Enamine Alkylation Via SN2 Reaction With A Bromo-alkane on:
[Wikipedia]
[Google]
[Amazon]
 An enamine is an
An enamine is an  The word "enamine" is derived from the affix ''en''-, used as the suffix of
The word "enamine" is derived from the affix ''en''-, used as the suffix of 


 There are many ways to modulate enamine reactivity in addition to altering the steric/electronics at the nitrogen center including changing temperature, solvent, amounts of other reagents, and type of electrophile. Tuning these parameters allows for the preferential formation of E/Z enamines and also affects the formation of the more/less substituted enamine from the ketone starting material.
There are many ways to modulate enamine reactivity in addition to altering the steric/electronics at the nitrogen center including changing temperature, solvent, amounts of other reagents, and type of electrophile. Tuning these parameters allows for the preferential formation of E/Z enamines and also affects the formation of the more/less substituted enamine from the ketone starting material.
 An enamine is an
An enamine is an unsaturated compound
In chemistry, a saturated compound is a chemical compound (or ion) that resists the addition reactions, such as hydrogenation, oxidative addition, and binding of a Lewis base. The term is used in many contexts and for many classes of chemical c ...
derived by the condensation of an aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
or ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bo ...
with a secondary amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituen ...
. Enamines are versatile intermediates.
: The word "enamine" is derived from the affix ''en''-, used as the suffix of
The word "enamine" is derived from the affix ''en''-, used as the suffix of alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
, and the root ''amine''. This can be compared with enol
In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene ( olefin) with a hydroxyl group attached to one end of the alkene double bond (). The te ...
, which is a functional group containing both alkene (''en''-) and alcohol
Alcohol most commonly refers to:
* Alcohol (chemistry), an organic compound in which a hydroxyl group is bound to a carbon atom
* Alcohol (drug), an intoxicant found in alcoholic drinks
Alcohol may also refer to:
Chemicals
* Ethanol, one of sev ...
(-''ol''). Enamines are considered to be nitrogen analogs of enols.
If one of the nitrogen substituents is a hydrogen atom, H, it is the tautomer
Tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert.
The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hydr ...
ic form of an imine
In organic chemistry, an imine ( or ) is a functional group or organic compound containing a carbon–nitrogen double bond (). The nitrogen atom can be attached to a hydrogen or an organic group (R). The carbon atom has two additional single bo ...
. This usually will rearrange to the imine; however there are several exceptions (such as aniline
Aniline is an organic compound with the formula C6 H5 NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine
In organic chemistry, an aromatic amine is an organic compound consisting of an aroma ...
). The enamine-imine tautomerism may be considered analogous to the keto-enol tautomerism
In organic chemistry, alkenols (shortened to enols) are a type of reactive structure or intermediate in organic chemistry that is represented as an alkene ( olefin) with a hydroxyl group attached to one end of the alkene double bond (). The te ...
. In both cases, a hydrogen atom switches its location between the heteroatom (oxygen or nitrogen) and the second carbon atom.
Enamines are both good nucleophiles and good bases. Their behavior as carbon-based nucleophiles is explained with reference to the following resonance structures.
:
Formation
Enamines are labile and therefore chemically useful moieties which can be easily produced from commercially available starting reagents. A common route for enamine production is via an acid-catalyzed nucleophilic reaction of ketone or aldehyde species containing anα-hydrogen
In the nomenclature of organic chemistry, a locant is a term to indicate the position of a functional group or substituent within a molecule.
Numeric locants
The International Union of Pure and Applied Chemistry (IUPAC) recommends the use of n ...
with secondary amines. Acid catalysis is not always required, if the pKaH of the reacting amine is sufficiently high (for example, pyrrolidine
Pyrrolidine, also known as tetrahydropyrrole, is an organic compound with the molecular formula (CH2)4NH. It is a cyclic secondary amine, also classified as a saturated heterocycle. It is a colourless liquid that is miscible with water and most ...
, which has a pKaH of 11.26). If the pKaH of the reacting amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituen ...
is low, however, then acid catalysis is required through both the addition and the dehydration steps (common dehydrating agents include MgSO4 and Na2SO4). Primary amines are usually not used for enamine synthesis due to the preferential formation of the more thermodynamically stable imine species. Methyl ketone self-condensation is a side-reaction which can be avoided through the addition of TiCl4 into the reaction mixture (to act as a water scavenger
Scavengers are animals that consume dead organisms that have died from causes other than predation or have been killed by other predators. While scavenging generally refers to carnivores feeding on carrion, it is also a herbivorous feeding b ...
). An example of an aldehyde reacting with a secondary amine to form an enamine via a carbinolamine
In organic chemistry, a hemiaminal (also carbinolamine) is a functional group or type of chemical compound that has a hydroxyl group and an amine attached to the same carbon atom: . R can be hydrogen or an alkyl group. Hemiaminals are intermediat ...
intermediate is shown below:

Reactions
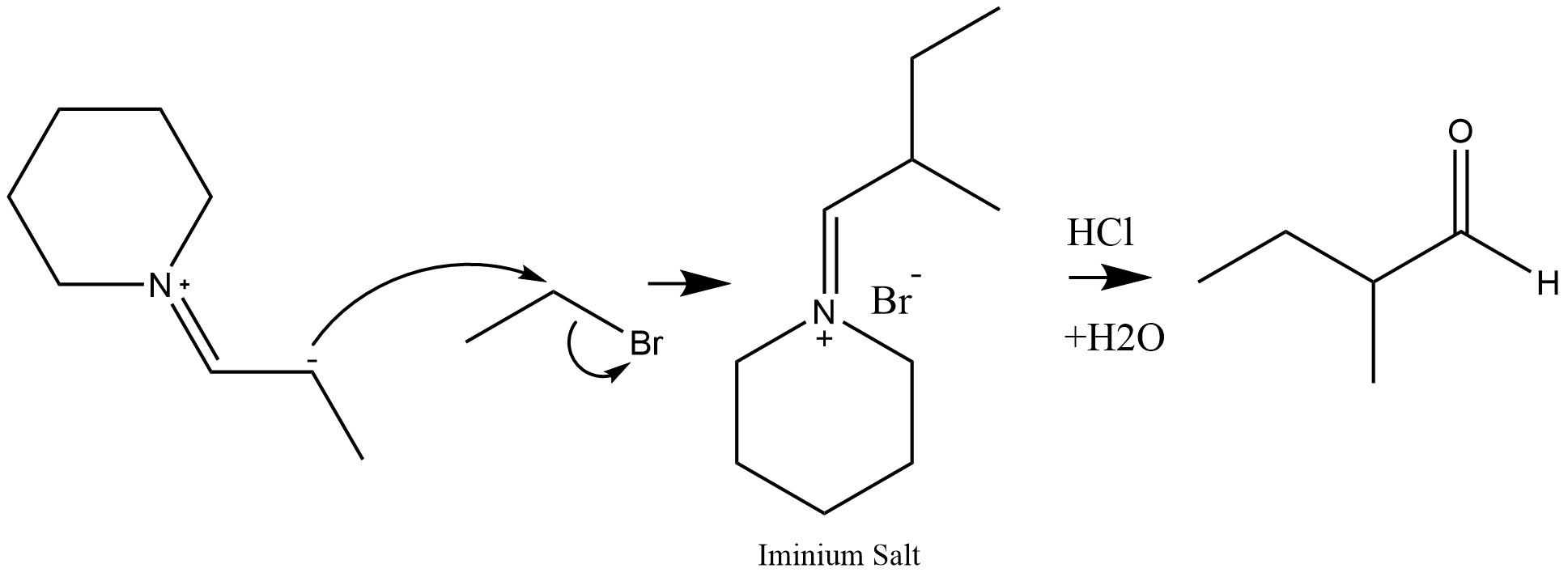
Alkylation
Even though enamines are more nucleophilic than their enol counterparts, they can still react selectively, rendering them useful for alkylation reactions. The enamine nucleophile can attackhaloalkanes
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely us ...
to form the alkylated iminium
In organic chemistry, an iminium cation is a polyatomic ion with the general structure . They are common in synthetic chemistry and biology.
Structure
Iminium cations adopt alkene-like geometries. The central C=N unit is nearly coplanar with al ...
salt intermediate which then hydrolyzes to regenerate a ketone (a starting material in enamine synthesis). This reaction was pioneered by Gilbert Stork
Gilbert Stork (December 31, 1921 – October 21, 2017) was an organic chemist. For a quarter of a century he was the Eugene Higgins Professor of Chemistry Emeritus at Columbia University. He is known for making significant contributions to the t ...
, and is sometimes referred to by the name of its inventor (the Stork enamine alkylation
The Stork enamine alkylation involves the addition of an enamine to a Michael acceptor (e.g, an α,β -unsaturated carbonyl compound) or another electrophilic alkylation reagent to give an alkylated iminium product, which is hydrolyzed by dilute ...
). Analogously, this reaction can be used as an effective means of acylation
In chemistry, acylation (or alkanoylation) is the chemical reaction in which an acyl group () is added to a compound. The compound providing the acyl group is called the acylating agent.
Because they form a strong electrophile when treated with ...
. A variety of alkylating and acylating agents including benzylic, allylic halides can be used in this reaction.

Acylation
In a reaction much similar to the enamine alkylation, enamines can be acylated to form a finaldicarbonyl
In organic chemistry, a dicarbonyl is a molecule containing two carbonyl () groups. Although this term could refer to any organic compound containing two carbonyl groups, it is used more specifically to describe molecules in which both carbonyls ...
product. The enamine starting material undergoes a nucleophilic addition to acyl halides
In organic chemistry, an acyl halide (also known as an acid halide) is a chemical compound derived from an oxoacid by replacing a hydroxyl group () with a halide group (, where X is a halogen).
If the acid is a carboxylic acid (), the compoun ...
forming the iminium salt intermediate which can hydrolyze in the presence of acid.

Metalloenamines
Strong bases such as LiNR2 can be used to deprotonate imines and form metalloenamines. Metalloenamines can prove synthetically useful due to their nucleophilicity (they are more nucleophilic than enolates). Thus they are better able to react with weaker electrophiles (for example, they can be used to openepoxides
In organic chemistry, an epoxide is a cyclic ether () with a three-atom ring. This ring approximates an equilateral triangle, which makes it strained, and hence highly reactive, more so than other ethers. They are produced on a large scale for ...
.) Most prominently, these reactions have allowed for asymmetric alkylations of ketones through transformation to chiral intermediate metalloenamines.
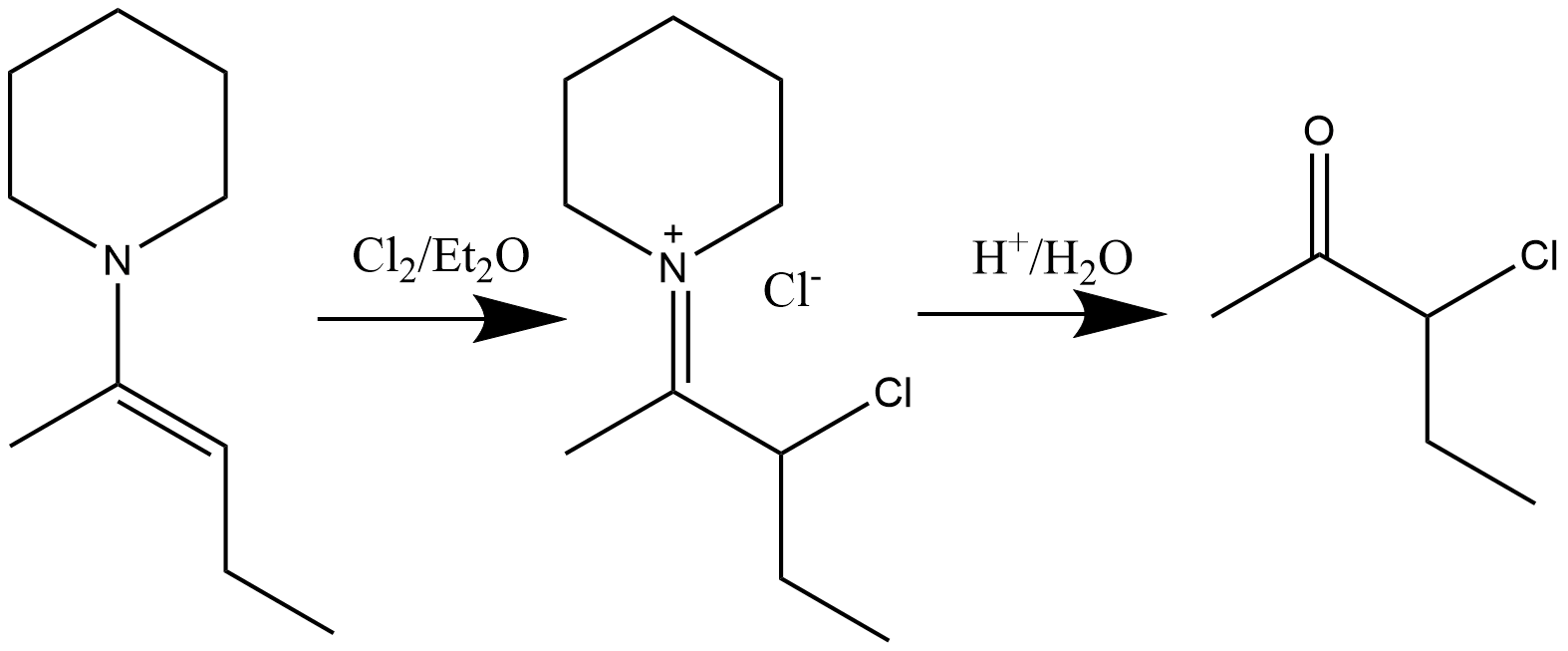
Halogenation
β-halo immonium compounds can be synthesized through the halogenation reaction of enamines with halides indiethyl ether
Diethyl ether, or simply ether, is an organic compound in the ether class with the formula , sometimes abbreviated as (see Pseudoelement symbols). It is a colourless, highly volatile, sweet-smelling ("ethereal odour"), extremely flammable liq ...
solvent. Hydrolysis will result in the formation of α-halo ketones. Chlorination, bromination, and even iodination have been shown to be possible. The general reaction is shown below:

Oxidative coupling
Enamines can be efficiently cross-coupled with enol silanes through treatment withceric ammonium nitrate
Ceric ammonium nitrate (CAN) is the inorganic compound with the formula . This orange-red, water-soluble cerium salt is a specialised oxidizing agent in organic synthesis and a standard oxidant in quantitative analysis.
Preparation, properties, ...
. These reactions were reported by the Narasaka group in 1935, providing a route to stable enamines as well as one instance of a 1,4-diketone (derived from a morpholine
Morpholine is an organic chemical compound having the chemical formula O( C H2CH2)2 NH. This heterocycle features both amine and ether functional groups. Because of the amine, morpholine is a base; its conjugate acid is called morpholinium. Fo ...
amine reagent). Later, these results were exploited by the MacMillan
MacMillan, Macmillan, McMillen or McMillan may refer to:
People
* McMillan (surname)
* Clan MacMillan, a Highland Scottish clan
* Harold Macmillan, British statesman and politician
* James MacMillan, Scottish composer
* William Duncan MacMillan ...
group with the development of an organocatalyst
In organic chemistry, organocatalysis is a form of catalysis in which the rate of a chemical reaction is increased by an Organic compound, organic catalyst. This "organocatalyst" consists of carbon, hydrogen, sulfur and other nonmetal elements fo ...
which used the Narasaka substrates to produce 1,4 dicarbonyls enantioselectively, with good yields. Oxidative dimerization of aldehydes in the presence of amines proceeds through the formation of an enamine followed by a final pyrrole
Pyrrole is a heterocyclic aromatic organic compound, a five-membered ring with the formula C4 H4 NH. It is a colorless volatile liquid that darkens readily upon exposure to air. Substituted derivatives are also called pyrroles, e.g., ''N''-meth ...
formation. This method for symmetric pyrrole synthesis was developed in 2010 by the Jia group, as a valuable new pathway for the synthesis of pyrrole-containing natural products.
Annulation
Enamines chemistry has been implemented for the purposes of producing a one-pot enantioselective version of theRobinson annulation
The Robinson annulation is a chemical reaction used in organic chemistry for ring formation. It was discovered by Robert Robinson (organic chemist), Robert Robinson in 1935 as a method to create a six membered ring by forming three new carbon–ca ...
. The Robinson annulation, published by Robert Robinson in 1935, is a base-catalyzed reaction that combines a ketone and a methyl vinyl ketone
Methyl vinyl ketone (MVK, IUPAC name: butenone) is the organic compound with the formula CH3C(O)CH=CH2. It is a reactive compound classified as an Alpha-beta Unsaturated carbonyl compounds, enone, in fact the simplest example thereof. It is a co ...
(commonly abbreviated to MVK) to form a cyclohexenone
Cyclohexenone is an organic compound which is a versatile intermediate used in the synthesis of a variety of chemical products such as pharmaceuticals and fragrances. It is colorless liquid, but commercial samples are often yellow.
Industrially, ...
fused ring system. This reaction may be catalyzed by proline
Proline (symbol Pro or P) is an organic acid classed as a proteinogenic amino acid (used in the biosynthesis of proteins), although it does not contain the amino group but is rather a secondary amine. The secondary amine nitrogen is in the prot ...
to proceed through chiral enamine intermediates which allow for good stereoselectivity. This is important, in particular in the field of natural product synthesis, for example, for the synthesis of the Wieland-Miescher ketone – a vital building block for more complex biologically active molecules.
Reactivity
Enamines act as nucleophiles that require less acid/base activation for reactivity than their enolate counterparts. They have also been shown to offer a greater selectivity with less side reactions. There is a gradient of reactivity among different enamine types, with a greater reactivity offered by ketone enamines than their aldehyde counterparts. Cyclic ketone enamines follow a reactivity trend where the five membered ring is the most reactive due to its maximally planar conformation at the nitrogen, following the trend 5>8>6>7 (the seven membered ring being the least reactive). This trend has been attributed to the amount of p-character on the nitrogen lone pair orbital - the higher p character corresponding to a greater nucleophilicity because the p-orbital would allow for donation into the alkene π- orbital. Analogously, if the N lone pair participates in stereoelectronic interactions on the amine moiety, the lone pair will pop out of the plane (will pyramidalize) and compromise donation into the adjacent π C-C bond. There are many ways to modulate enamine reactivity in addition to altering the steric/electronics at the nitrogen center including changing temperature, solvent, amounts of other reagents, and type of electrophile. Tuning these parameters allows for the preferential formation of E/Z enamines and also affects the formation of the more/less substituted enamine from the ketone starting material.
There are many ways to modulate enamine reactivity in addition to altering the steric/electronics at the nitrogen center including changing temperature, solvent, amounts of other reagents, and type of electrophile. Tuning these parameters allows for the preferential formation of E/Z enamines and also affects the formation of the more/less substituted enamine from the ketone starting material.
See also
* Enders SAMP/RAMP hydrazone-alkylation reaction *Hajos–Parrish–Eder–Sauer–Wiechert reaction The Hajos–Parrish–Eder–Sauer–Wiechert reaction in organic chemistry is a proline catalysed asymmetric aldol reaction. The reaction is named after its principal investigators, Zoltan Hajos others, from Hoffmann-La RocheZ. G. Hajos, D. R. P ...
*Michael Addition
In organic chemistry, the Michael reaction or Michael addition is a reaction between a Michael donor (an enolate or other nucleophile) and a Michael acceptor (usually an α,β-unsaturated carbonyl) to produce a Michael adduct by creating a carbon ...
*Nenitzescu indole synthesis
The Nenitzescu indole synthesis is a chemical reaction that forms 5-hydroxyindole derivatives from benzoquinone and β-aminocrotonic esters.
This reaction was named for its discoverer, Costin Nenițescu, who first reported it in 1929. It can b ...
*Organocatalysis
In organic chemistry, organocatalysis is a form of catalysis in which the rate of a chemical reaction is increased by an organic catalyst. This "organocatalyst" consists of carbon, hydrogen, sulfur and other nonmetal elements found in organic com ...
*Robinson annulation
The Robinson annulation is a chemical reaction used in organic chemistry for ring formation. It was discovered by Robert Robinson (organic chemist), Robert Robinson in 1935 as a method to create a six membered ring by forming three new carbon–ca ...
*Thorpe reaction The Thorpe reaction is a chemical reaction described as a self-condensation of aliphatic nitriles catalyzed by base to form enamines. The reaction was discovered by Jocelyn Field Thorpe.
Thorpe–Ziegler reaction
The Thorpe–Ziegler reaction ...
*Fluoxymesterone
Fluoxymesterone, sold under the brand names Halotestin and Ultandren among others, is an androgen and anabolic steroid (AAS) medication which is used in the treatment of low testosterone levels in men, delayed puberty in boys, breast cancer in w ...
References
{{Authority control Functional groups