Myotonic Dystrophy Protein Kinase on:
[Wikipedia]
[Google]
[Amazon]
Myotonin-protein kinase (MT-PK) also known as myotonic dystrophy protein kinase (MDPK) or dystrophia myotonica protein kinase (DMPK) is an
 Myotonic dystrophy (DM) 1 is an autosomal dominant neuromuscular disorder affecting approximately 1 in 8000 individuals. Affected individuals display a wide range of symptoms including myotonia, skeletal muscle weakness and wasting, cardiac conduction abnormalities, and cataracts. Despite cloning of the locus, the complex disease phenotype of DM has proven difficult to interpret, and the exact role of DMPK in the pathogenesis of DM remains unclear.
Myotonic dystrophy (DM) 1 is an autosomal dominant neuromuscular disorder affecting approximately 1 in 8000 individuals. Affected individuals display a wide range of symptoms including myotonia, skeletal muscle weakness and wasting, cardiac conduction abnormalities, and cataracts. Despite cloning of the locus, the complex disease phenotype of DM has proven difficult to interpret, and the exact role of DMPK in the pathogenesis of DM remains unclear.
GeneReviews/NCBI/NIH/UW entry on Myotonic Dystrophy Type 1
{{DEFAULTSORT:Myotonin-Protein Kinase EC 2.7.11 Human proteins
enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. A ...
that in humans is encoded by the '' DMPK'' gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a ba ...
.
The dmpk gene product is a Ser/Thr protein kinase homologous to the MRCK p21-activated kinases and Rho kinase family. Data obtained by using antibodies that detect specific isoforms of DMPK indicate that the most abundant isoform of DMPK is an 80-kDa protein expressed almost exclusively in smooth, skeletal, and cardiac muscles. This kinase exists both as a membrane-associated and as a soluble form in human left ventricular samples. The different C termini of DMPK that arise from alternative splicing determine its localization to the endoplasmic reticulum, mitochondria, or cytosol in transfected COS-1 cells. Among the substrates for DMPK proposed by in vitro studies are phospholemman, the dihydropyridine receptor, and the myosin phosphatase targeting subunit. However, an in vivo demonstration of the phosphorylation of these substrates by DMPK remains to be established, and a link between these substrates and the clinical manifestations of myotonic dystrophy (DM) is unclear.
Function
Myotonin-protein kinase is aserine-threonine kinase
A serine/threonine protein kinase () is a kinase enzyme, in particular a protein kinase, that phosphorylation, phosphorylates the hydroxyl, OH group of the amino acid, amino-acid residues serine or threonine, which have similar side chains. ...
that is closely related to other kinases that interact with members of the Rho family
The Rho family of GTPases is a family of small (~21 kDa) signaling G proteins, and is a subfamily of the Ras superfamily. The members of the Rho GTPase family have been shown to regulate many aspects of intracellular actin dynamics, and are found ...
of small GTPase
GTPases are a large family of hydrolase enzymes that bind to the nucleotide guanosine triphosphate (GTP) and hydrolyze it to guanosine diphosphate (GDP). The GTP binding and hydrolysis takes place in the highly conserved P-loop "G domain", a pro ...
s. Substrates for this enzyme include myogenin
Myogenin, is a transcriptional activator encoded by the MYOG gene.
Myogenin is a muscle-specific basic-helix-loop-helix (bHLH) transcription factor involved in the coordination of skeletal muscle development or myogenesis and repair. Myogenin is ...
, the beta-subunit of the L-type calcium channel
The L-type calcium channel (also known as the dihydropyridine channel, or DHP channel) is part of the high-voltage activated family of voltage-dependent calcium channel.
"L" stands for long-lasting referring to the length of activation. This c ...
s, and phospholemman. Although the specific function of this protein is unknown, it appears to play an important role in muscle, heart, and brain cells. This protein may be involved in communication within cells. It also appears to regulate the production and function of important structures inside muscle cells by interacting with other proteins. For example, myotonic dystrophy protein kinase has been shown to turn off (inhibit) part of a muscle protein called myosin phosphatase. Myosin phosphatase is an enzyme that plays a role in muscle tensing (contraction) and relaxation.
Structure
Dystrophia myotonica protein kinase (DMPK) is a serine/threonine kinase composed of a kinase domain and a coiled-coil domain involved in the multimerization. The crystal structure of the kinase domain of DMPK bound to the inhibitor bisindolylmaleimide VIII (BIM-8) revealed a dimeric enzyme associated by a conserved dimerization domain. The affinity of dimerisation suggested that the kinase domain alone is insufficient for dimerisation in vivo and that the coiled-coil domains are required for stable dimer formation. The kinase domain is in an active conformation, with a fully ordered and correctly positioned aC helix, and catalytic residues in a conformation competent for catalysis. The conserved hydrophobic motif at the C-terminal extension of the kinase domain is bound to the N-terminal lobe of the kinase domain, despite being unphosphorylated.Clinical significance
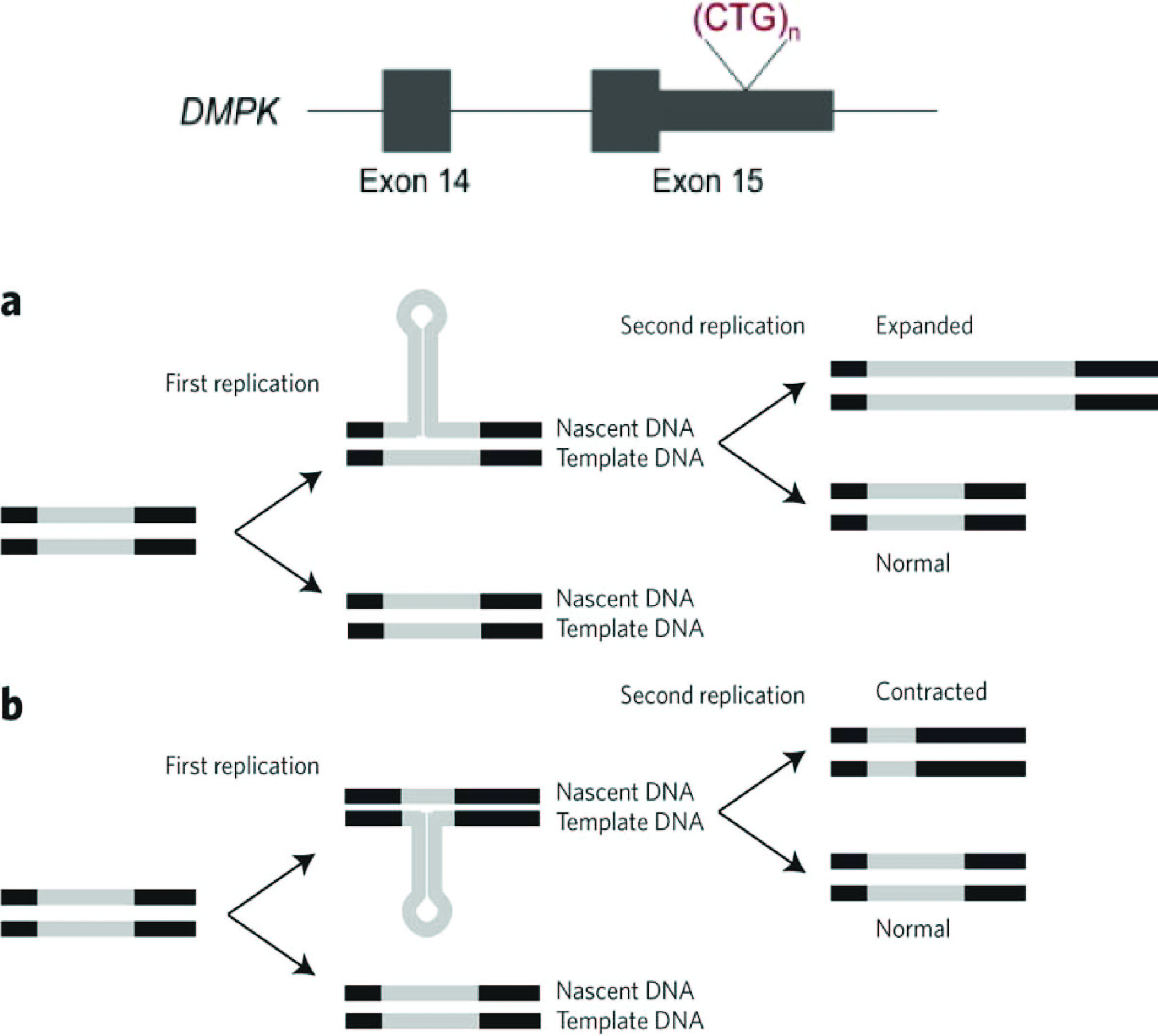
The 3' untranslated region of this gene contains 5-37 copies of a CTG trinucleotide repeat. Expansion of this unstable motif to 50-5,000 copies causesmyotonic dystrophy
Myotonic dystrophy (DM) is a type of muscular dystrophy, a group of genetic disorders that cause progressive muscle loss and weakness. In DM, muscles are often unable to relax after contraction. Other manifestations may include cataracts, intel ...
type I, which increases in severity with increasing repeat element copy number. Repeat expansion is associated with condensation of local chromatin structure that disrupts the expression of genes in this region. As the DMPK repeat is replicated, the hairpin loop that is formed leads to repeat expansion (a) or contractions (b).
 Myotonic dystrophy (DM) 1 is an autosomal dominant neuromuscular disorder affecting approximately 1 in 8000 individuals. Affected individuals display a wide range of symptoms including myotonia, skeletal muscle weakness and wasting, cardiac conduction abnormalities, and cataracts. Despite cloning of the locus, the complex disease phenotype of DM has proven difficult to interpret, and the exact role of DMPK in the pathogenesis of DM remains unclear.
Myotonic dystrophy (DM) 1 is an autosomal dominant neuromuscular disorder affecting approximately 1 in 8000 individuals. Affected individuals display a wide range of symptoms including myotonia, skeletal muscle weakness and wasting, cardiac conduction abnormalities, and cataracts. Despite cloning of the locus, the complex disease phenotype of DM has proven difficult to interpret, and the exact role of DMPK in the pathogenesis of DM remains unclear.
Interactions
Myotonic dystrophy protein kinase has been shown tointeract
Advocates for Informed Choice, dba interACT or interACT Advocates for Intersex Youth, is a 501(c)(3) nonprofit organization using innovative strategies to advocate for the legal and human rights of children with intersex traits. The organizati ...
with HSPB2
Heat shock protein beta-2 is a protein that in humans is encoded by the ''HSPB2'' gene.
Interactions
HSPB2 has been shown to interact with:
* CRYAB,
* HSPB8,
* Myotonic dystrophy protein kinase and
* TRAF6
TRAF6 is a TRAF human protein.
...
and RAC1.
Regulation
The close relationship of DMPK to the Rho-kinases has led to speculation whether DMPK activity may be regulated in vivo by small G proteins, particularly of the Rho family. Although DMPK lacks obvious binding sites for known G, DMPK-1 oligomers exhibit low basal catalytic activity due to the presence of the C-terminal autoinhibitory domain (AI). A protease (P) within the membrane cleaves DMPK-1, removing the C-terminal autoinhibitory and membrane association domains and releasing cytosolic, basally active DMPK-2. This processing event would produce longterm activation of the kinase. Short-term activation of DMPK-1 and -2 may be mediated by transitory interaction with a small GTPase (G). A general model that accounts for DMPK oligomerization, processing, and regulation has been proposed. In this model, transient activation of kinase activity would occur in response to G protein second messengers, while longterm activation of DMPK could be mediated by a membrane associated protease that cleaves DMPK-1 to release cytosolic DMPK-2 in a persistently activated form. The persistent activation of serine/threonine kinases has been shown to play a role in the determination of cell fate as well as memory production in the nervous system. In this respect, DMPK may be similar to PKA and PKC, two kinases that can be transiently activated in response to second messengers or persistently activated by proteolytic removal of an autoinhibitory domain. Thus, this model suggests that the two endogenous DMPK forms may possess different activities, localizations, regulators, and substrates and perform distinct physiological functions.References
Further reading
* * * * * * * * * * * * * * * * *External links
GeneReviews/NCBI/NIH/UW entry on Myotonic Dystrophy Type 1
{{DEFAULTSORT:Myotonin-Protein Kinase EC 2.7.11 Human proteins