Ribose on:
[Wikipedia]
[Google]
[Amazon]
 Ribose is a simple sugar and
Ribose is a simple sugar and
 Ribose is a simple sugar and
Ribose is a simple sugar and carbohydrate
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may o ...
with molecular formula C5H10O5 and the linear-form composition H−(C=O)−(CHOH)4−H. The naturally-occurring form, , is a component of the ribonucleotides from which RNA is built, and so this compound is necessary for coding, decoding, regulation and expression of gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a b ...
s. It has a structural analog, deoxyribose, which is a similarly essential component of DNA. is an unnatural sugar that was first prepared by Emil Fischer and Oscar Piloty in 1891. It was not until 1909 that Phoebus Levene and Walter Jacobs
Marion Walter Jacobs (May 1, 1930 – February 15, 1968), known as Little Walter, was an American blues musician, singer, and songwriter, whose revolutionary approach to the harmonica had a strong impact on succeeding generations, earning him ...
recognised that was a natural product, the enantiomer of Fischer and Piloty's product, and an essential component of nucleic acids. Fischer chose the name "ribose" as it is a partial rearrangement of the name of another sugar, arabinose
Arabinose is an aldopentose – a monosaccharide containing five carbon atoms, and including an aldehyde (CHO) functional group.
For biosynthetic reasons, most saccharides are almost always more abundant in nature as the "D"-form, or structural ...
, of which ribose is an epimer
In stereochemistry, an epimer is one of a pair of diastereomers. The two epimers have opposite configuration at only one stereogenic center out of at least two. All other stereogenic centers in the molecules are the same in each. Epimerization is ...
at the 2' carbon; both names also relate to gum arabic, from which arabinose was first isolated and from which they prepared .
Like most sugars, ribose exists as a mixture of cyclic forms in equilibrium with its linear form, and these readily interconvert especially in aqueous solution. The name "ribose" is used in biochemistry and biology to refer to all of these forms, though more specific names for each are used when required. In its linear form, ribose can be recognised as the pentose
In chemistry, a pentose is a monosaccharide (simple sugar) with five carbon atoms. The chemical formula of many pentoses is , and their molecular weight is 150.13 g/mol.hydroxyl functional groups on the same side in its Fischer projection. has these hydroxyl groups on the right hand side and is associated with the systematic name (2''R'',3''R'',4''R'')-2,3,4,5-tetrahydroxypentanal, whilst has its hydroxyl groups appear on the left hand side in a Fischer projection. Cyclisation of ribose occurs via hemiacetal formation due to attack on the aldehyde by the C4' hydroxyl group to produce a furanose form or by the C5' hydroxyl group to produce a pyranose form. In each case, there are two possible geometric outcomes, named as α- and β- and known as anomers, depending on the stereochemistry at the hemiacetal carbon atom (the "anomeric carbon"). At room temperature, about 76% of is present in pyranose forms (α:β = 1:2) and 24% in the furanose forms (α:β = 1:3), with only about 0.1% of the linear form present.
The
ribonucleoside A ribonucleoside is a type of nucleoside including ribose as a component.
One example of a ribonucleoside is cytidine
Cytidine (symbol C or Cyd) is a nucleoside molecule that is formed when cytosine is attached to a ribose ring (also known as ...
s adenosine, cytidine, guanosine, and uridine are all derivatives of β--ribofuranose. Metabolically-important species that include phosphorylated ribose include ADP
Adp or ADP may refer to:
Aviation
* Aéroports de Paris, airport authority for the Parisian region in France
* Aeropuertos del Perú, airport operator for airports in northern Peru
* SLAF Anuradhapura, an airport in Sri Lanka
* Ampara Airp ...
, ATP, coenzyme A, and NADH. cAMP
Camp may refer to:
Outdoor accommodation and recreation
* Campsite or campground, a recreational outdoor sleeping and eating site
* a temporary settlement for nomads
* Camp, a term used in New England, Northern Ontario and New Brunswick to descri ...
and cGMP serve as secondary messengers in some signaling pathways and are also ribose derivatives. The ribose moiety
Moiety may refer to:
Chemistry
* Moiety (chemistry), a part or functional group of a molecule
** Moiety conservation, conservation of a subgroup in a chemical species
Anthropology
* Moiety (kinship), either of two groups into which a society is ...
appears in some pharmaceutical agents, including the antibiotics neomycin and paromomycin
Paromomycin is an antimicrobial used to treat a number of parasitic infections including amebiasis, giardiasis, leishmaniasis, and tapeworm infection. It is a first-line treatment for amebiasis or giardiasis during pregnancy. Otherwise it is gen ...
.
Synthesis and sources
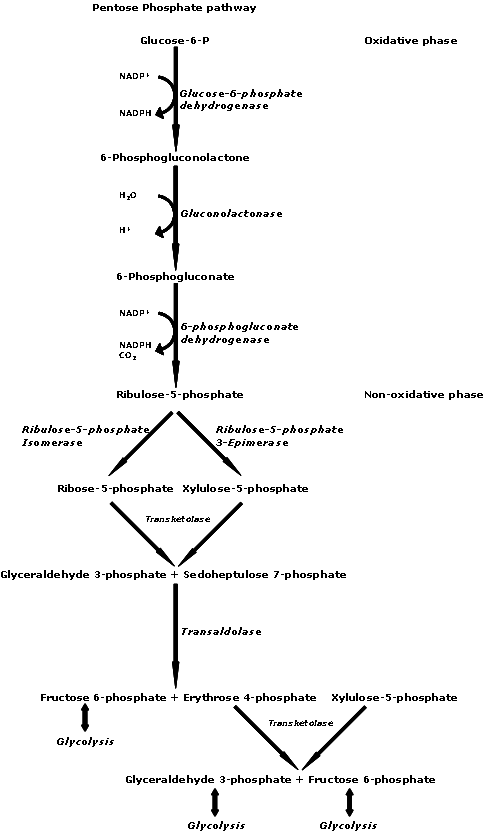
Ribose as its 5-phosphate ester is typically produced from glucose by the pentose phosphate pathway. In at least some archaea, alternative pathways have been identified. Ribose can be synthesized chemically, but commercial production relies on fermentation of glucose. Using genetically modified strains of ''B. subtilis
''Bacillus subtilis'', known also as the hay bacillus or grass bacillus, is a Gram-positive, catalase-positive bacterium, found in soil and the gastrointestinal tract of ruminants, humans and marine sponges. As a member of the genus ''Bacillus' ...
'', 90 g/liter of ribose can be produced from 200 g of glucose. The conversion entails the intermediacy of gluconate and ribulose.
Ribose has been detected in meteorites.
Structure
Ribose is analdopentose
In chemistry, a pentose is a monosaccharide (simple sugar) with five carbon atoms. The chemical formula of many pentoses is , and their molecular weight is 150.13 g/mol.carbon atoms that, in its open chain form, has an aldehyde functional group at one end). In the conventional numbering scheme for monosaccharides, the carbon atoms are numbered from C1' (in the aldehyde group) to C5'. The deoxyribose derivative found in DNA differs from ribose by having a
File:Alpha-D-Ribopyranose numbered.png, α--Ribopyranose
File:Beta-D-Ribopyranose numbered.png, β--Ribopyranose
File:Alpha-D-Ribofuranose numbered.png, α--Ribofuranose
File:Beta-D-Ribofuranose Numbered.png, β--Ribofuranose
Relative abundance of forms of ribose in solution: β--ribopyranose (59%), α--ribopyranose (20%), β--ribofuranose (13%), α--ribofuranose (7%) and open chain (0.1%).
For ribose residues in nucleosides and nucleotide, the torsion angles for the rotation encompassing the bonds influence the configuration of the respective nucleoside and nucleotide. The secondary structure of a nucleic acid is determined by the rotation of its 7 torsion angles. Having a large amount of torsion angles allows for greater flexibility.
In closed ring riboses, the observed flexibility mentioned above is not observed because the ring cycle imposes a limit on the number of torsion angles possible in the structure. Conformers of closed form riboses differ in regards to how the lone
File:2' endo.jpg, 2' endo
File:2' endo 3' exo.jpg, 2' endo 3' exo
File:3' endo 2' exo.jpg, 3' endo 2' exo
File:3' endo.jpg, 3' endo
A ribose molecule is typically represented as a planar molecule on paper. Despite this, it is typically non-planar in nature. Even between hydrogen atoms, the many constituents on a ribose molecule cause steric hindrance and strain between them. To relieve this crowding and ring strain, the ring puckers, i.e. becomes non-planar. This puckering is achieved by displacing an atom from the plane, relieving the strain and yielding a more stable configuration. Puckering, otherwise known as the sugar ring conformation (specifically ribose sugar), can be described by the amplitude of pucker as well as the pseudorotation angle. The pseudo-rotation angle can be described as either "north (N)" or "south (S)" range. While both ranges are found in double helices, the north range is commonly associated with RNA and the A form of DNA. In contrast, the south range is associated with B form DNA. Z-DNA contains sugars in both the north and south ranges. When only a single atom is displaced, it is referred to as an "envelope" pucker. When two atoms are displaced, it is referred to as a "twist" pucker, in reference to the zigzag orientation. In an "endo" pucker, the major displacement of atoms is on the β-face, the same side as the C4'-C5' bond and the base. In an "exo" pucker, the major displacement of atoms is on the α-face, on the opposite side of the ring. The major forms of ribose are the 3'-endo pucker (commonly adopted by RNA and A-form DNA) and 2'-endo pucker (commonly adopted by B-form DNA). These ring puckers are developed from changes in ring torsion angles; there are infinite combinations of angles so therefore, there is an infinite number of transposable pucker conformations, each separated by disparate activation energies.

hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-to ...
atom in place of the hydroxyl group at C2'. This hydroxyl group performs a function in RNA splicing.
The "-" in the name -ribose refers to the stereochemistry of the chiral carbon atom farthest away from the aldehyde group (C4'). In -ribose, as in all -sugars, this carbon atom has the same configuration as in -glyceraldehyde.oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements ...
in the molecule is positioned respective to the nitrogenous base (also known as a nucleobase
Nucleobases, also known as ''nitrogenous bases'' or often simply ''bases'', are nitrogen-containing biological compounds that form nucleosides, which, in turn, are components of nucleotides, with all of these monomers constituting the basi ...
or just a base) attached to the ribose. If a carbon is facing towards the base, then the ribose is labeled as endo. If a carbon is facing away from the base, then the ribose is labeled as exo. If there is an oxygen molecule attached to the 2' carbon of a closed cycle ribose, then the exo confirmation is more stable because it decreases the interactions of the oxygen with the base. The difference itself is quite small, but when looking at an entire chain of RNA the slight difference amounts to a sizable impact.Functions
ATP is derived from ribose; it contains one ribose, three phosphate groups, and an adenine base. ATP is created duringcellular respiration
Cellular respiration is the process by which biological fuels are oxidised in the presence of an inorganic electron acceptor such as oxygen to produce large amounts of energy, to drive the bulk production of ATP. Cellular respiration may be des ...
from adenosine diphosphate (ATP with one less phosphate group).
Signaling pathways
Ribose is a building block in secondary signaling molecules such as cyclic adenosine monophosphate (cAMP) which is derived from ATP. One specific case in which cAMP is used is in cAMP-dependent signaling pathways. In cAMP signaling pathways, either a stimulative or inhibitory hormone receptor is activated by a signal molecule. These receptors are linked to a stimulative or inhibitory regulative G-protein. When a stimulative G-protein is activated,adenylyl cyclase
Adenylate cyclase (EC 4.6.1.1, also commonly known as adenyl cyclase and adenylyl cyclase, abbreviated AC) is an enzyme with systematic name ATP diphosphate-lyase (cyclizing; 3′,5′-cyclic-AMP-forming). It catalyzes the following reaction:
:A ...
catalyzes ATP into cAMP by using Mg2+ or Mn2+. cAMP, a secondary messenger, then goes on to activate protein kinase A, which is an enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
that regulates cell metabolism
Metabolism (, from el, μεταβολή ''metabolē'', "change") is the set of life-sustaining chemical reactions in organisms. The three main functions of metabolism are: the conversion of the energy in food to energy available to run ...
. Protein kinase A regulates metabolic enzymes by phosphorylation which causes a change in the cell depending on the original signal molecule. The opposite occurs when an inhibitory G-protein is activated; the G-protein inhibits adenylyl cyclase and ATP is not converted to cAMP. 
Metabolism
Ribose is referred to as the "molecular currency" because of its involvement in intracellular energy transfers. For example, nicotinamide adenine dinucleotide (NAD), flavin adenine dinucleotide (FAD), and nicotinamide adenine dinucleotide phosphate (NADP) all contain the -ribofuranosemoiety
Moiety may refer to:
Chemistry
* Moiety (chemistry), a part or functional group of a molecule
** Moiety conservation, conservation of a subgroup in a chemical species
Anthropology
* Moiety (kinship), either of two groups into which a society is ...
. They can each be derived from -ribose after it is converted to -ribose 5-phosphate by the enzyme ribokinase
In enzymology, a ribokinase () is an enzyme that catalyzes the chemical reaction
:ATP + -ribose ⇌ ADP + -ribose 5-phosphate
Thus, the two substrates of this enzyme are ATP and -ribose, whereas its two products are ADP and -ribose 5-phospha ...
. NAD, FAD, and NADP act as electron acceptors in biochemical redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or ...
reactions in major metabolic pathways including glycolysis, the citric acid cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and prot ...
, fermentation, and the electron transport chain.

Nucleotide biosynthesis
Nucleotides are synthesized through salvage orde novo synthesis
In chemistry, ''de novo'' synthesis () refers to the synthesis of complex molecules from simple molecules such as sugars or amino acids, as opposed to recycling after partial degradation. For example, nucleotides are not needed in the diet as ...
. Nucleotide salvage uses pieces of previously made nucleotides and re-synthesizes them for future use. In de novo, amino acids, carbon dioxide, folate derivatives, and phosphoribosyl pyrophosphate (PRPP) are used to synthesize nucleotides. Both de novo and salvage require PRPP which is synthesized from ATP and ribose 5-phosphate by an enzyme called PRPP synthetase.
Modifications
Modifications in nature
Ribokinase
In enzymology, a ribokinase () is an enzyme that catalyzes the chemical reaction
:ATP + -ribose ⇌ ADP + -ribose 5-phosphate
Thus, the two substrates of this enzyme are ATP and -ribose, whereas its two products are ADP and -ribose 5-phospha ...
catalyzes the conversion of -ribose to -ribose 5-phosphate. Once converted, -ribose-5-phosphate is available for the manufacturing of the amino acids tryptophan and histidine
Histidine (symbol His or H) is an essential amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated –NH3+ form under biological conditions), a carboxylic acid group (which is in the ...
, or for use in the pentose phosphate pathway. The absorption of -ribose is 88–100% in the small intestines (up to 200 mg/kg·h).
One important modification occurs at the C2' position of the ribose molecule. By adding an O-alkyl group, the nuclear resistance of the RNA is increased because of additional stabilizing forces. These forces are stabilizing because of the increase of intramolecular hydrogen bonding and an increase in the glycosidic bond stability. The resulting increase of resistance leads to increases in the half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ...
of siRNA and the potential therapeutic potential in cells and animals. The methylation of ribose at particular sites is correlated with a decrease in immune stimulation.
Synthetic modifications
Along with phosphorylation, ribofuranose molecules can exchange their oxygen with selenium and sulfur to produce similar sugars that only vary at the 4' position. These derivatives are more lipophilic than the original molecule. Increased lipophilicity makes these species more suitable for use in techniques such as PCR, RNA aptamer post-modification, antisense technology, and for phasing X-ray crystallographic data. Similar to the 2' modifications in nature, a synthetic modification of ribose includes the addition of fluorine at the 2' position. Thisfluorinated
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers ...
ribose acts similar to the methylated ribose because it is capable of suppressing immune stimulation depending on the location of the ribose in the DNA strand. The big difference between methylation and fluorination, is the latter only occurs through synthetic modifications. The addition of fluorine leads to an increase in the stabilization of the glycosidic bond and an increase of intramolecular hydrogen bonds.
Medical uses
-ribose has been suggested for use in management of congestive heart failure (as well as other forms of heart disease) and for chronic fatigue syndrome (CFS), also called myalgic encephalomyelitis (ME) in an open-label non-blinded, non-randomized, and non-crossover subjective study. Supplemental -ribose can bypass part of the pentose phosphate pathway, an energy-producing pathway, to produce -ribose-5-phosphate. The enzyme glucose-6-phosphate-dehydrogenase (G-6-PDH) is often in short supply in cells, but more so in diseased tissue, such as in myocardial cells in patients with cardiac disease. The supply of -ribose in the mitochondria is directly correlated with ATP production; decreased -ribose supply reduces the amount of ATP being produced. Studies suggest that supplementing -ribose following tissue ischemia (e.g. myocardial ischemia) increases myocardial ATP production, and therefore mitochondrial function. Essentially, administering supplemental -ribose bypasses an enzymatic step in the pentose phosphate pathway by providing an alternate source of 5-phospho--ribose 1- pyrophosphate for ATP production. Supplemental -ribose enhances recovery of ATP levels while also reducing cellular injury in humans and other animals. One study suggested that the use of supplemental -ribose reduces the instance of angina in men with diagnosed coronary artery disease. -Ribose has been used to treat many pathological conditions, such as chronic fatigue syndrome, fibromyalgia, and myocardial dysfunction. It is also used to reduce symptoms of cramping, pain, stiffness, etc. after exercise and to improve athletic performance.References