deoxygenation on:
[Wikipedia]
[Google]
[Amazon]
Deoxygenation is a
Link
/ref> For example, in the deoxygenation of the
chemical reaction
A chemical reaction is a process that leads to the chemistry, chemical transformation of one set of chemical substances to another. When chemical reactions occur, the atoms are rearranged and the reaction is accompanied by an Gibbs free energy, ...
involving the removal of oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
atoms from a molecule. The term also refers to the removal of molecular oxygen (O2) from gases and solvents, a step in air-free technique Air-free techniques refer to a range of manipulations in the chemistry laboratory for the handling of compounds that are air-sensitive. These techniques prevent the compounds from reacting with components of air, usually water and oxygen; less com ...
and gas purifiers. As applied to organic compounds, deoxygenation is a component of fuels production as well a type of reaction employed in organic synthesis
Organic synthesis is a branch of chemical synthesis concerned with the construction of organic compounds. Organic compounds are molecules consisting of combinations of covalently-linked hydrogen, carbon, oxygen, and nitrogen atoms. Within the gen ...
, e.g. of pharmaceuticals
Medication (also called medicament, medicine, pharmaceutical drug, medicinal product, medicinal drug or simply drug) is a drug used to diagnose, cure, treat, or prevent disease. Drug therapy ( pharmacotherapy) is an important part of the ...
.
Deoxygenation of C-O bonds
With replacement by H2
The main examples involving the replacement of an oxo group by two hydrogen atoms (A=O → AH2) arehydrogenolysis
Hydrogenolysis is a chemical reaction whereby a carbon–carbon or carbon–heteroatom single bond is cleaved or undergoes lysis (breakdown) by hydrogen.Ralph Connor, Homer Adkins. Hydrogenolysis Of Oxygenated Organic Compounds. J. Am. Chem. Soc. ...
. Typical examples use metal catalysts and H2 as the reagent. Conditions are typically more forcing than hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to redox, reduce or Saturated ...
.
Stoichiometric reactions that effect deoxygenation include the Wolff–Kishner reduction for aryl ketones. The replacement of a hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydroxy ...
group by hydrogen (A-OH → A-H) is the point of the Barton–McCombie deoxygenation and the Markó–Lam deoxygenation.
Biomass valorization
Deoxygenation is an important goal of the conversion of biomass to useful fuels and chemicals. Partial deoxygenation is effected bydehydration
In physiology, dehydration is a lack of total body water that disrupts metabolic processes. It occurs when free water loss exceeds intake, often resulting from excessive sweating, health conditions, or inadequate consumption of water. Mild deh ...
and decarboxylation
Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2). Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain. The reverse process, which is ...
.
Other routes
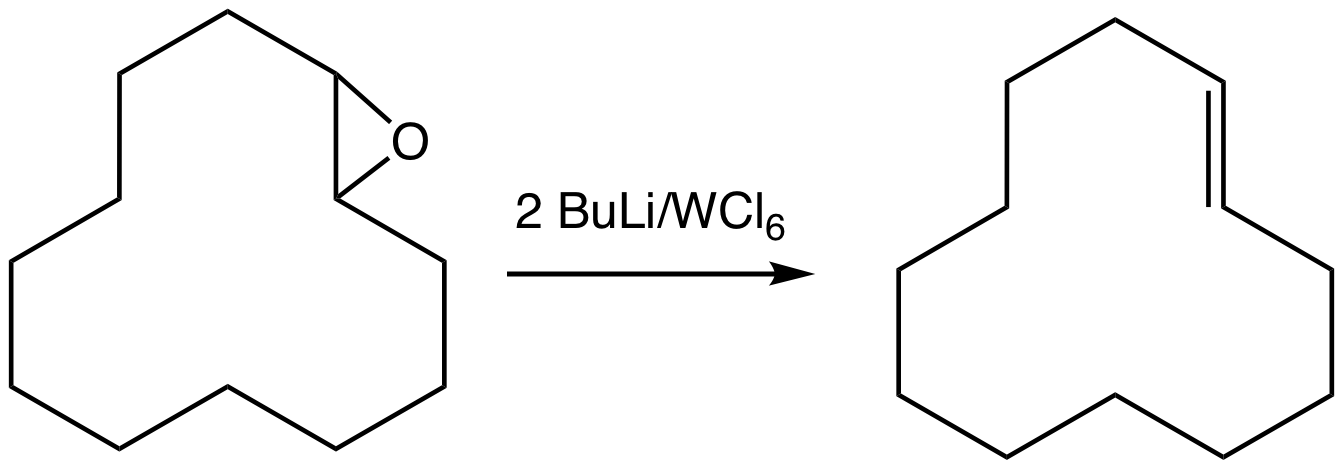
Oxygen groups can also be removed by the reductive coupling of ketones, as illustrated by the McMurry reaction. :Epoxide
In organic chemistry, an epoxide is a cyclic ether, where the ether forms a three-atom ring: two atoms of carbon and one atom of oxygen. This triangular structure has substantial ring strain, making epoxides highly reactive, more so than other ...
s can be deoxygenated using the oxophilic reagent produced by combining tungsten hexachloride
Tungsten hexachloride is an inorganic chemical compound of tungsten and chlorine with the chemical formula . This dark violet-blue compound exists as volatile crystals under standard conditions. It is an important starting reagent in the prepar ...
and ''n''-butyllithium generates the alkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins.
The Internationa ...
. This reaction can proceed with loss or retention of configuration.
:
Deoxygenation of S-O and P-O bonds
N=O bonds
Nitroaromatics are deoxygenated by strongly reducing silyl reagents such as N,N'-bis(trimethylsilyl)-4,4'-bipyridinylidene.P=O bonds
Phosphorus occurs in nature as oxides, so to produce elemental form of the element, deoxygenation is required. The main method involves carbothermic reduction (i.e., carbon is the deoxygenation agent). :4 Ca5(PO4)3F + 18 SiO2 + 30 C → 3 P4 + 30 CO + 18 CaSiO3 + 2 CaF2Oxophilic
Oxophilicity is the tendency of certain chemical compounds to form oxides by hydrolysis or abstraction of an oxygen atom from another molecule, often from organic compounds. The term is often used to describe metal centers, commonly the early trans ...
main group compounds are useful reagents for certain deoxygenations conducted on laboratory scale. The highly oxophilic reagent hexachlorodisilane (Si2Cl6) stereospecifically deoxygenates phosphine oxide
Phosphine oxide is the inorganic compound with the formula H3PO. Although stable as a dilute gas, liquid or solid samples are unstable. Unlike many other compounds of the type POxHy, H3PO is rarely discussed and is not even mentioned in major so ...
s.
S=O bonds
A chemical reagent for the deoxygenation of many sulfur and nitrogen oxo compounds is the combination trifluoroacetic anhydride/sodium iodide
Sodium iodide (chemical formula NaI) is an ionic compound formed from the chemical reaction of sodium metal and iodine. Under standard conditions, it is a white, water-soluble solid comprising a 1:1 mix of sodium cations (Na+) and iodide anions ...
.''Trifluoroacetic anhydride-sodium iodide reagent. Nature and applications'' Arkivoc
''Arkivoc'' (''Archive for Organic Chemistry'') is a peer-reviewed open access scientific journal covering all aspects of organic chemistry. It is published by the non-profit organization Arkat USA, which was established in 2000 through a personal ...
2007 (JE-2136MR) Zbigniew H. Kudzin, Marcin H. Kudzin, Józef Drabowicz, and Andrzej KotyńskLink
/ref> For example, in the deoxygenation of the
sulfoxide
In organic chemistry, a sulfoxide, also called a sulphoxide, is an organosulfur compound containing a sulfinyl () functional group attached to two carbon atoms. It is a polar functional group. Sulfoxides are oxidized derivatives of sulfides. E ...
''diphenylsulfoxide'' to the sulfide
Sulfide (also sulphide in British English) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to large families o ...
''diphenylsulfide'':
:
The reaction mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical reaction occurs.
A chemical mechanism is a theoretical conjecture that tries to describe in detail what takes place at each stage ...
is based on the activation of the sulfoxide by a trifluoroacetyl group and oxidation of iodine. Iodine
Iodine is a chemical element; it has symbol I and atomic number 53. The heaviest of the stable halogens, it exists at standard conditions as a semi-lustrous, non-metallic solid that melts to form a deep violet liquid at , and boils to a vi ...
is formed quantitatively in this reaction and therefore the reagent is used for the analytical detection of many oxo compounds.
:
See also
*Degassing
Degassing, also known as degasification, is the removal of dissolved gases from liquids, especially water or aqueous solutions. There are numerous methods for removing gases from liquids.
Gases are removed for various reasons. Chemists remove gas ...
* Preparation of stable carbenes
* Ocean deoxygenation
Ocean deoxygenation is the reduction of the oxygen content in different parts of the ocean due to human activities. There are two areas where this occurs. Firstly, it occurs in coastal zones where eutrophication has driven some quite rapid (in ...
* Oxophilicity
References
{{Reflist Organic redox reactions Gas technologies