in limiting global warming to a given level with a given probability, taking into account the effect of other anthropogen ...
. 99.6% of all carbon on Earth (equating to roughly 108 billion tons of carbon) is sequestered in the longterm rock reservoir. And essentially all carbon has spent time in the form of carbonate. By contrast, only 0.002% of carbon exists in the biosphere.
Feedbacks
Changes to the surface of the planet, such as an absence of volcanoes or higher sea levels, which would reduce the amount of land surface exposed to weathering can change the rates at which different processes in this cycle take place. Over tens to hundreds of millions of years, carbon dioxide levels in the atmosphere may vary due to natural perturbations in the cycle but even more generally, it serves as a critical negative feedback loop between carbon dioxide levels and climate changes. For example, if CO2 builds up in the atmosphere, the greenhouse effect will serve to increase the surface temperature, which will in turn increase the rate of rainfall and silicate weathering, which will remove carbon from the atmosphere. In this way, over long timescales, the carbonate-silicate cycle has a stabilizing effect on the Earth's climate, which is why it has been called the Earth's thermostat.Changes through Earth history
Aspects of the carbonate-silicate cycle have changed throughEarth history
The history of Earth concerns the development of planet Earth from its formation to the present day. Nearly all branches of natural science have contributed to understanding of the main events of Earth's past, characterized by constant geologi ...
as a result of biological evolution
Evolution is change in the heritable characteristics of biological populations over successive generations. These characteristics are the expressions of genes, which are passed on from parent to offspring during reproduction. Variation t ...
and tectonic
Tectonics (; ) are the processes that control the structure and properties of the Earth's crust and its evolution through time. These include the processes of mountain building, the growth and behavior of the strong, old cores of continents ...
changes. Generally, the formation of carbonates has outpaced that of silicates, effectively removing carbon dioxide from the atmosphere. The advent of carbonate biomineralization
Biomineralization, also written biomineralisation, is the process by which living organisms produce minerals, often to harden or stiffen existing tissues. Such tissues are called mineralized tissues. It is an extremely widespread phenomenon; ...
near the Precambrian- Cambrian boundary would have allowed more efficient removal of weathering products from the ocean. Biological processes in soils can significantly increase weathering rates. Plants produce organic acids that increase weathering
Weathering is the deterioration of rocks, soils and minerals as well as wood and artificial materials through contact with water, atmospheric gases, and biological organisms. Weathering occurs ''in situ'' (on site, with little or no movement) ...
. These acids are secreted by root and mycorrhizal fungi
A mycorrhiza (from Greek μύκης ', "fungus", and ῥίζα ', "root"; pl. mycorrhizae, mycorrhiza or mycorrhizas) is a symbiotic association between a fungus and a plant. The term mycorrhiza refers to the role of the fungus in the pla ...
, as well as microbial plant decay. Root respiration and oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a ...
of organic soil matter also produce carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
, which is converted to carbonic acid, which increases weathering.
Tectonics can induce changes in the carbonate-silicate cycle. For example, the uplift of major mountain ranges, such as Himalayas
The Himalayas, or Himalaya (; ; ), is a mountain range in Asia, separating the plains of the Indian subcontinent from the Tibetan Plateau. The range has some of the planet's highest peaks, including the very highest, Mount Everest. Over 10 ...
and the Andes
The Andes, Andes Mountains or Andean Mountains (; ) are the longest continental mountain range in the world, forming a continuous highland along the western edge of South America. The range is long, wide (widest between 18°S – 20°S ...
, is thought to have initiated the Late Cenozoic Ice Age due to increased rates of silicate weathering and draw down of carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
.
Seafloor weather is linked both to solar luminosity and carbon dioxide concentration. However, it presented a challenge to modelers who have tried to relate the rate of outgassing and subduction to the related rates of seafloor change. Proper, uncomplicated proxy data is difficult to attain for such questions. For example, sediment cores, from which scientists can deduce past sea levels, are not ideal because sea levels change as a result of more than just seafloor adjustment. Recent modeling studies have investigated the role of seafloor weathering on the early evolution of life, showing that relatively fast seafloor creation rates worked to draw down carbon dioxide levels to a moderate extent.
Observations of so-called deep time indicate that Earth has a relatively insensitive rock weathering feedback, allowing for large temperature swings. With about twice as much carbon dioxide in the atmosphere, paleoclimate records show that global temperatures reached up to 5 to 6 °C higher than current temperatures. However, other factors such as changes in orbital/solar forcing contribute to global temperature change in the paleo-record.
Human emissions of CO2 have been steadily increasing, and the consequent concentration of CO2 in the Earth system has reached unprecedented levels in a very short amount of time. Excess carbon in the atmosphere that is dissolved in seawater can alter the rates of carbonate-silicate cycle. Dissolved CO2 may react with water to form bicarbonate ions, HCO3−, and hydrogen ions, H+. These hydrogen ions quickly react with carbonate, CO32- to produce more bicarbonate ions and reduce the available carbonate ions, which presents an obstacle to the carbon carbonate precipitation process. Put differently, 30% of excess carbon emitted into the atmosphere is absorbed by the oceans. Higher concentrations of carbon dioxide in the oceans work to push the carbonate precipitation process in the opposite direction (to the left), producing less CaCO3. This process, which harms shell-building organisms, is called ocean acidification
Ocean acidification is the reduction in the pH value of the Earth’s ocean. Between 1751 and 2021, the average pH value of the ocean surface has decreased from approximately 8.25 to 8.14. The root cause of ocean acidification is carbon dioxid ...
.
The cycle on other planets
One should not assume that a carbonate-silicate cycle would appear on allterrestrial planet
A terrestrial planet, telluric planet, or rocky planet, is a planet that is composed primarily of silicate rocks or metals. Within the Solar System, the terrestrial planets accepted by the IAU are the inner planets closest to the Sun: Mercury, ...
s. To begin, the carbonate-silicate cycle requires the presence of a water cycle. It therefore breaks down at the inner edge of the Solar System's habitable zone
In astronomy and astrobiology, the circumstellar habitable zone (CHZ), or simply the habitable zone, is the range of orbits around a star within which a planetary surface can support liquid water given sufficient atmospheric pressure.J. F. Kast ...
. Even if a planet starts out with liquid water on the surface, if it becomes too warm, it will undergo a runaway greenhouse, losing surface water. Without the requisite rainwater, no weathering will occur to produce carbonic acid from gaseous CO2. Furthermore, at the outer edge, CO2 may condense, consequently reducing the greenhouse effect
The greenhouse effect is a process that occurs when energy from a planet's host star goes through the planet's atmosphere and heats the planet's surface, but greenhouse gases in the atmosphere prevent some of the heat from returning directly ...
and reducing the surface temperature. As a result, the atmosphere would collapse into polar caps.
Mars
Mars is the fourth planet from the Sun and the second-smallest planet in the Solar System, only being larger than Mercury. In the English language, Mars is named for the Roman god of war. Mars is a terrestrial planet with a thin at ...
is such a planet. Located at the edge of the solar system's habitable zone, its surface is too cold for liquid water to form without a greenhouse effect. With its thin atmosphere, Mars' mean surface temperature is . In attempting to explain topographical features resembling fluvial channels, despite seemingly insufficient incoming solar radiation, some have suggested that a cycle similar to Earth's carbonate-silicate cycle could have existed – similar to a retreat from Snowball Earth periods. It has been shown using modeling studies that gaseous CO2 and H2O acting as greenhouse gases could not have kept Mars warm during its early history when the sun was fainter because CO2 would condense out into clouds. Even though CO2 clouds do not reflect in the same way that water clouds do on Earth, it could not have had much of a carbonate-silicate cycle in the past.
By contrast, Venus
Venus is the second planet from the Sun. It is sometimes called Earth's "sister" or "twin" planet as it is almost as large and has a similar composition. As an interior planet to Earth, Venus (like Mercury) appears in Earth's sky never f ...
is located at the inner edge of the habitable zone and has a mean surface temperature of . After losing its water by photodissociation and hydrogen escape, Venus stopped removing carbon dioxide from its atmosphere, and began instead to build it up, and experience a runaway greenhouse effect.
On tidally locked exoplanets, the location of the substellar point will dictate the release of carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is trans ...
from the lithosphere.
See also
*Carbon cycle
The carbon cycle is the biogeochemical cycle by which carbon is exchanged among the biosphere, pedosphere, geosphere, hydrosphere, and atmosphere of the Earth. Carbon is the main component of biological compounds as well as a major componen ...
* Reverse weathering
* Daisyworld
* Gaia hypothesis
*Runaway greenhouse effect
A runaway greenhouse effect occurs when a planet's atmosphere contains greenhouse gas in an amount sufficient to block thermal radiation from leaving the planet, preventing the planet from cooling and from having liquid water on its surface. A ...
* Major ice ages
* Snowball earth
*Lake Nyos
Lake Nyos ( ) is a crater lake in the Region of Cameroon, located about northwest of Yaoundé, the capital. Nyos is a deep lake high on the flank of an inactive volcano in the Oku volcanic plain along the Cameroon line of volcanic activity ...
*Ocean acidification
Ocean acidification is the reduction in the pH value of the Earth’s ocean. Between 1751 and 2021, the average pH value of the ocean surface has decreased from approximately 8.25 to 8.14. The root cause of ocean acidification is carbon dioxid ...
*Faint young Sun paradox
The faint young Sun paradox or faint young Sun problem describes the apparent contradiction between observations of liquid water early in Earth's history and the astrophysical expectation that the Sun's output would be only 70 percent as intense ...
References
External links
Understanding the long-term carbon-cycle: weathering of rocks - a vitally important carbon-sink
by John Mason, Skeptical Science {{DEFAULTSORT:Carbonate-silicate cycle Geochemistry History of climate variability and change Climate forcing