E1cB Elimination Reaction on:
[Wikipedia]
[Google]
[Amazon]
 The E1cB elimination reaction is a type of
The E1cB elimination reaction is a type of
 There are two main requirements to have a reaction proceed down an E1cB mechanistic pathway. The compound must have an acidic hydrogen on its
There are two main requirements to have a reaction proceed down an E1cB mechanistic pathway. The compound must have an acidic hydrogen on its 
 In an E2-elimination reaction, both the deprotonation of the α-carbon and the loss of the leaving group occur simultaneously in one concerted step. Molecules that undergo E2-elimination mechanisms have more acidic α-carbons than those that undergo E1 mechanisms, but their α-carbons are not as acidic as those of molecules that undergo E1cB mechanisms. The key difference between the E2 vs E1cb pathways is a distinct carbanion intermediate as opposed to one concerted mechanism. Studies have been shown that the pathways differ by using different halogen
In an E2-elimination reaction, both the deprotonation of the α-carbon and the loss of the leaving group occur simultaneously in one concerted step. Molecules that undergo E2-elimination mechanisms have more acidic α-carbons than those that undergo E1 mechanisms, but their α-carbons are not as acidic as those of molecules that undergo E1cB mechanisms. The key difference between the E2 vs E1cb pathways is a distinct carbanion intermediate as opposed to one concerted mechanism. Studies have been shown that the pathways differ by using different halogen
 Assuming that there is a steady-state carbanion concentration in the mechanism, the rate law for an E1cB mechanism.
:
From this equation, it is clear the second order kinetics will be exhibited.
E1cB mechanisms kinetics can vary slightly based on the rate of each step. As a result, the E1cB mechanism can be broken down into three categories:
#E1cBanion is when the carbanion is stable and/or a strong base is used in excess of the substrate, making deprotonation irreversible, followed by rate-determining loss of the leaving group (k1
Assuming that there is a steady-state carbanion concentration in the mechanism, the rate law for an E1cB mechanism.
:
From this equation, it is clear the second order kinetics will be exhibited.
E1cB mechanisms kinetics can vary slightly based on the rate of each step. As a result, the E1cB mechanism can be broken down into three categories:
#E1cBanion is when the carbanion is stable and/or a strong base is used in excess of the substrate, making deprotonation irreversible, followed by rate-determining loss of the leaving group (k1
If the reactant contains deuterium at the β position, a primary kinetic isotope effect indicates that deprotonation is rate determining. Of the three E1cB mechanisms, this result is only consistent with the E1cBirr mechanism, since the isotope is already removed in E1cBanion and leaving group departure is rate determining in E1cBrev.
 The E1cB elimination reaction is a type of
The E1cB elimination reaction is a type of elimination reaction
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 r ...
which occurs under basic conditions, where the hydrogen to be removed is relatively acidic, while the leaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
(such as -OH or -OR) is a relatively poor one. Usually a moderate to strong base is present. E1cB is a two-step process, the first step of which may or may not be reversible. First, a base abstracts the relatively acidic proton to generate a stabilized anion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
. The lone pair of electrons on the anion then moves to the neighboring atom, thus expelling the leaving group and forming double
A double is a look-alike or doppelgänger; one person or being that resembles another.
Double, The Double or Dubble may also refer to:
Film and television
* Double (filmmaking), someone who substitutes for the credited actor of a character
* ...
or triple bond
A triple bond in chemistry is a chemical bond between two atoms involving six bonding electrons instead of the usual two in a covalent single bond. Triple bonds are stronger than the equivalent single bonds or double bonds, with a bond order o ...
. The name of the mechanism - E1cB - stands for Elimination Unimolecular conjugate Base. ''Elimination'' refers to the fact that the mechanism is an elimination reaction
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 r ...
and will lose two substituents. ''Unimolecular'' refers to the fact that the rate-determining step of this reaction only involves one molecular entity A molecular entity, or chemical entity, is "any constitutionally or isotopically distinct atom, molecule, ion, ion pair, radical, radical ion, complex, conformer, etc., identifiable as a separately distinguishable entity".{{GoldBookRef, title=molec ...
. Finally, ''conjugate base
A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid donates a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as in the reverse reaction it loses a ...
'' refers to the formation of the carbanion intermediate, which is the conjugate base of the starting material.
E1cB should be thought of as being on one end of a continuous spectrum, which includes the E1 mechanism at the opposite end and the E2 mechanism in the middle. The E1 mechanism usually has the opposite characteristics: the leaving group is a good one (like -OTs or -Br), while the hydrogen is not particularly acidic and a strong base is absent. Thus, in the E1 mechanism, the leaving group leaves first to generate a carbocation. Due to the presence of an empty p orbital after departure of the leaving group, the hydrogen on the neighboring carbon becomes much more acidic, allowing it to then be removed by the weak base in the second step. In an E2 reaction, the presence of a strong base and a good leaving group allows proton abstraction by the base and the departure of the leaving group to occur simultaneously, leading to a concerted transition state in a one-step process.
Mechanism
β-carbon
In the nomenclature of organic chemistry, a locant is a term to indicate the position of a functional group or substituent within a molecule.
Numeric locants
The International Union of Pure and Applied Chemistry (IUPAC) recommends the use of n ...
and a relatively poor leaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
on the α- carbon.
The first step of an E1cB mechanism is the deprotonation of the β-carbon, resulting in the formation of an anion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
ic transition state, such as a carbanion. The greater the stability of this transition state, the more the mechanism will favor an E1cB mechanism. This transition state can be stabilized through induction
Induction, Inducible or Inductive may refer to:
Biology and medicine
* Labor induction (birth/pregnancy)
* Induction chemotherapy, in medicine
* Induced stem cells, stem cells derived from somatic, reproductive, pluripotent or other cell t ...
or delocalization
In chemistry, delocalized electrons are electrons in a molecule, ion or solid metal that are not associated with a single atom or a covalent bond.IUPAC Gold Boo''delocalization''/ref>
The term delocalization is general and can have slightly dif ...
of the electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have no ...
lone pair through resonance
Resonance describes the phenomenon of increased amplitude that occurs when the frequency of an applied Periodic function, periodic force (or a Fourier analysis, Fourier component of it) is equal or close to a natural frequency of the system ...
. In general it can be claimed that an electron withdrawing group on the substrate, a strong base, a poor leaving group and a polar solvent triggers the E1cB mechanism. An example of an E1cB mechanism that has a stable transition state can be seen in the degradation of ethiofencarb
Ethiofencarb is a carbamate insecticide which is useful in controlling aphids on hard and soft fruits and some vegetables. It is not as dangerous as organophosphorous pesticides, but is considered highly toxic to humans in the UK, moderately toxi ...
- a carbamate insecticide that has a relatively short half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable at ...
in earth's atmosphere. Upon deprotonation of the amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen
Hydrogen is the chemical element wi ...
, the resulting amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it i ...
is relatively stable because it is conjugated with the neighboring carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
.
In addition to containing an acidic hydrogen on the β-carbon, a relatively poor leaving group is also necessary. A bad leaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
is necessary because a good leaving group will leave before the ionization
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecul ...
of the molecule. As a result, the compound will likely proceed through an E2 pathway. Some examples of compounds that contain poor leaving groups and can undergo the E1cB mechanism are alcohols and fluoroalkanes.
It has also been suggested that the E1cB mechanism is more common among alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s eliminating to alkynes than from an alkane to alkene. One possible explanation for this is that the sp2 hybridization creates slightly more acidic protons. Although this mechanism is not limited to carbon-based eliminations. It has been observed with other heteroatom
In chemistry, a heteroatom () is, strictly, any atom that is not carbon or hydrogen.
Organic chemistry
In practice, the term is usually used more specifically to indicate that non-carbon atoms have replaced carbon in the backbone of the molecula ...
s, such as nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
in the elimination of a phenol
Phenol (also called carbolic acid) is an aromatic organic compound with the molecular formula . It is a white crystalline solid that is volatile. The molecule consists of a phenyl group () bonded to a hydroxy group (). Mildly acidic, it ...
derivative from ethiofencarb
Ethiofencarb is a carbamate insecticide which is useful in controlling aphids on hard and soft fruits and some vegetables. It is not as dangerous as organophosphorous pesticides, but is considered highly toxic to humans in the UK, moderately toxi ...
.
Distinguishing E1cB-elimination reactions from E1- and E2-elimination reactions
All elimination reactions involve the removal of two substituents from a pair of atoms in a compound. Alkene, alkynes, or similar heteroatom variations (such ascarbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
and cyano
Cyanide is a naturally occurring, rapidly acting, toxic chemical that can exist in many different forms.
In chemistry, a cyanide () is a chemical compound that contains a functional group. This group, known as the cyano group, consists of a ...
) will form. The E1cB mechanism is just one of three types of elimination reaction. The other two elimination reactions are E1 and E2 reactions. Although the mechanisms are similar, they vary in the timing of the deprotonation of the α-carbon and the loss of the leaving group. E1 stands for unimolecular elimination, and E2 stands for bimolecular elimination.
In an E1 mechanism, the molecule contains a good leaving group that departs before deprotonation of the α-carbon. This results in the formation of a carbocation intermediate. The carbocation is then deprotonated resulting in the formation of a new pi bond. The molecule involved must also have a very good leaving group such as bromine or chlorine, and it should have a relatively less acidic α-carbon.
 In an E2-elimination reaction, both the deprotonation of the α-carbon and the loss of the leaving group occur simultaneously in one concerted step. Molecules that undergo E2-elimination mechanisms have more acidic α-carbons than those that undergo E1 mechanisms, but their α-carbons are not as acidic as those of molecules that undergo E1cB mechanisms. The key difference between the E2 vs E1cb pathways is a distinct carbanion intermediate as opposed to one concerted mechanism. Studies have been shown that the pathways differ by using different halogen
In an E2-elimination reaction, both the deprotonation of the α-carbon and the loss of the leaving group occur simultaneously in one concerted step. Molecules that undergo E2-elimination mechanisms have more acidic α-carbons than those that undergo E1 mechanisms, but their α-carbons are not as acidic as those of molecules that undergo E1cB mechanisms. The key difference between the E2 vs E1cb pathways is a distinct carbanion intermediate as opposed to one concerted mechanism. Studies have been shown that the pathways differ by using different halogen leaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
s. One example uses chlorine
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine i ...
as a better stabilizing halogen for the anion
An ion () is an atom or molecule with a net electrical charge.
The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conve ...
than fluorine, which makes fluorine the leaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
even though chlorine is a much better leaving group. This provides evidence that the carbanion is formed because the products are not possible through the most stable concerted E2 mechanism.
The following table summarizes the key differences between the three elimination reactions; however, the best way to identify which mechanism is playing a key role in a particular reaction involves the application of chemical kinetics
Chemical kinetics, also known as reaction kinetics, is the branch of physical chemistry that is concerned with understanding the rates of chemical reactions. It is to be contrasted with chemical thermodynamics, which deals with the direction in ...
.
Chemical kinetics of E1cB-elimination mechanisms
When trying to determine whether or not a reaction follows the E1cB mechanism,chemical kinetics
Chemical kinetics, also known as reaction kinetics, is the branch of physical chemistry that is concerned with understanding the rates of chemical reactions. It is to be contrasted with chemical thermodynamics, which deals with the direction in ...
are essential. The best way to identify the E1cB mechanism involves the use of rate laws and the kinetic isotope effect
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for th ...
. These techniques can also help further differentiate between E1cB, E1, and E2-elimination reactions.
Rate law
When trying to experimentally determine whether or not a reaction follows the E1cB mechanism,chemical kinetics
Chemical kinetics, also known as reaction kinetics, is the branch of physical chemistry that is concerned with understanding the rates of chemical reactions. It is to be contrasted with chemical thermodynamics, which deals with the direction in ...
are essential. The best ways to identify the E1cB mechanism involves the use of rate laws and the kinetic isotope effect.
The rate law that governs E1cB mechanisms is relatively simple to determine. Consider the following reaction scheme.
ase
Ase may refer to:
* Ase, Nigeria, a town in Delta State, Nigeria
* -ase, a suffix used for the names of enzymes
* Aṣẹ, a West African philosophical concept
* American Sign Language (ISO 639-3 code: ase)
See also
* Åse (disambiguation)
* ...
≫ k2).
#E1cBrev is when the first step is reversible but the formation of product is slower than reforming the starting material, this again results from a slow second step (k−1 onjugate acid≫ k2).
#E1cBirr is when the first step is slow, but once the anion is formed the product quickly follows (k2 ≫ k−1 onjugate acid. This leads to an irreversible first step but unlike E1cBanion, deprotonation is rate determining.
Kinetic isotope effect
Deuterium
Deuterium exchange and a deuteriumkinetic isotope effect
In physical organic chemistry, a kinetic isotope effect (KIE) is the change in the reaction rate of a chemical reaction when one of the atoms in the reactants is replaced by one of its isotopes. Formally, it is the ratio of rate constants for th ...
can help distinguish among E1cBrev, E1cBanion, and E1cBirr. If the solvent is protic and contains deuterium
Deuterium (or hydrogen-2, symbol or deuterium, also known as heavy hydrogen) is one of two stable isotopes of hydrogen (the other being protium, or hydrogen-1). The nucleus of a deuterium atom, called a deuteron, contains one proton and one ...
in place of hydrogen (e.g., CH3OD), then the exchange of protons into the starting material can be monitored. If the recovered starting material contains deuterium, then the reaction is most likely undergoing an E1cBrev type mechanism. Recall, in this mechanism protonation of the carbanion (either by the conjugate acid or by solvent) is faster than loss of the leaving group. This means after the carbanion is formed, it will quickly remove a proton from the solvent to form the starting material.If the reactant contains deuterium at the β position, a primary kinetic isotope effect indicates that deprotonation is rate determining. Of the three E1cB mechanisms, this result is only consistent with the E1cBirr mechanism, since the isotope is already removed in E1cBanion and leaving group departure is rate determining in E1cBrev.
Fluorine-19 and carbon-11
Another way that the kinetic isotope effect can help distinguish E1cB mechanisms involves the use of 19F. Fluorine is a relatively poor leaving group, and it is often employed in E1cB mechanisms. Fluorine kinetic isotope effects are also applied in the labeling ofRadiopharmaceuticals
Radiopharmaceuticals, or medicinal radiocompounds, are a group of pharmaceutical drugs containing radioactive isotopes. Radiopharmaceuticals can be used as diagnostic and therapeutic agents. Radiopharmaceuticals emit radiation themselves, which is ...
and other compounds in medical research. This experiment is very useful in determining whether or not the loss of the leaving group is the rate-determining step in the mechanism and can help distinguish between E1cBirr and E2 mechanisms. 11C can also be used to probe the nature of the transition state structure. The use of 11C can be used to study the formation of the carbanion as well as study its lifetime which can not only show that the reaction is a two-step E1cB mechanism (as opposed to the concerted E2 mechanism), but it can also address the lifetime and stability of the transition state structure which can further distinguish between the three different types of E1cB mechanisms.
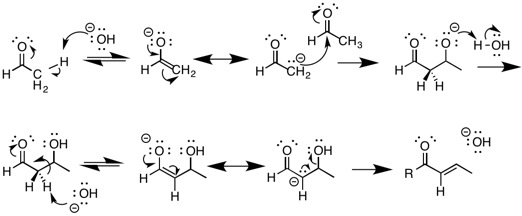
Aldol reactions
The most well known reaction that undergoes E1cB elimination is thealdol condensation
An aldol condensation is a condensation reaction in organic chemistry in which two carbonyl moieties (of aldehydes or ketones) react to form a β-hydroxyaldehyde or β-hydroxyketone (an aldol reaction), and this is then followed by dehydration to ...
reaction under basic conditions. This involves the deprotonation
Deprotonation (or dehydronation) is the removal (transfer) of a proton (or hydron, or hydrogen cation), (H+) from a Brønsted–Lowry acid in an acid–base reaction.Henry Jakubowski, Biochemistry Online Chapter 2A3, https://employees.csbsju. ...
of a compound containing a carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
group that results in the formation of an enolate
In organic chemistry, enolates are organic anions derived from the deprotonation of carbonyl () compounds. Rarely isolated, they are widely used as reagents in the synthesis of organic compounds.
Bonding and structure
Enolate anions are electr ...
. The enolate is the very stable conjugate base
A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid donates a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as in the reverse reaction it loses a ...
of the starting material, and is one of the intermediates in the reaction. This enolate then acts as a nucleophile and can attack an electrophilic aldehyde. The Aldol product is then deprotonated forming another enolate followed by the elimination of water in an E1cB dehydration reaction
In chemistry, a dehydration reaction is a chemical reaction that involves the loss of water from the reacting molecule or ion. Dehydration reactions are common processes, the reverse of a hydration reaction.
Dehydration reactions in organic ch ...
. Aldol reactions are a key reaction in organic chemistry because they provide a means of forming carbon-carbon bonds, allowing for the synthesis of more complex molecules.

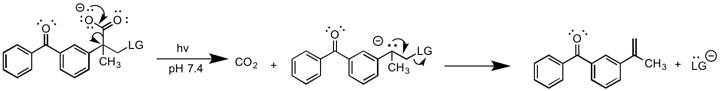
Photo-induced E1cB
A photochemical version of E1cB has been reported by Lukeman ''et al.'' In this report, a photochemically induced decarboxylation reaction generates a carbanion intermediate, which subsequently eliminates the leaving group. The reaction is unique from other forms of E1cB since it does not require a base to generate the carbanion. The carbanion formation step is irreversible, and should thus be classified as E1cBirr.
In biology
The E1cB-elimination reaction is an important reaction in biology. For example, the penultimate step of glycolysis involves an E1cB mechanism. This step involves the conversion of 2-phosphoglycerate tophosphoenolpyruvate
Phosphoenolpyruvate (2-phosphoenolpyruvate, PEP) is the ester derived from the enol of pyruvate and phosphate. It exists as an anion. PEP is an important intermediate in biochemistry. It has the highest-energy phosphate bond found (−61.9 kJ/m ...
, facilitated by the enzyme enolase
Phosphopyruvate hydratase, usually known as enolase, is a metalloenzyme () that catalyses the conversion of 2-phosphoglycerate (2-PG) to phosphoenolpyruvate (PEP), the ninth and penultimate step of glycolysis. The chemical reaction is:
:2-p ...
.
See also
*Elimination reaction
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 r ...
* Reaction mechanism
* Carbocation
* Carbanion
References
{{Organic reactions Elimination reactions Reaction mechanisms