Migratory Insertion on:
[Wikipedia]
[Google]
[Amazon]
In
 CO insertion does not always involve migration. Treatment of CpFe(L)(CO)CH3 with 13CO yields a mix of both alkyl migration products and products formed by true insertion of bound
CO insertion does not always involve migration. Treatment of CpFe(L)(CO)CH3 with 13CO yields a mix of both alkyl migration products and products formed by true insertion of bound 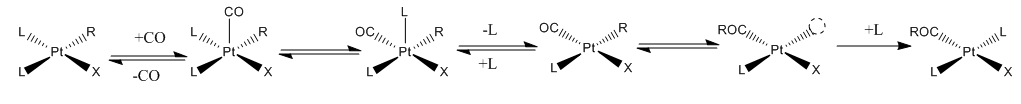


 The Cativa process
The Cativa process
Migratory Insertion
{{DEFAULTSORT:Migratory Insertion Organometallic chemistry Reaction mechanisms
organometallic chemistry
Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and so ...
, a migratory insertion is a type of reaction
Reaction may refer to a process or to a response to an action, event, or exposure:
Physics and chemistry
*Chemical reaction
*Nuclear reaction
*Reaction (physics), as defined by Newton's third law
*Chain reaction (disambiguation).
Biology and me ...
wherein two ligand
In coordination chemistry, a ligand is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electr ...
s on a metal complex
A coordination complex consists of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many ...
combine. It is a subset of reactions that very closely resembles the insertion reaction
An insertion reaction is a chemical reaction where one chemical entity (a molecule or molecular fragment) interposes itself into an existing bond of typically a second chemical entity ''e.g.'':
: + \longrightarrow
The term only refers to the ...
s, and both are differentiated by the mechanism
Mechanism may refer to:
* Mechanism (engineering), rigid bodies connected by joints in order to accomplish a desired force and/or motion transmission
*Mechanism (biology), explaining how a feature is created
*Mechanism (philosophy), a theory that ...
that leads to the resulting stereochemistry
Stereochemistry, a subdiscipline of chemistry, involves the study of the relative spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereois ...
of the products. However, often the two are used interchangeably because the mechanism is sometimes unknown. Therefore, migratory insertion reactions or insertion reactions, for short, are defined not by the mechanism but by the overall regiochemistry
In chemistry, regioselectivity is the preference of chemical bonding or breaking in one direction over all other possible directions. It can often apply to which of many possible positions a reagent will affect, such as which proton a strong base ...
wherein one chemical entity interposes itself into an existing bond
Bond or bonds may refer to:
Common meanings
* Bond (finance), a type of debt security
* Bail bond, a commercial third-party guarantor of surety bonds in the United States
* Chemical bond, the attraction of atoms, ions or molecules to form chemica ...
of typically a second chemical entity e.g.:
:
Overview
In the migratory insertion, a ligand that is viewed as ananion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
(X) ligand in and a ligand that is viewed as neutral
Neutral or neutrality may refer to:
Mathematics and natural science Biology
* Neutral organisms, in ecology, those that obey the unified neutral theory of biodiversity
Chemistry and physics
* Neutralization (chemistry), a chemical reaction in ...
couple, generating a new anionic ligand. The anion and neutral ligands that react are adjacent. If the precursor
Precursor or Precursors may refer to:
* Precursor (religion), a forerunner, predecessor
** The Precursor, John the Baptist
Science and technology
* Precursor (bird), a hypothesized genus of fossil birds that was composed of fossilized parts of u ...
complex is coordinatively saturated, migratory insertion often result in a coordinatively unsaturated
In chemistry, a saturated compound is a chemical compound (or ion) that resists the addition reactions, such as hydrogenation, oxidative addition, and binding of a Lewis base. The term is used in many contexts and for many classes of chemical ...
product. A new (neutral) ligand can then react with the metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
leading to a further insertion. The process can occur many times on a single metal, as in olefin polymerization.
The anionic ligand can be: H− (hydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride of ...
), R− (alkyl), acyl
In chemistry, an acyl group is a moiety derived by the removal of one or more hydroxyl groups from an oxoacid, including inorganic acids. It contains a double-bonded oxygen atom and an alkyl group (). In organic chemistry, the acyl group (IUPAC ...
, Ar− (aryl
In organic chemistry, an aryl is any functional group or substituent derived from an aromatic ring, usually an aromatic hydrocarbon, such as phenyl and naphthyl. "Aryl" is used for the sake of abbreviation or generalization, and "Ar" is used as ...
), or OR− ( alkoxide). The ability of these groups to migrate is called their migratory aptitude Migratory aptitude is the relative ability of a migrating group to migrate in a rearrangement reaction. Migratory aptitudes vary in different reactions, depending on multiple factors.
In the Baeyer-Villiger reaction, the more substituted group, i ...
. The neutral ligand can be CO, alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
, alkyne
\ce
\ce
Acetylene
\ce
\ce
\ce
Propyne
\ce
\ce
\ce
\ce
1-Butyne
In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and n ...
, or in some cases, even carbene
In organic chemistry, a carbene is a molecule containing a neutral carbon atom with a valence of two and two unshared valence electrons. The general formula is or where the R represents substituents or hydrogen atoms.
The term "carbene" ma ...
.
Diverse reactions apply to the migratory insertion. One mechanism involves the attack of the anionic ligand on the electrophilic
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carri ...
part of the neutral ligand (the anionic ligand migrates to the neutral ligand). The other mechanism involves the neutral ligand inserts itself between the metal and the anionic ligand.
CO insertion
The insertion ofcarbon monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simple ...
into a metal-carbon bond to form an acyl
In chemistry, an acyl group is a moiety derived by the removal of one or more hydroxyl groups from an oxoacid, including inorganic acids. It contains a double-bonded oxygen atom and an alkyl group (). In organic chemistry, the acyl group (IUPAC ...
group is the basis of carbonylation
Carbonylation refers to reactions that introduce carbon monoxide into organic and inorganic substrates. Carbon monoxide is abundantly available and conveniently reactive, so it is widely used as a reactant in industrial chemistry. The term carbon ...
reactions, which provides many commercially useful products.
Mechanism
CO inserts into a metal-alkyl
In organic chemistry, an alkyl group is an alkane missing one hydrogen.
The term ''alkyl'' is intentionally unspecific to include many possible substitutions.
An acyclic alkyl has the general formula of . A cycloalkyl is derived from a cycloalk ...
bond via migratory insertion. The key concept is that both the CO and the alkyl groups are ligands on the same metal. For example, the reaction of 13CO with Mn(CO)5CH3 exclusively form Mn(CO)4(13CO)COCH3. The alkyl group migrates intramolecularly to an adjacent CO ligand within the coordination sphere
In coordination chemistry, the first coordination sphere refers to the array of molecules and ions (the ligands) directly attached to the central metal atom. The second coordination sphere consists of molecules and ions that attached in various ...
of the Mn(I) centre. Subsequent to the migration, the metal binds free CO (see figure below).
: CO insertion does not always involve migration. Treatment of CpFe(L)(CO)CH3 with 13CO yields a mix of both alkyl migration products and products formed by true insertion of bound
CO insertion does not always involve migration. Treatment of CpFe(L)(CO)CH3 with 13CO yields a mix of both alkyl migration products and products formed by true insertion of bound carbonyls
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a ...
into the methyl
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula . In formulas, the group is often abbreviated as Me. This hydrocarbon group occurs in many ...
group. Product distribution is influenced by the choice of solvent
A solvent (s) (from the Latin '' solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for ...
.
Alkyl derivatives of square planar complexes undergo CO insertions particularly readily. Insertion reactions on square planar complexes are of particular interest because of their industrial applications. Since square planar complexes are often coordinatively unsaturated
In chemistry, a saturated compound is a chemical compound (or ion) that resists the addition reactions, such as hydrogenation, oxidative addition, and binding of a Lewis base. The term is used in many contexts and for many classes of chemical ...
, they are susceptible to formation of 5-coordinate adducts, which undergo migratory insertion readily. In most cases the in-plane migration pathway is preferred, but, unlike the nucleophilic
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are ...
pathway, it is inhibited by an excess of CO.

Effects on reaction rates
* Steric effects strain – Increasing the steric strain of thechelate
Chelation is a type of bonding of ions and molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central metal atom. These ligands are ...
backbone in square planar complexes pushes the carbonyl and methyl groups closer together, increasing the reactivity of insertion reactions.
*Oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
– Oxidation of the metal tends to increase insertion reaction rates. The main rate-limiting step in the mechanism is the migration of the methyl group onto a carbonyl ligand, oxidizing the metal by imparting a greater partial positive charge on the acetyl carbon, and thus increasing the rate of reaction.
*Lewis acids
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any spe ...
– Lewis acids also increase the reaction rates, for reasons similar to metal oxidation increasing the positive charge on the carbon. Lewis acids bind to the CO oxygen and remove charge, increasing the electrophilicity of the carbon. This can increase the reaction rate by a factor of up to 108, and the complex formed is stable enough that the reaction proceeds even without additional CO to bind to the metal.
*Electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
of the leaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
- Increasing the electronegativity of the leaving alkyl group stabilizes the metal-carbon bond interaction and thus increases the activation energy
In chemistry and physics, activation energy is the minimum amount of energy that must be provided for compounds to result in a chemical reaction. The activation energy (''E''a) of a reaction is measured in joules per mole (J/mol), kilojoules pe ...
required for migration, decreasing the reaction rate.
* ''Trans''-effect – Ligands in an octahedral or square planar complex are known to influence the reactivity of the group to which they are ''trans''. This ligand influence is often referred to as the ''trans''-influence, and it varies in intensity between ligands. A partial list of ''trans''-influencing ligands is as follows, from highest ''trans''-effect to lowest: aryl
In organic chemistry, an aryl is any functional group or substituent derived from an aromatic ring, usually an aromatic hydrocarbon, such as phenyl and naphthyl. "Aryl" is used for the sake of abbreviation or generalization, and "Ar" is used as ...
, alkyl > NR3 > PR3 > AsR3 > CO > Cl. Ligands with a greater ''trans''-influence impart greater electrophilicity to the active site. Increasing the electrophilicity of the CO group has been shown experimentally to greatly increase the reaction rate, while decreasing the electrophilicity of the methyl group slightly increases the reaction rate. This can be demonstrated by reacting a square planar PN)M(CO)(CH3)complex with CO, where PN is a bidentate phosphorus- or nitrogen-bound ligand. This reaction proceeds in much greater yield when the methyl group is ''trans''-P and the CO ''trans''-N, owing to the higher ''trans''-influence of the more electronegative nitrogen.
Reverse reaction
Decarbonylation
Decarbonylation is a type of organic reaction that involves loss of CO. It is often an undesirable reaction since it represents a degradation. In the chemistry of metal carbonyls, decarbonylation describes a substitution process, whereby a CO lig ...
of aldehydes
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
, the reverse of CO insertion, is a well-recognized reaction:
:RCHO → RH + CO
The reaction is not widely practiced in part because the alkanes
In organic chemistry, an alkane, or paraffin (a historical trivial name that also has other meanings), is an acyclic saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in whi ...
are less useful materials than are the aldehyde precursors. Furthermore, the reaction is not often conducted catalytically because the extruded CO can be slow to dissociate. Extrusion of CO from an organic aldehyde is most famously demonstrated using Wilkinson's catalyst
Wilkinson's catalyst is the common name for chloridotris(triphenylphosphine)rhodium(I), a coordination complex of rhodium with the formula hCl(PPh3)3(Ph = phenyl). It is a red-brown colored solid that is soluble in hydrocarbon solvents such as ...
:
:RhCl(PPh3)3 + RCHO → RhCl(CO)(PPh3)2 + RH + PPh3
Please see Tsuji-Wilkinson Decarbonylation Reaction for an example of this elementary organometallic step in synthesis
Insertion of other oxides
Many electrophilic oxides insert into metal carbon bonds; these includesulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a toxic gas responsible for the odor of burnt matches. It is released naturally by volcanic activ ...
, carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
, and nitric oxide. These reactions have limited or no practical significance, but are of historic interest. With transition metal alkyls, these oxides behave as electrophiles and insert into the bond between metals and their relatively nucleophilic alkyl ligands. As discussed in the article on Metal sulfur dioxide complex
In organometallic chemistry, metal sulfur dioxide complexes are complexes that contain sulfur dioxide, , bonded to a transition metal. Such compounds are common but are mainly of theoretical interest. Historically, the study of these compounds ...
es, the insertion of SO2 has been examined in particular detail. SO2 inserts to give both ''O''-sulphinates and ''S''-sulphinates, depending on the metal centre. With square planar alkyl complexes, a pre-equilibrium is assumed involving formation of an adduct.
Insertion of alkenes into metal-carbon bonds
The insertion ofalkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s into both metal-carbon is important. The insertion of ethylene and propylene into titanium alkyls is the cornerstone of Ziegler–Natta catalysis, the main source of polyethylene and polypropylene. The majority of this technology involves heterogeneous catalysts, but it is widely assumed that the principles and observations on homogeneous systems are applicable to the solid-state versions. Related technologies include the Shell Higher Olefin Process
The Shell higher olefin process (SHOP) is a chemical process for the production of linear alpha olefins via ethylene oligomerization and olefin metathesis invented and exploited by Royal Dutch Shell.''Industrial Organic Chemistry'', Klaus Weisserm ...
which produces detergent
A detergent is a surfactant or a mixture of surfactants with cleansing properties when in dilute solutions. There are a large variety of detergents, a common family being the alkylbenzene sulfonates, which are soap-like compounds that are more ...
precursors.
:
Mechanism
Factors affecting the rate of olefin insertions include the formation of the cyclic, planar, four-center transition state involving incipient formation of a bond between the metal and an olefin carbon. From this transition state, it can be seen that a partial positive charge forms on the β-carbon with a partial negative charge formed on the carbon initially bonded to the metal. This polarization explains the subsequently observed formation of the bond between the negatively charged carbon/hydrogen and the positively charged β-carbon as well as the simultaneous formation of the metal-α-carbon bond. This transition state also highlights the two factors that most strongly contribute to the rate of olefin insertion reactions: (i) orbital overlap of the alkyl group initially attached to the metal and (ii) the strength of the metal-alkyl bond. With greater orbital overlap between the partially positive β-carbon and the partially negative hydrogen/alkyl group carbon, the formation of the new C-C bond is facilitated. With increasing strength of the metal-alkyl bond, the breaking of the bond between the metal and the hydrogen/alkyl carbon bond to form the two new bonds with the α-carbon and β-carbon (respectively) is slower, thus decreasing the rate of the insertion reaction.Insertion of alkenes into M–H bonds
The insertion of alkenes into metal-hydrogen bonds is a key step inhydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a Catalysis, catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or S ...
and hydroformylation
Hydroformylation, also known as oxo synthesis or oxo process, is an industrial process for the production of aldehydes from alkenes. This chemical reaction entails the net addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon d ...
reactions. The reaction involves the alkene and the hydride ligands combining within the coordination sphere of a catalyst. In hydrogenation, the resulting alkyl ligand combines with a second hydride to give the alkane. Analogous reactions apply to the hydrogenation of alkynes: an alkenyl ligand combines with a hydride to eliminate an alkene.

Mechanism
In terms of mechanism, the insertion of alkenes into M–H bond and into M–C bonds are described similarly. Both involve four-membered transition states that place the less substituted carbon on the metal. The reverse of olefin insertion into a metal-hydrogen bond is β-hydride elimination. The Principle of Microscopic Reversibility requires that the mechanism of β-hydride elimination follow the same pathway as the insertion of alkenes into metal hydride bonds. The first requirement for β-hydride elimination is the presence of a hydrogen at a position that is β with respect to the metal. β-elimination requires a vacant coordination position on the metal that will accommodate the hydrogen that is abstracted.Industrial applications
Carbonylation
Two widely employed applications of migratory insertion of carbonyl groups arehydroformylation
Hydroformylation, also known as oxo synthesis or oxo process, is an industrial process for the production of aldehydes from alkenes. This chemical reaction entails the net addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon d ...
and the production of acetic acid
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula (also written as , , or ). Vinegar is at least 4% acetic acid by volume, making acetic acid the main component ...
by carbonylation
Carbonylation refers to reactions that introduce carbon monoxide into organic and inorganic substrates. Carbon monoxide is abundantly available and conveniently reactive, so it is widely used as a reactant in industrial chemistry. The term carbon ...
of methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical and the simplest aliphatic alcohol, with the formula C H3 O H (a methyl group linked to a hydroxyl group, often abbreviated as MeOH). It is a ...
. The former converts alkenes, hydrogen, and carbon monoxide into aldehydes. The production of acetic acid by carbonylation proceeds via two similar industrial processes. More traditional is the Monsanto acetic acid process, which relies on a rhodium-iodine catalyst to transform methanol into acetic acid. This process has been superseded by the Cativa process
The Cativa process is a method for the production of acetic acid by the carbonylation of methanol. The technology, which is similar to the Monsanto process, was developed by BP Chemicals and is under license by BP Plc. The process is based on an ...
which uses a related iridium catalyst, r(CO)2I2sup>− (1). By 2002, worldwide annual production of acetic acid stood at 6 million tons, of which approximately 60% is produced by the Cativa process.
: The Cativa process
The Cativa process catalytic cycle
In chemistry, a catalytic cycle is a multistep reaction mechanism that involves a catalyst. The catalytic cycle is the main method for describing the role of catalysts in biochemistry, organometallic chemistry, bioinorganic chemistry, materials s ...
, shown above, includes both insertion and de-insertion steps. The oxidative addition reaction of methyl iodide with (1) involves the formal insertion of the iridium(I) centre into the carbon-iodine bond, whilst step (3) to (4) is an example of migratory insertion of carbon monoxide into the iridium-carbon bond. The active catalyst species is regenerated by the reductive elimination
Reductive elimination is an elementary step in organometallic chemistry in which the oxidation state of the metal center decreases while forming a new covalent bond between two ligands. It is the microscopic reverse of oxidative addition, and is ...
of acetyl iodide
Acetyl iodide is an organoiodine compound with the formula CH3COI. It is a colourless liquid. It is formally derived from acetic acid. Although far rarer in the laboratory than the related acetyl bromide and acetyl chloride, acetyl iodide is p ...
from (4), a de-insertion reaction.
Alkene polymerization
Industrial applications of alkene insertions include metal-catalyzed routes to polyethylene and polypropylene. Typically these conversions are catalyzed heterogeneously by titanium trichloride which are activated by aluminium alkyls. This technology is known asZiegler–Natta catalyst A Ziegler–Natta catalyst, named after Karl Ziegler and Giulio Natta, is a catalyst used in the synthesis of polymers of 1-alkenes ( alpha-olefins). Two broad classes of Ziegler–Natta catalysts are employed, distinguished by their solubility:
* ...
s. In these reactions, ethylene coordinates to titanium metal followed by its insertion. These steps can be repeated multiple times, potentially leading to high molecular weight polymers.
References
External links
Migratory Insertion
{{DEFAULTSORT:Migratory Insertion Organometallic chemistry Reaction mechanisms