meta-Chloroperoxybenzoic acid on:
[Wikipedia]
[Google]
[Amazon]
''meta''-Chloroperoxybenzoic acid (mCPBA or ''m''CPBA) is a peroxycarboxylic acid. A white solid, it is used widely as an
 The epoxidation mechanism is concerted: the ''cis'' or ''trans'' geometry of the alkene starting material is retained in the epoxide ring of the product. The transition state of the Prilezhaev reaction is given below:
The epoxidation mechanism is concerted: the ''cis'' or ''trans'' geometry of the alkene starting material is retained in the epoxide ring of the product. The transition state of the Prilezhaev reaction is given below:
 The geometry of the transition state, with the peracid bisecting the C-C double bond, allows the two primary frontier orbital interactions to occur: πC=C (HOMO) to σ*O-O (LUMO) and nO (HOMO, regarded as a filled p orbital on a sp2 hybridized oxygen) to π*C=C (LUMO), corresponding, in arrow-pushing terms, to formation of one C-O bond and cleavage of the O-O bond and formation of the other C-O bond and cleavage of the C=C π bond.
The geometry of the transition state, with the peracid bisecting the C-C double bond, allows the two primary frontier orbital interactions to occur: πC=C (HOMO) to σ*O-O (LUMO) and nO (HOMO, regarded as a filled p orbital on a sp2 hybridized oxygen) to π*C=C (LUMO), corresponding, in arrow-pushing terms, to formation of one C-O bond and cleavage of the O-O bond and formation of the other C-O bond and cleavage of the C=C π bond.
oxidant
An oxidizing agent (also known as an oxidant, oxidizer, electron recipient, or electron acceptor) is a substance in a redox chemical reaction that gains or " accepts"/"receives" an electron from a (called the , , or ). In other words, an oxi ...
in organic synthesis
Organic synthesis is a special branch of chemical synthesis and is concerned with the intentional construction of organic compounds. Organic molecules are often more complex than inorganic compounds, and their synthesis has developed into one o ...
. mCPBA is often preferred to other peroxy acids because of its relative ease of handling. mCPBA is a strong oxidizing agent that may cause fire upon contact with flammable material.
Preparation and purification
mCPBA can be prepared by reacting m-Chlorobenzoyl chloride with a basic solution ofhydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3 ...
, followed by acidification.
It is sold commercially as a shelf-stable mixture that is less than 72% mCPBA, with the balance made up of ''m''-chlorobenzoic acid (10%) and water. The peroxyacid can be purified by washing the commercial material with a sodium hydroxide and potassium phosphate solution buffered at pH = 7.5. Peroxyacids are generally slightly less acidic than their carboxylic acid counterparts, so one can extract the acid impurity by careful control of pH. The purified material is reasonably stable against decomposition if stored at low temperatures in a plastic container.
In reactions where the exact amount of mCPBA must be controlled, a sample can be titrated to determine the exact amount of active oxidant.
Reactions
The main areas of use are the conversion ofketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double b ...
s to ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides ...
s ( Baeyer-Villiger oxidation), epoxidation
In organic chemistry, an epoxide is a cyclic ether () with a three-atom ring. This ring approximates an equilateral triangle, which makes it strained, and hence highly reactive, more so than other ethers. They are produced on a large scale ...
of alkenes
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, a ...
(Prilezhaev reaction
The Prilezhaev reaction, also known as the Prileschajew reaction or Prilezhaev epoxidation, is the chemical reaction of an alkene with a peroxy acid to form epoxides. It is named after Nikolai Prilezhaev, who first reported this reaction in 1909 ...
), conversion of silyl enol ethers
Silyl enol ethers in organic chemistry are a class of organic compounds that share a common functional group composed of an enolate bonded through its oxygen end to an organosilicon group. They are important intermediates in organic synthesis.
Syn ...
to silyl α-hydroxy ketones ( Rubottom oxidation), oxidation of sulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds la ...
s to sulfoxide
In organic chemistry, a sulfoxide, also called a sulphoxide, is an organosulfur compound containing a sulfinyl () functional group attached to two carbon atoms. It is a polar functional group. Sulfoxides are oxidized derivatives of sulfides. E ...
s and sulfone
In organic chemistry, a sulfone is a organosulfur compound containing a sulfonyl () functional group attached to two carbon atoms. The central hexavalent sulfur atom is double-bonded to each of two oxygen atoms and has a single bond to each of ...
s, and oxidation of amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent ...
s to produce amine oxide
In chemistry, an amine oxide, also known as an amine ''N''-oxide or simply ''N''-oxide, is a chemical compound that contains the functional group , a nitrogen-oxygen coordinate covalent bond with three additional hydrogen and/or substituent-grou ...
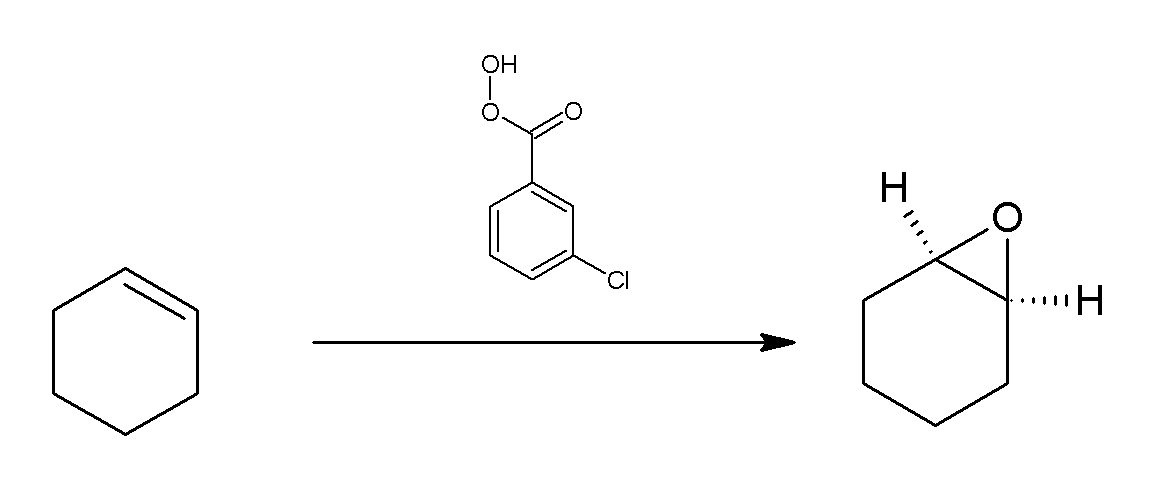
s. The following scheme shows the epoxidation of cyclohexene
Cyclohexene is a hydrocarbon with the formula C6H10. This cycloalkene is a colorless liquid with a sharp smell. It is an intermediate in various industrial processes. Cyclohexene is not very stable upon long term storage with exposure to light ...
with mCPBA.
: The epoxidation mechanism is concerted: the ''cis'' or ''trans'' geometry of the alkene starting material is retained in the epoxide ring of the product. The transition state of the Prilezhaev reaction is given below:
The epoxidation mechanism is concerted: the ''cis'' or ''trans'' geometry of the alkene starting material is retained in the epoxide ring of the product. The transition state of the Prilezhaev reaction is given below:
 The geometry of the transition state, with the peracid bisecting the C-C double bond, allows the two primary frontier orbital interactions to occur: πC=C (HOMO) to σ*O-O (LUMO) and nO (HOMO, regarded as a filled p orbital on a sp2 hybridized oxygen) to π*C=C (LUMO), corresponding, in arrow-pushing terms, to formation of one C-O bond and cleavage of the O-O bond and formation of the other C-O bond and cleavage of the C=C π bond.
The geometry of the transition state, with the peracid bisecting the C-C double bond, allows the two primary frontier orbital interactions to occur: πC=C (HOMO) to σ*O-O (LUMO) and nO (HOMO, regarded as a filled p orbital on a sp2 hybridized oxygen) to π*C=C (LUMO), corresponding, in arrow-pushing terms, to formation of one C-O bond and cleavage of the O-O bond and formation of the other C-O bond and cleavage of the C=C π bond.
References
{{DEFAULTSORT:Chloroperoxybenzoic acid, meta- Chlorobenzenes Oxidizing agents Organic peroxy acids Reagents for organic chemistry