sucrase-isomaltase on:
[Wikipedia]
[Google]
[Amazon]
Oligo-1,6-glucosidase (EC 3.2.1.10, sucrase-isomaltase, SI; systematic name oligosaccharide 6-α-glucohydrolase) is a glucosidase
 Sucrase-isomaltase consists of two enzymatic subunits:
Sucrase-isomaltase consists of two enzymatic subunits:
Structure and evolution of the mammalian maltase-glucoamylase and sucrase-isomaltase
* {{Portal bar, Biology, border=no EC 3.2.1
enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different molecule ...
located on the brush border of the small intestine, which catalyses the following reaction:
:Hydrolysis of (1→6)-α-D-glucosidic linkages in some oligosaccharides produced from starch and glycogen by (α-amylase), and in isomaltose
It is a dual-function enzyme with two GH31 domains, one serving as the isomaltase, the other as a sucrose alpha-glucosidase. It has preferential expression in the apical membranes of enterocytes. The enzyme’s purpose is to digest dietary carbohydrates
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may or may ...
such as starch
Starch or amylum is a polymeric carbohydrate consisting of numerous glucose units joined by glycosidic bonds. This polysaccharide is produced by most green plants for energy storage. Worldwide, it is the most common carbohydrate in human diets ...
, sucrose and isomaltose. By further processing the broken-down products, energy in the form of ATP can be generated.Berg, J. M. et al. ''Biochemistry'', 7th Ed. W.H. Freeman and Company: New York, 2012.
Structure
 Sucrase-isomaltase consists of two enzymatic subunits:
Sucrase-isomaltase consists of two enzymatic subunits: sucrase Sucrase is a digestive enzyme that catalyzes the hydrolysis of sucrose to its subunits fructose and glucose. One form, sucrase-isomaltase, is secreted in the small intestine on the brush border. The sucrase enzyme invertase, which occurs more com ...
and isomaltase. The subunits originate from a polypeptide precursor, pro-SI. By heterodimerizing the two subunits, the sucrase-isomaltase complex is formed. The enzyme is anchored in the intestinal brush border membrane by a hydrophobic segment located near the N-terminal of the isomaltase subunit. Before the enzyme is anchored to the membrane, pro-SI is mannose-rich and glycosylated; it moves from the ER to the Golgi, where it becomes a protein complex that is N- and O- glycosylated. The O-linked glycosylation
''O''-linked glycosylation is the attachment of a sugar molecule to the oxygen atom of serine (Ser) or threonine (Thr) residues in a protein. ''O''-glycosylation is a post-translational modification that occurs after the protein has been synthes ...
is necessary to target the protein to the apical membrane. In addition, there is a segment that is both O-linked glycosylated and Ser/Thr-rich. A similarly-arranged enzyme is the maltase-glucoamylase
Maltase-glucoamylase, intestinal is an enzyme that in humans is encoded by the ''MGAM'' gene.
Maltase-glucoamylase is an alpha-glucosidase digestive enzyme. It consists of two subunits with differing substrate specificity. Recombinant enzyme st ...
, also a member of GH31.
Sucrase-isomaltase is composed of duplicated catalytic domains, N- and C-terminal. Each domain displays overlapping specificities. Scientists have discovered the crystal structure for N-terminal human sucrase-isomaltase (ntSI) in apo form to 3.2 Å and in complex with the inhibitor kotalanol to 2.15 Å resolution. Sucrase-isomaltase’s mechanism results in a net retention of configuration at the anomeric center.
The crystal structure shows that sucrase-isomaltase exists as a monomer
In chemistry, a monomer ( ; ''mono-'', "one" + '' -mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or three-dimensional network in a process called polymerization.
Classification
...
. The researchers claim that the observance of SI dimers is dependent on experimental conditions. ntSI’s four monomers, A, B, C, and D are included in the crystal asymmetric unit and have identical active sites. The active site is composed of a shallow-substrate binding pocket including -1 and +1 subsites. The non-reducing end of substrates binds to the pocket. While the non-reducing sugar ring has interactions with the buried -1 subsite, the reducing ring has interactions with the surface exposed +1 subsite.
The interactions between the active site of sucrase-isomaltase and the following compounds have been identified:
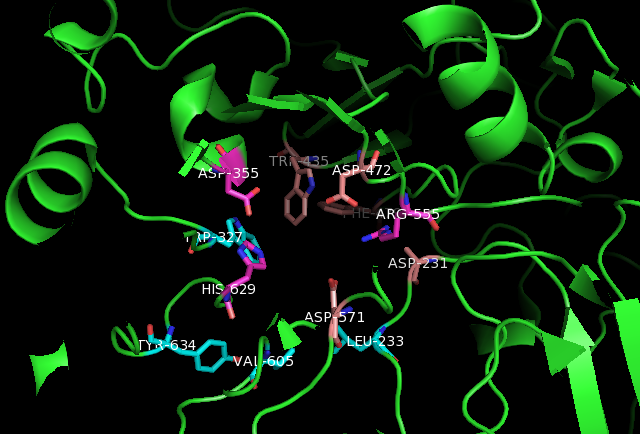
* Man2GlcNAc2 glycan: Within the active site, Man2GlcNAc2 hydrogen bonds
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
with hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydrox ...
side chains of Asp231 and Asp571. Furthermore, hydrophobic interactions with Leu233, Trp327, Trp435, Phe479, Val605, and Tyr634 provide additional stabilization for Man2GlcNAc2.
* Kotalonal, the inhibitor: It interacts with the catalytic nucleophile
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they a ...
Asp472 and acid base catalyst Asp571. In addition, ntSI residues His629, Asp355, Arg555, Asp231, Trp435, and Phe479 bind to the substrate.
Currently, there are no crystal structures of ntSI in complex with an α-1,6-linked substrate or inhibitor analogue. In order to predict isomaltose binding in sucrase-isomaltase structure, a model was produced by hand. Within the -1 subsite, isomaltose’s non-reducing glucose ring was aligned to that of acarbose.
Not only has the structure of human sucrase-isomaltase been studied, but also sucrase-isomaltase’s structure in sea lions and pigs have also been analyzed.
Disease relevance
A deficiency is responsible for sucrose intolerance. Congenital sucrase-isomaltase deficiency (CSID), also called genetic sucrase-isomaltase deficiency (GSID), and sucrose intolerance, is a genetic, intestinal disorder that is caused by a reduction or absence ofsucrase Sucrase is a digestive enzyme that catalyzes the hydrolysis of sucrose to its subunits fructose and glucose. One form, sucrase-isomaltase, is secreted in the small intestine on the brush border. The sucrase enzyme invertase, which occurs more com ...
and isomaltase Explanations for GSID include:
* Mutations C1229Y and F1745C, which are present in the sucrase domain of SI, block SI path to anchor in the cell’s aprical membrane but does not impact protein folding or isomaltase activity.
* Substitution of a cysteine by an arginine at amino acid residue 635 in the isomaltase subunit of SI was present in the cDNA encoding for a patient with CSID. SIC635R had an altered folding pattern, which influenced the sorting profile and increased the turnover rate.
* A factor that can be attributed to congenital sucrase-isomaltase deficiency is the retention of SI in the cis-Golgi. This inability to transport is a result of a glutamine to proline substitution at amino acid residue 1098 of the sucrase subunit.
Furthermore, a relationship between mutations in sucrase-isomaltase and chronic lymphocytic leukemia
Chronic lymphocytic leukemia (CLL) is a type of cancer in which the bone marrow makes too many lymphocytes (a type of white blood cell). Early on, there are typically no symptoms. Later, non-painful lymph node swelling, feeling tired, fever, n ...
(CLL) has been identified. These mutations cause a loss of enzyme function by blocking the biosynthesis of SI at the cell surface.
See also
*Sucrase Sucrase is a digestive enzyme that catalyzes the hydrolysis of sucrose to its subunits fructose and glucose. One form, sucrase-isomaltase, is secreted in the small intestine on the brush border. The sucrase enzyme invertase, which occurs more com ...
* Isomaltase
* Brush border
References
External links
Structure and evolution of the mammalian maltase-glucoamylase and sucrase-isomaltase
* {{Portal bar, Biology, border=no EC 3.2.1