Sharpless Epoxidation on:
[Wikipedia]
[Google]
[Amazon]
The Sharpless epoxidation reaction is an  2,3-Epoxyalcohols can be converted into
2,3-Epoxyalcohols can be converted into
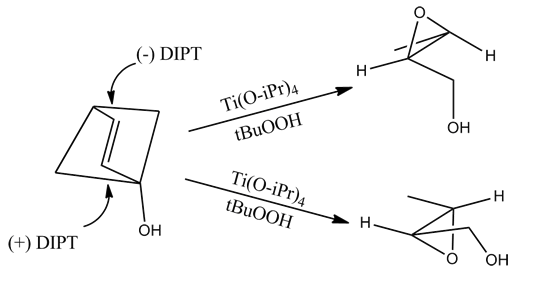 However, this method incorrectly predicts the product of allylic 1,2-diols.
However, this method incorrectly predicts the product of allylic 1,2-diols.


 As one of the few highly enantioselective reactions during its time, many manipulations of the 2,3-epoxyalcohols have been developed.
The Sharpless epoxidation has been used for the total synthesis of various
As one of the few highly enantioselective reactions during its time, many manipulations of the 2,3-epoxyalcohols have been developed.
The Sharpless epoxidation has been used for the total synthesis of various
Sharpless Asymmetric Epoxidation Reaction
{{DEFAULTSORT:Sharpless Epoxidation Epoxidation reactions Organic redox reactions Name reactions Epoxides Catalysis
enantioselective
In chemistry, an enantiomer ( /ɪˈnænti.əmər, ɛ-, -oʊ-/ ''ih-NAN-tee-ə-mər''; from Ancient Greek ἐνάντιος ''(enántios)'' 'opposite', and μέρος ''(méros)'' 'part') – also called optical isomer, antipode, or optical anti ...
chemical reaction
A chemical reaction is a process that leads to the IUPAC nomenclature for organic transformations, chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the pos ...
to prepare 2,3-epoxyalcohols from primary and secondary allylic alcohol
Allyl alcohol (IUPAC name: prop-2-en-1-ol) is an organic compound with the structural formula . Like many alcohols, it is a water-soluble, colourless liquid. It is more toxic than typical small alcohols. Allyl alcohol is used as a raw material ...
s. The oxidizing agent is ''tert''-butyl hydroperoxide. The method relies on a catalyst formed from titanium tetra(isopropoxide) and diethyl tartrate
Diethyl tartrate is an organic compound with the formula (HOCHCO2Et)2 (Et = ethyl). Three stereoisomers exist, R,R-, S,S-, and R,S (=S,R-). They are the ethyl esters of the respective R,R-, S,S-, and R,S-tartaric acids. The R,R- and S,S- isomers ...
.
diol
A diol is a chemical compound containing two hydroxyl groups ( groups). An aliphatic diol is also called a glycol. This pairing of functional groups is pervasive, and many subcategories have been identified.
The most common industrial diol is e ...
s, aminoalcohols, and ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be c ...
s. The reactants for the Sharpless epoxidation are commercially available and relatively inexpensive.
K. Barry Sharpless was awarded the 2001 Nobel Prize in Chemistry for this and related work on asymmetric oxidations. The prize was shared with William S. Knowles and Ryōji Noyori
is a Japanese chemist. He won the Nobel Prize in Chemistry in 2001, Noyori shared a half of the prize with William S. Knowles for the study of chirally catalyzed hydrogenations; the second half of the prize went to K. Barry Sharpless for his st ...
.
Catalyst
5–10 mol% of the catalyst is typical. The presence of 3Åmolecular sieve
A molecular sieve is a material with pores (very small holes) of uniform size. These pore diameters are similar in size to small molecules, and thus large molecules cannot enter or be adsorbed, while smaller molecules can. As a mixture of molecu ...
s (3Å MS) is necessary. The structure of the catalyst is uncertain although it is thought to be a dimer of [].
Selectivity
The epoxidation of allylic alcohols is a well-utilized conversion in fine chemical synthesis. The chirality of the product of a Sharpless epoxidation is sometimes predicted with the followingmnemonic
A mnemonic ( ) device, or memory device, is any learning technique that aids information retention or retrieval (remembering) in the human memory for better understanding.
Mnemonics make use of elaborative encoding, retrieval cues, and imag ...
. A rectangle is drawn around the double bond in the same plane as the carbons of the double bond (the ''xy-plane''), with the allylic alcohol in the bottom right corner and the other substituents in their appropriate corners. In this orientation, the (−) diester tartrate preferentially interacts with the top half of the molecule, and the (+) diester tartrate preferentially interacts with the bottom half of the molecule. This model seems to be valid despite substitution on the olefin. Selectivity decreases with larger R1, but increases with larger R2 and R3 (see introduction).
 However, this method incorrectly predicts the product of allylic 1,2-diols.
However, this method incorrectly predicts the product of allylic 1,2-diols.

Kinetic resolution
The Sharpless epoxidation can also givekinetic resolution
In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resul ...
of a racemic mixture of secondary 2,3-epoxyalcohols. While the yield of a kinetic resolution process cannot be higher than 50%, the enantiomeric excess
In stereochemistry, enantiomeric excess (ee) is a measurement of purity used for chiral substances. It reflects the degree to which a sample contains one enantiomer in greater amounts than the other. A racemic mixture has an ee of 0%, while a sin ...
approaches 100% in some reactions.

Synthetic utility
The Sharpless epoxidation is viable with a large range of primary and secondary alkenic alcohols. Furthermore, with the exception noted above, a given dialkyl tartrate will preferentially add to the same face independent of the substitution on thealkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
.To demonstrate the synthetic utility of the Sharpless epoxidation, the Sharpless group created synthetic intermediates of various natural products: methymycin, erythromycin
Erythromycin is an antibiotic used for the treatment of a number of bacterial infections. This includes respiratory tract infections, skin infections, chlamydia infections, pelvic inflammatory disease, and syphilis. It may also be used duri ...
, leukotriene
Leukotrienes are a family of eicosanoid inflammatory mediators produced in leukocytes by the oxidation of arachidonic acid (AA) and the essential fatty acid eicosapentaenoic acid (EPA) by the enzyme arachidonate 5-lipoxygenase.
Leukotrienes ...
C-1, and (+)- disparlure.
 As one of the few highly enantioselective reactions during its time, many manipulations of the 2,3-epoxyalcohols have been developed.
The Sharpless epoxidation has been used for the total synthesis of various
As one of the few highly enantioselective reactions during its time, many manipulations of the 2,3-epoxyalcohols have been developed.
The Sharpless epoxidation has been used for the total synthesis of various saccharides
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or may ...
, terpenes
Terpenes () are a class of natural products consisting of compounds with the formula (C5H8)n for n > 1. Comprising more than 30,000 compounds, these unsaturated hydrocarbons are produced predominantly by plants, particularly conifers. Terpenes ar ...
, leukotrienes
Leukotrienes are a family of eicosanoid inflammatory mediators produced in leukocytes by the oxidation of arachidonic acid (AA) and the essential fatty acid eicosapentaenoic acid (EPA) by the enzyme arachidonate 5-lipoxygenase.
Leukotrienes ...
, pheromones
A pheromone () is a secreted or excreted chemical factor that triggers a social response in members of the same species. Pheromones are chemicals capable of acting like hormones outside the body of the secreting individual, to affect the behavio ...
, and antibiotics
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention o ...
.
The main drawback of this protocol is the necessity of the presence of an allylic alcohol
Allyl alcohol (IUPAC name: prop-2-en-1-ol) is an organic compound with the structural formula . Like many alcohols, it is a water-soluble, colourless liquid. It is more toxic than typical small alcohols. Allyl alcohol is used as a raw material ...
. The Jacobsen epoxidation
The Jacobsen epoxidation, sometimes also referred to as Jacobsen-Katsuki epoxidation is a chemical reaction which allows enantioselective epoxidation of unfunctionalized alkyl- and aryl- substituted alkenes. It is complementary to the Sharpless e ...
, an alternative method to enantioselectively oxidise alkenes, overcomes this issue and tolerates a wider array of functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the rest ...
s.
References of historic interest
* *See also
*Asymmetric catalytic oxidation Asymmetric catalytic oxidation is a technique of oxidizing various substrates to give an enantio-enriched product using a catalyst. Typically, but not necessarily, asymmetry is induced by the chirality of the catalyst. Typically, but again not ne ...
* Juliá–Colonna epoxidation The Juliá–Colonna epoxidation is an asymmetric poly-leucine catalyzed nucleophilic epoxidation of electron deficient olefins in a triphasic system. The reaction was reported by Sebastian Juliá at the Chemical Institute of Sarriá in 1980, with ...
References
External links
Sharpless Asymmetric Epoxidation Reaction
{{DEFAULTSORT:Sharpless Epoxidation Epoxidation reactions Organic redox reactions Name reactions Epoxides Catalysis