Electronvolt on:
[Wikipedia]
[Google]
[Amazon]
In

 The energy ''E'', frequency ''v'', and wavelength ''λ'' of a photon are related by
where ''h'' is the
The energy ''E'', frequency ''v'', and wavelength ''λ'' of a photon are related by
where ''h'' is the

BIPM's definition of the electronvoltphysical constants reference; CODATA data
{{DEFAULTSORT:Electron Volt Particle physics Units of chemical measurement Units of energy Voltage
physics
Physics is the natural science that studies matter, its fundamental constituents, its motion and behavior through space and time, and the related entities of energy and force. "Physical science is that department of knowledge which r ...
, an electronvolt (symbol eV, also written electron-volt and electron volt) is the measure of an amount of kinetic energy
In physics, the kinetic energy of an object is the energy that it possesses due to its motion.
It is defined as the work needed to accelerate a body of a given mass from rest to its stated velocity. Having gained this energy during its acc ...
gained by a single electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
accelerating from rest through an electric potential difference of one volt
The volt (symbol: V) is the unit of electric potential, electric potential difference (voltage), and electromotive force in the International System of Units (SI). It is named after the Italian physicist Alessandro Volta (1745–1827).
Defin ...
in vacuum
A vacuum is a space devoid of matter. The word is derived from the Latin adjective ''vacuus'' for "vacant" or " void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often di ...
. When used as a unit of energy
Energy is defined via work, so the SI unit of energy is the same as the unit of work – the joule (J), named in honour of James Prescott Joule and his experiments on the mechanical equivalent of heat. In slightly more fundamental terms, is ...
, the numerical value of 1 eV in joule
The joule ( , ; symbol: J) is the unit of energy in the International System of Units (SI). It is equal to the amount of work done when a force of 1 newton displaces a mass through a distance of 1 metre in the direction of the force applie ...
s (symbol J) is equivalent to the numerical value of the charge
Charge or charged may refer to:
Arts, entertainment, and media Films
* '' Charge, Zero Emissions/Maximum Speed'', a 2011 documentary
Music
* ''Charge'' (David Ford album)
* ''Charge'' (Machel Montano album)
* ''Charge!!'', an album by The Aqu ...
of an electron in coulombs (symbol C). Under the 2019 redefinition of the SI base units
In 2019, four of the seven SI base units specified in the International System of Quantities were redefined in terms of natural physical constants, rather than human artifacts such as the standard kilogram.
Effective 20 May 2019, the 144th ...
, this sets 1 eV equal to the exact value
Historically, the electronvolt was devised as a standard unit of measure through its usefulness in electrostatic particle accelerator
An electrostatic particle accelerator is a particle accelerator in which charged particles are accelerated to a high energy by a static high voltage potential. This contrasts with the other major category of particle accelerator, oscillating fie ...
sciences, because a particle with electric charge
Electric charge is the physical property of matter that causes charged matter to experience a force when placed in an electromagnetic field. Electric charge can be ''positive'' or ''negative'' (commonly carried by protons and electrons respe ...
''q'' gains an energy after passing through a voltage of ''V.'' Since ''q'' must be an integer
An integer is the number zero (), a positive natural number (, , , etc.) or a negative integer with a minus sign ( −1, −2, −3, etc.). The negative numbers are the additive inverses of the corresponding positive numbers. In the languag ...
multiple of the elementary charge ''e'' for any isolated particle, the gained energy in units of electronvolts conveniently equals that integer times the voltage.
It is a common unit of energy
Energy is defined via work, so the SI unit of energy is the same as the unit of work – the joule (J), named in honour of James Prescott Joule and his experiments on the mechanical equivalent of heat. In slightly more fundamental terms, is ...
within physics, widely used in solid state, atomic, nuclear, and particle physics
Particle physics or high energy physics is the study of fundamental particles and forces that constitute matter and radiation. The fundamental particles in the universe are classified in the Standard Model as fermions (matter particles) an ...
, and high-energy astrophysics. It is commonly used with SI prefixes milli-, kilo-, mega-, giga-, tera-, peta- or exa- (meV, keV, MeV, GeV, TeV, PeV and EeV respectively). In some older documents, and in the name Bevatron
The Bevatron was a particle accelerator — specifically, a weak-focusing proton synchrotron — at Lawrence Berkeley National Laboratory, U.S., which began operating in 1954. The antiproton was discovered there in 1955, resulting in ...
, the symbol BeV is used, which stands for billion
Billion is a word for a large number, and it has two distinct definitions:
*1,000,000,000, i.e. one thousand million, or (ten to the ninth power), as defined on the short scale. This is its only current meaning in English.
* 1,000,000,000,000, i. ...
(109) electronvolts; it is equivalent to the GeV.
Definition
An electronvolt is the amount of kinetic energy gained or lost by a singleelectron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
accelerating from rest through an electric potential difference of one volt
The volt (symbol: V) is the unit of electric potential, electric potential difference (voltage), and electromotive force in the International System of Units (SI). It is named after the Italian physicist Alessandro Volta (1745–1827).
Defin ...
in vacuum. Hence, it has a value of one volt
The volt (symbol: V) is the unit of electric potential, electric potential difference (voltage), and electromotive force in the International System of Units (SI). It is named after the Italian physicist Alessandro Volta (1745–1827).
Defin ...
, , multiplied by the elementary charge Therefore, one electronvolt is equal to
The electronvolt (eV) is a unit of energy, but is not an SI unit
The International System of Units, known by the international abbreviation SI in all languages and sometimes pleonastically as the SI system, is the modern form of the metric system and the world's most widely used system of measurement. ...
. The SI unit of energy is the joule (J).
Relation to other physical properties and units
Mass
By mass–energy equivalence, the electronvolt corresponds to a unit ofmass
Mass is an intrinsic property of a body. It was traditionally believed to be related to the quantity of matter in a physical body, until the discovery of the atom and particle physics. It was found that different atoms and different eleme ...
. It is common in particle physics
Particle physics or high energy physics is the study of fundamental particles and forces that constitute matter and radiation. The fundamental particles in the universe are classified in the Standard Model as fermions (matter particles) an ...
, where units of mass and energy are often interchanged, to express mass in units of eV/''c''2, where ''c'' is the speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
in vacuum (from ). It is common to informally express mass in terms of eV as a unit of mass, effectively using a system of natural units
In physics, natural units are physical units of measurement in which only universal physical constants are used as defining constants, such that each of these constants acts as a coherent unit of a quantity. For example, the elementary charge ma ...
with ''c'' set to 1. The kilogram equivalent of is:
For example, an electron and a positron, each with a mass of , can annihilate to yield of energy. A proton has a mass of . In general, the masses of all hadron
In particle physics, a hadron (; grc, ἁδρός, hadrós; "stout, thick") is a composite subatomic particle made of two or more quarks held together by the strong interaction. They are analogous to molecules that are held together by the e ...
s are of the order of , which makes the GeV/''c''2 a convenient unit of mass for particle physics:
The atomic mass constant (''m''u), one twelfth of the mass a carbon-12 atom, is close to the mass of a proton. To convert to electronvolt mass-equivalent, use the formula:
Momentum
By dividing a particle's kinetic energy in electronvolts by the fundamental constant ''c'' (the speed of light), one can describe the particle's momentum in units of eV/''c''. In natural units in which the fundamental velocity constant ''c'' is numerically 1, the ''c'' may informally be omitted to express momentum as electronvolts. The energy momentum relation in natural units (with ) is a Pythagorean equation. When a relatively high energy is applied to a particle with relatively lowrest mass
The invariant mass, rest mass, intrinsic mass, proper mass, or in the case of bound systems simply mass, is the portion of the total mass of an object or system of objects that is independent of the overall motion of the system. More precisely, i ...
, it can be approximated as in high-energy physics such that an applied energy in units of eV conveniently results in an approximately equivalent change of momentum in units of eV/''c''.
The dimensions of momentum units are . The dimensions of energy units are . Dividing the units of energy (such as eV) by a fundamental constant (such as the speed of light) that has units of velocity () facilitates the required conversion for using energy units to describe momentum.
For example, if the momentum ''p'' of an electron is said to be , then the conversion to MKS system of units can be achieved by:
Distance
Inparticle physics
Particle physics or high energy physics is the study of fundamental particles and forces that constitute matter and radiation. The fundamental particles in the universe are classified in the Standard Model as fermions (matter particles) an ...
, a system of natural units in which the speed of light in vacuum ''c'' and the reduced Planck constant
The Planck constant, or Planck's constant, is a fundamental physical constant of foundational importance in quantum mechanics. The constant gives the relationship between the energy of a photon and its frequency, and by the mass-energy equivalen ...
''ħ'' are dimensionless and equal to unity is widely used: . In these units, both distances and times are expressed in inverse energy units (while energy and mass are expressed in the same units, see mass–energy equivalence). In particular, particle scattering length The scattering length in quantum mechanics describes low-energy scattering. For potentials that decay faster than 1/r^3 as r\to \infty, it is defined as the following low-energy limit (mathematics), limit:
:
\lim_ k\cot\delta(k) =- \frac\;,
wher ...
s are often presented in units of inverse particle masses.
Outside this system of units, the conversion factors between electronvolt, second, and nanometer are the following:
:
The above relations also allow expressing the mean lifetime ''τ'' of an unstable particle (in seconds) in terms of its decay width
Decay may refer to:
Science and technology
* Bit decay, in computing
* Software decay, in computing
* Distance decay, in geography
* Decay time (fall time), in electronics
Biology
* Decomposition of organic matter
* Tooth decay (dental caries ...
Γ (in eV) via . For example, the meson has a lifetime of 1.530(9) picoseconds, mean decay length is , or a decay width of .
Conversely, the tiny meson mass differences responsible for meson oscillations are often expressed in the more convenient inverse picoseconds.
Energy in electronvolts is sometimes expressed through the wavelength of light with photons of the same energy:
:
Temperature
In certain fields, such as , it is convenient to use the electronvolt to express temperature. The electronvolt is divided by theBoltzmann constant
The Boltzmann constant ( or ) is the proportionality factor that relates the average relative kinetic energy of particles in a gas with the thermodynamic temperature of the gas. It occurs in the definitions of the kelvin and the gas constant, ...
to convert to the Kelvin scale:
:
where ''k''B is the Boltzmann constant
The Boltzmann constant ( or ) is the proportionality factor that relates the average relative kinetic energy of particles in a gas with the thermodynamic temperature of the gas. It occurs in the definitions of the kelvin and the gas constant, ...
.
The ''k''B is assumed when using the electronvolt to express temperature, for example, a typical magnetic confinement fusion
Magnetic confinement fusion is an approach to generate thermonuclear fusion power that uses magnetic fields to confine fusion fuel in the form of a plasma. Magnetic confinement is one of two major branches of fusion energy research, along with ...
plasma is (kiloelectronvolt), which is equal to 174 MK (megakelvin).
As an approximation: ''k''B''T'' is about (≈ ) at a temperature of .
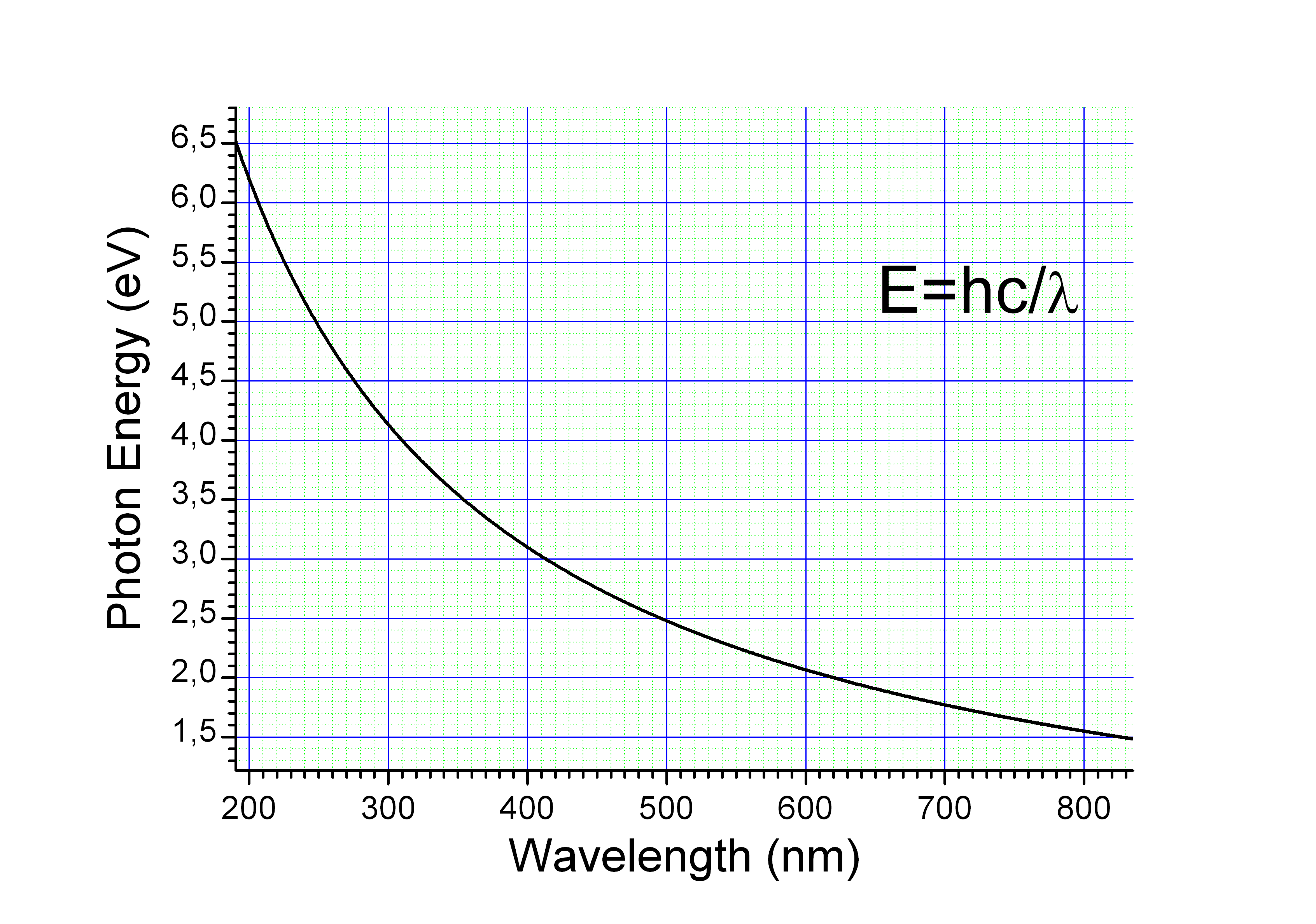
Wavelength
 The energy ''E'', frequency ''v'', and wavelength ''λ'' of a photon are related by
where ''h'' is the
The energy ''E'', frequency ''v'', and wavelength ''λ'' of a photon are related by
where ''h'' is the Planck constant
The Planck constant, or Planck's constant, is a fundamental physical constant of foundational importance in quantum mechanics. The constant gives the relationship between the energy of a photon and its frequency, and by the mass-energy equivale ...
, ''c'' is the speed of light
The speed of light in vacuum, commonly denoted , is a universal physical constant that is important in many areas of physics. The speed of light is exactly equal to ). According to the special theory of relativity, is the upper limit ...
. This reduces to
A photon with a wavelength of (green light) would have an energy of approximately . Similarly, would correspond to an infrared photon of wavelength or frequency .
Scattering experiments
In a low-energy nuclear scattering experiment, it is conventional to refer to the nuclear recoil energy in units of eVr, keVr, etc. This distinguishes the nuclear recoil energy from the "electron equivalent" recoil energy (eVee, keVee, etc.) measured by scintillation light. For example, the yield of aphototube
A phototube or photoelectric cell is a type of gas-filled or vacuum tube that is sensitive to light. Such a tube is more correctly called a 'photoemissive cell' to distinguish it from photovoltaic or photoconductive cells. Phototubes were previ ...
is measured in phe/keVee (photoelectron
The photoelectric effect is the emission of electrons when electromagnetic radiation, such as light, hits a material. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, and solid sta ...
s per keV electron-equivalent energy). The relationship between eV, eVr, and eVee depends on the medium the scattering takes place in, and must be established empirically for each material.
Energy comparisons
Per mole
One mole of particles given 1 eV of energy each has approximately 96.5 kJ of energy – this corresponds to theFaraday constant
In physical chemistry, the Faraday constant, denoted by the symbol and sometimes stylized as ℱ, is the electric charge per mole of elementary charges. It is named after the English scientist Michael Faraday. Since the 2019 redefinition of ...
(''F'' ≈ ), where the energy in joules of ''n'' moles of particles each with energy ''E'' eV is equal to ''E''·''F''·''n''.
See also
*Orders of magnitude (energy)
This list compares various energies in joules (J), organized by order of magnitude.
Below 1 J
1 to 105 J
106 to 1011 J
1012 to 1017 J
1018 to 1023 J
Over 1023 J
}
, -
, 1050
,
, ≳1050 J
, Upper limit of 'apparent'/isotropic energy ...
References
External links
BIPM's definition of the electronvolt
{{DEFAULTSORT:Electron Volt Particle physics Units of chemical measurement Units of energy Voltage