UF6 Cylinder Rupture Bw on:
[Wikipedia]
[Google]
[Amazon]
Uranium hexafluoride (), (sometimes called "hex") is an inorganic compound with the formula UF6. Uranium hexafluoride is a volatile white solid that reacts with water, releasing corrosive hydrofluoric acid. The compound reacts mildly with
Image:Uranium-hexafluoride-unit-cell-3D-balls.png,
 As one of the most volatile compounds of uranium, uranium hexafluoride is relatively convenient to process and is used in both of the main uranium enrichment methods, namely
As one of the most volatile compounds of uranium, uranium hexafluoride is relatively convenient to process and is used in both of the main uranium enrichment methods, namely
Uranium
, in: Lester R. Morss, Norman M. Edelstein, Jean Fuger (Hrsg.): ''The Chemistry of the Actinide and Transactinide Elements'', Springer, Dordrecht 2006; , p. 253–698; (p. 530–531, 557–564). * US-Patent 2535572
26. December 1950. * US-Patent 5723837
3. March 1998.
Uranium Hexafluoride (UF6) – Physical and chemical properties of UF6, and its use in uranium processing – Uranium Hexafluoride and Its Properties
Import of Western depleted uranium hexafluoride (uranium tails) to Russia
ead link 30 June 2017
Uranium Hexafluoride in www.webelements.com
{{fluorine compounds Uranium(VI) compounds Hexafluorides Actinide halides Octahedral compounds Nuclear materials
aluminium
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. I ...
, forming a thin surface layer of AlF3 that resists any further reaction from the compound. UF6 is used in the process of enriching uranium, which produces fuel for nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s and nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or a combination of fission and fusion reactions ( thermonuclear bomb), producing a nuclear explosion. Both bom ...
s.
Preparation
Milled uranium ore—U3O8 or "yellowcake
Yellowcake (also called urania) is a type of uranium concentrate powder obtained from leach solutions, in an intermediate step in the processing of uranium ores. It is a step in the processing of uranium after it has been mined but before f ...
"—is dissolved in nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available nitri ...
, yielding a solution of uranyl nitrate
Uranyl nitrate is a water-soluble yellow uranium salt with the formula . The hexa-, tri-, and dihydrates are known. The compound is mainly of interest because it is an intermediate in the preparation of nuclear fuels.
Uranyl nitrate can be prepa ...
UO2(NO3)2. Pure uranyl nitrate is obtained by solvent extraction
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for ...
, then treated with ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous wa ...
to produce ammonium diuranate ("ADU", (NH4)2U2O7). Reduction with hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic ...
gives UO2, which is converted with hydrofluoric acid (HF) to uranium tetrafluoride
Uranium tetrafluoride is the inorganic compound with the formula UF4. It is a green solid with an insignificant vapor pressure and low solubility in water. Uranium in its tetravalent ( uranous) state is important in various technological process ...
, UF4. Oxidation with fluorine yields UF6.
During nuclear reprocessing, uranium is reacted with chlorine trifluoride
Chlorine trifluoride is an interhalogen compound with the formula ClF3. This colorless, poisonous, corrosive, and extremely reactive gas condenses to a pale-greenish yellow liquid, the form in which it is most often sold (pressurized at room temp ...
to give UF6:
:U + 2 ClF3 → UF6 + Cl2
Properties
Physical properties
Atatmospheric pressure
Atmospheric pressure, also known as barometric pressure (after the barometer), is the pressure within the atmosphere of Earth. The standard atmosphere (symbol: atm) is a unit of pressure defined as , which is equivalent to 1013.25 millibars, ...
, it sublimes at 56.5 °C.
The solid state structure was determined by neutron diffraction
Neutron diffraction or elastic neutron scattering is the application of neutron scattering to the determination of the atomic and/or magnetic structure of a material. A sample to be examined is placed in a beam of thermal or cold neutrons to o ...
at 77 K and 293 K.
Ball-and-stick model
In chemistry, the ball-and-stick model is a molecular model of a chemical substance which displays both the three-dimensional position of the atoms and the bonds between them. The atoms are typically represented by spheres, connected by rods ...
of the unit cell of uranium hexafluorideJ. C. Taylor, P. W. Wilson, J. W. Kelly: „The structures of fluorides. I. Deviations from ideal symmetry in the structure of crystalline UF6: a neutron diffraction analysis", '' Acta Crystallogr.'', 1973, ''B29'', p. 7–12; .
Image:Uranium hexafluoride dimensions.svg, Bond lengths and angles of gaseous uranium hexafluoride
Chemical properties
It has been shown that uranium hexafluoride is anoxidant
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
and a Lewis acid that is able to bind to fluoride; for instance, the reaction of copper(II) fluoride with uranium hexafluoride in acetonitrile is reported to form copper(II) heptafluorouranate(VI), Cu(UF7)2.
Polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part")
is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic a ...
ic uranium(VI) fluorides containing organic cations have been isolated and characterized by X-ray diffraction.
Application in the fuel cycle
 As one of the most volatile compounds of uranium, uranium hexafluoride is relatively convenient to process and is used in both of the main uranium enrichment methods, namely
As one of the most volatile compounds of uranium, uranium hexafluoride is relatively convenient to process and is used in both of the main uranium enrichment methods, namely gaseous diffusion
Gaseous diffusion is a technology used to produce enriched uranium by forcing gaseous uranium hexafluoride (UF6) through semipermeable membranes. This produces a slight separation between the molecules containing uranium-235 (235U) and uranium-2 ...
and the gas centrifuge
A gas centrifuge is a device that performs isotope separation of gases. A centrifuge relies on the principles of centrifugal force accelerating molecules so that particles of different masses are physically separated in a gradient along the radiu ...
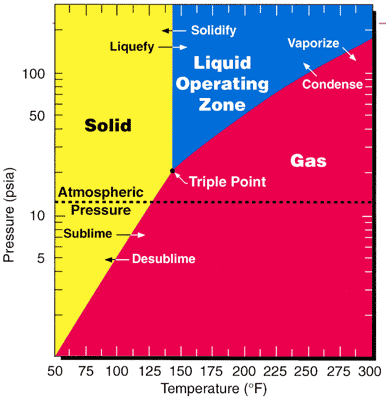
method. Since the triple point
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.. It is that temperature and pressure at which the sub ...
of UF6—64 °C(147 °F; 337 K) and 152 kPa (22 psi; 1.5 atm)— is close to ambient conditions, phase transitions can be achieved in a processing facility with little thermodynamic work
In thermodynamics, work is one of the principal processes by which a thermodynamic system can interact with its surroundings and exchange energy. An exchange of energy is facilitated by a mechanism through which the system can spontaneously exer ...
.
Fluorine has only a single naturally occurring stable isotope, so isotopologue
In chemistry, isotopologues are molecules that differ only in their isotopic composition. They have the same chemical formula and bonding arrangement of atoms, but at least one atom has a different number of neutrons than the parent.
An exampl ...
s of UF6 differ in their molecular weight based solely on the uranium isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers (mass numb ...
present. This difference is the basis for the physical separation of isotopes in enrichment.
All the other uranium fluorides are nonvolatile solids that are coordination polymers.
The conversion factor for the 238U isotopologue of UF6 ("hex") to "U mass" is .676.
Gaseous diffusion requires about 60 times as much energy as the gas centrifuge process: gaseous diffusion-produced nuclear fuel produces 25 times more energy than is used in the diffusion process, while centrifuge-produced fuel produces 1,500 times more energy than is used in the centrifuge process.
In addition to its use in enrichment, uranium hexafluoride has been used in an advanced reprocessing method (fluoride volatility
Fluoride volatility is the tendency of highly fluorinated molecules to vaporize at comparatively low temperatures. Heptafluorides, hexafluorides and pentafluorides have much lower boiling points than the lower- valence fluorides. Most difluor ...
), which was developed in the Czech Republic
The Czech Republic, or simply Czechia, is a landlocked country in Central Europe. Historically known as Bohemia, it is bordered by Austria to the south, Germany to the west, Poland to the northeast, and Slovakia to the southeast. The ...
. In this process, spent nuclear fuel
Spent nuclear fuel, occasionally called used nuclear fuel, is nuclear fuel that has been irradiated in a nuclear reactor (usually at a nuclear power plant). It is no longer useful in sustaining a nuclear reaction in an ordinary thermal reactor and ...
is treated with fluorine gas to transform the oxides or elemental metals into a mixture of fluorides. This mixture is then distilled to separate the different classes of material. Some fission products form nonvolatile fluorides which remain as solids and can then either be prepared for storage as nuclear waste or further processed either by solvation
Solvation (or dissolution) describes the interaction of a solvent with dissolved molecules. Both ionized and uncharged molecules interact strongly with a solvent, and the strength and nature of this interaction influence many properties of the ...
-based methods or electrochemically.
Uranium enrichment produces large quantities of depleted uranium hexafluoride, or DUF6, as a waste product. The long-term storage of DUF6 presents environmental, health, and safety risks because of its chemical instability. When UF6 is exposed to moist air, it reacts with the water in the air to produce UO2F2 (uranyl fluoride
Uranyl fluoride is the inorganic compound with the formula UO2F2. As shown by x-ray crystallography, the uranyl (UO22+) centers are complemented by six fluoride ligands.
This salt is very soluble in water as well as hygroscopic
Hygroscopy is t ...
) and HF ( hydrogen fluoride) both of which are highly corrosive and toxic. In 2005, 686,500 tonnes of DUF6 was housed in 57,122 storage cylinders located near Portsmouth, Ohio
Portsmouth is a city in and the county seat of Scioto County, Ohio, United States. Located in southern Ohio south of Chillicothe, it lies on the north bank of the Ohio River, across from Kentucky, just east of the mouth of the Scioto River. ...
; Oak Ridge, Tennessee; and Paducah, Kentucky
Paducah ( ) is a home rule-class city in and the county seat of McCracken County, Kentucky. The largest city in the Jackson Purchase region, it is located at the confluence of the Tennessee and the Ohio rivers, halfway between St. Louis, Miss ...
. Storage cylinders must be regularly inspected for signs of corrosion and leaks. The estimated lifetime of the steel cylinders is measured in decades.
There have been several accidents involving uranium hexafluoride in the US, including a cylinder-filling accident and material release at the Sequoyah Fuels Corporation in 1986. The U.S. government has been converting DUF6 to solid uranium oxides for disposal. Such disposal of the entire DUF6 stockpile could cost anywhere from $15 million to $450 million.
References
Further reading
* '' Gmelins Handbuch der anorganischen Chemie'', System Nr. 55, Uran, Teil A, p. 121–123. * ''Gmelins Handbuch der anorganischen Chemie'', System Nr. 55, Uran, Teil C 8, p. 71–163. * R. DeWitt: ''Uranium hexafluoride: A survey of the physico-chemical properties'', Technical report, GAT-280; Goodyear Atomic Corp., Portsmouth, Ohio; 12. August 1960; . * Ingmar Grenthe, Janusz Drożdżynński, Takeo Fujino, Edgar C. Buck, Thomas E. Albrecht-Schmitt, Stephen F. WolfUranium
, in: Lester R. Morss, Norman M. Edelstein, Jean Fuger (Hrsg.): ''The Chemistry of the Actinide and Transactinide Elements'', Springer, Dordrecht 2006; , p. 253–698; (p. 530–531, 557–564). * US-Patent 2535572
26. December 1950. * US-Patent 5723837
3. March 1998.
External links
* Simon Cotton (Uppingham School, Rutland, UK)Uranium Hexafluoride (UF6) – Physical and chemical properties of UF6, and its use in uranium processing – Uranium Hexafluoride and Its Properties
Import of Western depleted uranium hexafluoride (uranium tails) to Russia
ead link 30 June 2017
Uranium Hexafluoride in www.webelements.com
{{fluorine compounds Uranium(VI) compounds Hexafluorides Actinide halides Octahedral compounds Nuclear materials