Trioxygen difluoride on:
[Wikipedia]
[Google]
[Amazon]
 Oxygen fluorides are compounds of elements
Oxygen fluorides are compounds of elements  Oxygen fluorides are strong
Oxygen fluorides are strong
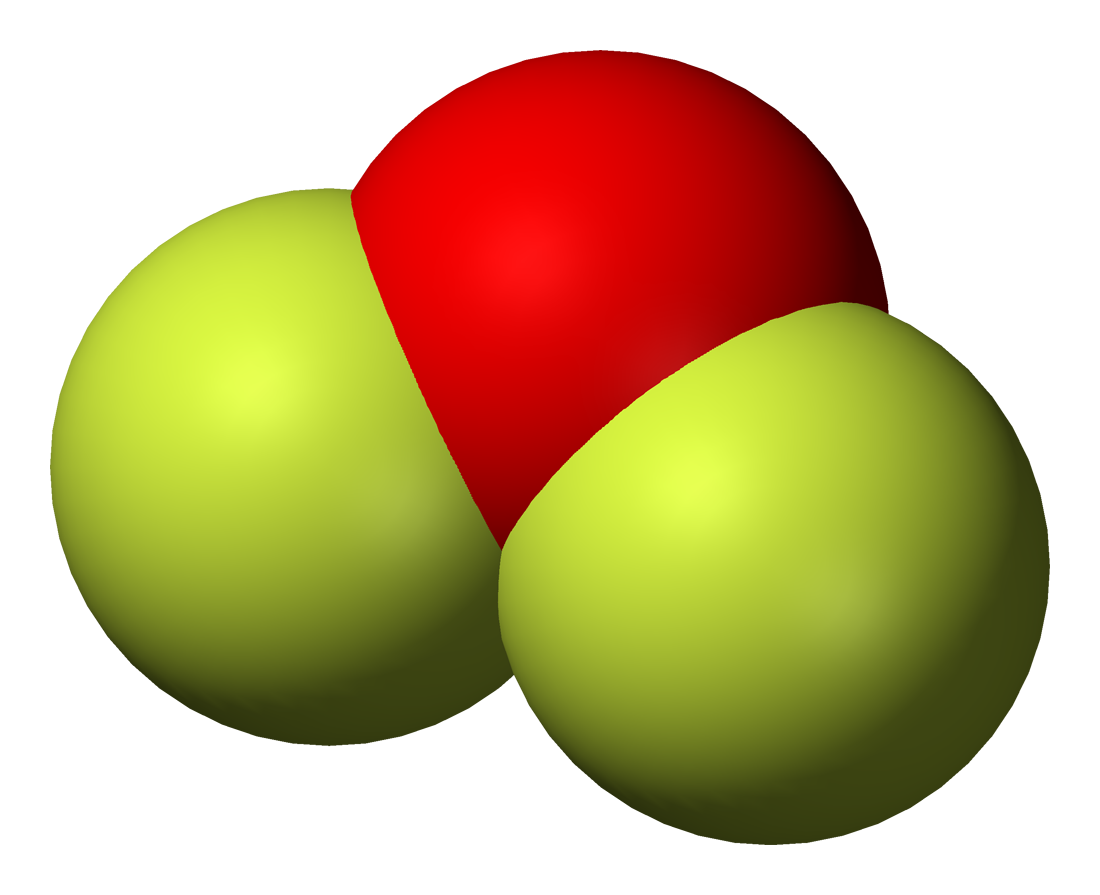 A common preparative method involves fluorination of sodium hydroxide:
:
is a colorless gas at room temperature and a yellow liquid below 128 K. Oxygen difluoride has an irritating odor and is poisonous. It reacts quantitatively with aqueous haloacids to give free halogens:
:
It can also displace halogens from their salts. It is both an effective
A common preparative method involves fluorination of sodium hydroxide:
:
is a colorless gas at room temperature and a yellow liquid below 128 K. Oxygen difluoride has an irritating odor and is poisonous. It reacts quantitatively with aqueous haloacids to give free halogens:
:
It can also displace halogens from their salts. It is both an effective
 precipitates as a brown solid upon the
precipitates as a brown solid upon the
National Pollutant Inventory - Fluoride and compounds fact sheetOxygen Fluoride PubChem
{{fluorides Oxygen compounds Fluorides Nonmetal halides Chalcohalides
oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements as ...
and fluorine with the general formula , where ''n'' = 1 to 6. Many different oxygen fluorides are known:
*oxygen difluoride
Oxygen difluoride is a chemical compound with the formula . As predicted by VSEPR theory, the molecule adopts a "bent" molecular geometry. It is strong oxidizer and has attracted attention in rocketry for this reason. With a boiling point of -144.7 ...
()
*dioxygen difluoride
Dioxygen difluoride is a compound of fluorine and oxygen with the molecular formula O2F2. It can exist as an orange-colored solid which melts into a red liquid at . It is an extremely strong oxidant and decomposes into oxygen and fluorine even ...
()
* trioxygen difluoride or ozone difluoride ()
* tetraoxygen difluoride ()
* pentaoxygen difluoride ()
* hexaoxygen difluoride ()
*dioxygen monofluoride or fluoroperoxyl ()
oxidizing agent
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
s with high energy and can release their energy either instantaneously or at a controlled rate. Thus, these compounds attracted much attention as potential fuels in jet propulsion systems.
Synthesis
Here are some synthesis methods and reactions of the three most common oxygen fluorides – oxygen difluoride (), dioxygen difluoride () and ozone difluoride ().Oxygen difluoride ()
 A common preparative method involves fluorination of sodium hydroxide:
:
is a colorless gas at room temperature and a yellow liquid below 128 K. Oxygen difluoride has an irritating odor and is poisonous. It reacts quantitatively with aqueous haloacids to give free halogens:
:
It can also displace halogens from their salts. It is both an effective
A common preparative method involves fluorination of sodium hydroxide:
:
is a colorless gas at room temperature and a yellow liquid below 128 K. Oxygen difluoride has an irritating odor and is poisonous. It reacts quantitatively with aqueous haloacids to give free halogens:
:
It can also displace halogens from their salts. It is both an effective fluorinating agent
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polyme ...
and a strong oxidizing agent
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
. When reacted with unsaturated nitrogen fluorides with electrical discharge, it results in the formation of nitrogen trifluoride, oxide fluorides and other oxides.
Dioxygen difluoride ()
 precipitates as a brown solid upon the
precipitates as a brown solid upon the UV irradiation
Ultraviolet germicidal irradiation (UVGI) is a disinfection method that uses short-wavelength ultraviolet (ultraviolet C or UV-C) light to kill or inactivate microorganisms by destroying nucleic acids and disrupting their DNA, leaving them unabl ...
of a mixture of liquid and at −196 °C. It also only appears to be stable below −160 °C.Solomon, Irvine J. ''Research on Chemistry of and ''. No. IITRI-C227-6. IIT RESEARCH INST CHICAGO IL, 1964. The general method of preparation of many oxygen fluorides is a gas-phase electric discharge in cold containers including .
: (electric discharge, 183 °C)
It is typically an orange-yellow solid which rapidly decomposes to and close to its normal boiling point of about 216 K.
reacts violently with red phosphorus
Elemental phosphorus can exist in several allotropes, the most common of which are white and red solids. Solid violet and black allotropes are also known. Gaseous phosphorus exists as diphosphorus and atomic phosphorus.
White phosphorus
White ...
, even at −196 °C. Explosions can also occur if Freon-13 is used to moderate the reaction.
Trioxygen difluoride or ozone difluoride ()
is a viscous, blood-red liquid. It remains liquid at 90 K and so can be differentiated from which has a melting point of about 109 K. Like the other oxygen fluorides, isendothermic
In thermochemistry, an endothermic process () is any thermodynamic process with an increase in the enthalpy (or internal energy ) of the system.Oxtoby, D. W; Gillis, H.P., Butler, L. J. (2015).''Principle of Modern Chemistry'', Brooks Cole. ...
and decomposes at about 115 K with the evolution of heat, which is given by the following reaction:
:
is safer to work with than ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lo ...
, and can be evaporated, or thermally decomposed, or exposed to electric sparks, without any explosions. But on contact with organic matter or oxidizable compounds, it can detonate or explode. Thus, the addition of even one drop of ozone difluoride to solid anhydrous ammonia
Ammonia is an inorganic compound of nitrogen and hydrogen with the formula . A stable binary hydride, and the simplest pnictogen hydride, ammonia is a colourless gas with a distinct pungent smell. Biologically, it is a common nitrogenous wa ...
will result in a mild explosion, when they are both at 90 K each.
Fluoroperoxyl
Fluoroperoxyl is a molecule such as O–O–F, whosechemical formula
In chemistry, a chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbol ...
is and is stable only at low temperature. It has been reported to be produced from atomic fluorine and dioxygen.
:
General preparation of polyoxygen difluorides
Effects on ozone
Oxygen- and fluorine-containing radicals like and OF occur in the atmosphere. These along with other halogen radicals have been implicated in the destruction of ozone in the atmosphere. However, the oxygen monofluoride radicals are assumed to not play as big a role in the ozone depletion because free fluorine atoms in the atmosphere are believed to react withmethane
Methane ( , ) is a chemical compound with the chemical formula (one carbon atom bonded to four hydrogen atoms). It is a group-14 hydride, the simplest alkane, and the main constituent of natural gas. The relative abundance of methane on Ea ...
to produce hydrofluoric acid which precipitates in rain. This decreases the availability of free fluorine atoms for oxygen atoms to react with and destroy ozone molecules.
:
:
Net reaction:
:
Hypergolic propellant
Despite the low solubility of in liquid oxygen, it has been shown to behypergolic
A hypergolic propellant is a rocket propellant combination used in a rocket engine, whose components spontaneously ignite when they come into contact with each other.
The two propellant components usually consist of a fuel and an oxidizer. T ...
with most rocket propellant fuels. The mechanism involves the boiling off oxygen from the solution containing , making it more reactive to have a spontaneous reaction with the rocket fuel. The degree of reactivity is also dependent on the type of fuel used.
See also
*Bromine oxide
Bromine can form several different unstable oxides:
* Dibromine monoxide (Br2O)
* Bromine dioxide (BrO2)
* Dibromine trioxide (Br2O3)
* Dibromine pentoxide (Br2O5)
* Tribromine octoxide (Br3O8)
Also, a number of ions are bromine oxides:
* Hy ...
* Chlorine oxide
Chlorine and oxygen can bond in many ways:
* chlorine monoxide, , chlorine (II) oxide
* chlorine peroxide, , dimer of chlorine (II) oxide
* chlorine dioxide, , chlorine (IV) oxide
* chloroperoxyl,
*chlorine trioxide, ClO3, chlorine (VI) oxide
* ...
* Iodine oxide
Iodine oxides are chemical compounds of oxygen and iodine. Iodine has only two stable oxides which are isolatable in bulk, iodine tetroxide and iodine pentoxide, but a number of other oxides are formed in trace quantities or have been hypothesize ...
* Ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lo ...
References
External links
National Pollutant Inventory - Fluoride and compounds fact sheet
{{fluorides Oxygen compounds Fluorides Nonmetal halides Chalcohalides