Crystal Structure Of Boron-rich Metal Borides on:
[Wikipedia]
[Google]
[Amazon]

 In metal borides, the bonding of boron varies depending on the atomic ratio B/M. Diborides have B/M = 2, as in the well-known superconductor MgB2; they crystallize in a
In metal borides, the bonding of boron varies depending on the atomic ratio B/M. Diborides have B/M = 2, as in the well-known superconductor MgB2; they crystallize in a  YB25 and YB50 decompose without melting that hinders their growth as single crystals by the floating zone method. However, addition of a small amount of Si solves this problem and results in single crystals with the stoichiometry of YB41Si1.2. This stabilization technique allowed the synthesis of some other boron-rich rare-earth borides.
Albert and Hillebrecht reviewed binary and selected ternary boron compounds containing main-group elements, namely, borides of the alkali and alkaline-earth metals,
YB25 and YB50 decompose without melting that hinders their growth as single crystals by the floating zone method. However, addition of a small amount of Si solves this problem and results in single crystals with the stoichiometry of YB41Si1.2. This stabilization technique allowed the synthesis of some other boron-rich rare-earth borides.
Albert and Hillebrecht reviewed binary and selected ternary boron compounds containing main-group elements, namely, borides of the alkali and alkaline-earth metals,
 Figure 3 shows the crystal structure of YAlB14 viewed along the ''x''-axis. The large black spheres are Y atoms, the small blue spheres are Al atoms and the small green spheres are the bridging boron sites; B12 clusters are depicted as the green icosahedra. Boron framework of YAlB14 is one of the simplest among icosahedron-based borides – it consists of only one kind of icosahedra and one bridging boron site. The bridging boron site is tetrahedrally coordinated by four boron atoms. Those atoms are another boron atom in the counter bridge site and three equatorial boron atoms of one of three B12 icosahedra. Aluminium atoms are separated by 0.2911 nm and are arranged in lines parallel to the ''x''-axis, whereas yttrium atoms are separated by 0.3405 nm. Both the Y atoms and B12 icosahedra form zigzags along the ''x''-axis. The bridging boron atoms connect three equatorial boron atoms of three icosahedra and those icosahedra make up a network parallel to the (101) crystal plane (''x''-''z'' plane in the figure). The bonding distance between the bridging boron and the equatorial boron atoms is 0.1755 nm, which is typical for the strong covalent B-B bond (bond length 0.17–0.18 nm); thus, the bridging boron atoms strengthen the individual network planes. On the other hand, the large distance between the boron atoms within the bridge (0.2041 nm) suggests weaker interaction, and thus the bridging sites contribute little to the bonding between the network planes.
The boron framework of YAlB14 needs donation of four electrons from metal elements: two electrons for a B12 icosahedron and one electron for each of the two bridging boron atoms – to support their tetrahedral coordination. The actual chemical composition of YAlB14, determined by the structure analysis, is Y0.62Al0.71B14 as described in table I. If both metal elements are trivalent ions then 3.99 electrons can be transferred to the boron framework, which is very close to the required value of 4. However, because the bonding between the bridging boron atoms is weaker than in a typical B-B covalent bond, less than 2 electrons are donated to this bond, and metal atoms need not be trivalent. On the other hand, the electron transfer from metal atoms to the boron framework implies that not only strong covalent B-B bonding within the framework but also ionic interaction between metal atoms and the framework contribute to the YAlB14 phase stabilization.
Figure 3 shows the crystal structure of YAlB14 viewed along the ''x''-axis. The large black spheres are Y atoms, the small blue spheres are Al atoms and the small green spheres are the bridging boron sites; B12 clusters are depicted as the green icosahedra. Boron framework of YAlB14 is one of the simplest among icosahedron-based borides – it consists of only one kind of icosahedra and one bridging boron site. The bridging boron site is tetrahedrally coordinated by four boron atoms. Those atoms are another boron atom in the counter bridge site and three equatorial boron atoms of one of three B12 icosahedra. Aluminium atoms are separated by 0.2911 nm and are arranged in lines parallel to the ''x''-axis, whereas yttrium atoms are separated by 0.3405 nm. Both the Y atoms and B12 icosahedra form zigzags along the ''x''-axis. The bridging boron atoms connect three equatorial boron atoms of three icosahedra and those icosahedra make up a network parallel to the (101) crystal plane (''x''-''z'' plane in the figure). The bonding distance between the bridging boron and the equatorial boron atoms is 0.1755 nm, which is typical for the strong covalent B-B bond (bond length 0.17–0.18 nm); thus, the bridging boron atoms strengthen the individual network planes. On the other hand, the large distance between the boron atoms within the bridge (0.2041 nm) suggests weaker interaction, and thus the bridging sites contribute little to the bonding between the network planes.
The boron framework of YAlB14 needs donation of four electrons from metal elements: two electrons for a B12 icosahedron and one electron for each of the two bridging boron atoms – to support their tetrahedral coordination. The actual chemical composition of YAlB14, determined by the structure analysis, is Y0.62Al0.71B14 as described in table I. If both metal elements are trivalent ions then 3.99 electrons can be transferred to the boron framework, which is very close to the required value of 4. However, because the bonding between the bridging boron atoms is weaker than in a typical B-B covalent bond, less than 2 electrons are donated to this bond, and metal atoms need not be trivalent. On the other hand, the electron transfer from metal atoms to the boron framework implies that not only strong covalent B-B bonding within the framework but also ionic interaction between metal atoms and the framework contribute to the YAlB14 phase stabilization.

Another structure unit of YB66, shown in figure 4b, is B80 cluster of 80 boron sites formed by the B10 to B13 sites. All those 80 sites are partially occupied and in total contain only about 42 boron atoms. The B80 cluster is located at the body center of the octant of the unit cell, i.e., at the 8''a'' position (1/4, 1/4, 1/4); thus, there are eight such clusters (336 boron atoms) per unit cell. Two independent structure analyses came to the same conclusion that the total number of boron atoms in the unit cell is 1584. The boron framework structure of YB66 is shown in figure 5a. To indicate relative orientations of the supericosahedra, a schematic drawing is shown in figure 5b, where the supericosahedra and the B80 clusters are depicted by light green and dark green spheres, respectively; at the top surface of the unit cell, the relative orientations of the supericosahedra are indicated by arrows. There are 48 yttrium sites ((0.0563, 1/4, 1/4) for YB62) in the unit cell. Richards and Kasper fixed the Y site occupancy to 0.5 that resulted in 24 Y atoms in the unit cell and the chemical composition of YB66. As shown in figure 6, Y sites form a pair separated by only 0.264 nm in YB62. This pair is aligned normal to the plane formed by four supericosahedra. The Y site occupancy 0.5 implies that the pair has always one Y atom with one empty site.
Slack ''et al.'' reported that the total number of boron atoms in the unit cell, calculated from the measured values of density, chemical composition and lattice constant, is 1628 ± 4, which is larger than the value 1584 obtained from the structural analysis. The number of B atoms in the unit cell remains nearly constant when the chemical composition changes from YB56 to YB66. On the other hand, the total number of yttrium atoms per unit cell varies, and it is, for example, ~26.3 for YB62 (see right table). If the total number of Y atoms stays below or equal to 24 then it is possible that one Y atom accommodates in each Y pair (partial occupancy). However, the experimental value of 26.3 significantly exceeds 24, and thus both pair sites might be occupied. In this case, because of the small separation between the two Y atoms, they must be repelled by the
 Initially these were described as ternary RE-B-Si compounds, but later carbon was included to improve the structure description that resulted in a quaternary RE-B-C-Si composition. RExB12C0.33Si3.0 (RE=Y and Gd–Lu) have a unique crystal structure with two units – a cluster of B12 icosahedra and a Si8 ethane-like complex – and one bonding configuration (B12)3≡Si-C≡(B12)3. A representative compound of this group is YxB12C0.33Si3.0 (x=0.68). It has a trigonal crystal structure with space group ''Rm'' (No. 166) and lattice constants ''a'' = ''b'' = 1.00841(4) nm, ''c'' = 1.64714(5) nm, α = β = 90° and γ = 120°.
Initially these were described as ternary RE-B-Si compounds, but later carbon was included to improve the structure description that resulted in a quaternary RE-B-C-Si composition. RExB12C0.33Si3.0 (RE=Y and Gd–Lu) have a unique crystal structure with two units – a cluster of B12 icosahedra and a Si8 ethane-like complex – and one bonding configuration (B12)3≡Si-C≡(B12)3. A representative compound of this group is YxB12C0.33Si3.0 (x=0.68). It has a trigonal crystal structure with space group ''Rm'' (No. 166) and lattice constants ''a'' = ''b'' = 1.00841(4) nm, ''c'' = 1.64714(5) nm, α = β = 90° and γ = 120°.
 The crystal has layered structure. Figure 15 shows a network of boron icosahedra that spreads parallel to the (001) plane, connecting with four neighbors through B1–B1 bonds. The C3 and Si3 site atoms strengthen the network by bridging the boron icosahedra. Contrary to other boron-rich icosahedral compounds, the boron icosahedra from different layers are not directly bonded. The icosahedra within one layer are linked through Si8 ethane-like clusters with (B12)3≡Si-C≡(B12)3 bonds, as shown in figures 16a and b.
There are eight atomic sites in the unit cell: one yttrium Y, four boron B1–B4, one carbon C3 and three silicon sites Si1–Si3. Atomic coordinates, site occupancy and isotropic displacement factors are listed in table Va; 68% of the Y sites are randomly occupied and remaining Y sites are vacant. All boron sites and Si1 and Si2 sites are fully occupied. The C3 and Si3 sites can be occupied by either carbon or silicon atoms (mixed occupancy) with a probability of about 50%. Their separation is only 0.413 Å, and thus either the C3 or Si3 sites, but not both, are occupied. These sites form Si-C pairs, but not Si-Si or C-C pairs. The distances between the C3 and Si3 sites and the surrounding sites for YxB12C0.33Si3.0 are summarized in table Vb and the overall crystal structure is shown in figure 14.
Salvador ''et al.'' reported an isotypic terbium compound
Tb3–xC2Si8(B12)3. Most parts of the crystal structure are the same as those described above; however, its bonding configuration is deduced as (B12)3≡C-C≡(B12)3 instead of (B12)3≡Si-C≡(B12)3. The authors intentionally added carbon to grow single crystals whereas the previous crystals were accidentally contaminated by carbon during their growth. Thus, higher carbon concentration was achieved. Existence of both bonding schemes of (B12)3≡Si-C≡(B12)3 and (B12)3≡C-C≡(B12)3 suggests the occupancy of the carbon sites of 50–100%. On the other hand, (B12)3≡Si-Si≡(B12)3 bonding scheme is unlikely because of too short Si-Si distance, suggesting that the minimum carbon occupancy at the site is 50%. Some B atoms may replace C atoms at the C3 site, as previously assigned to the B site. However, the carbon occupation is more likely because the site is tetrahedrally coordinated whereas the B occupation of the site needs an extra electron to complete tetrahedral bonding. Thus, carbon is indispensable for this group of compounds.
The crystal has layered structure. Figure 15 shows a network of boron icosahedra that spreads parallel to the (001) plane, connecting with four neighbors through B1–B1 bonds. The C3 and Si3 site atoms strengthen the network by bridging the boron icosahedra. Contrary to other boron-rich icosahedral compounds, the boron icosahedra from different layers are not directly bonded. The icosahedra within one layer are linked through Si8 ethane-like clusters with (B12)3≡Si-C≡(B12)3 bonds, as shown in figures 16a and b.
There are eight atomic sites in the unit cell: one yttrium Y, four boron B1–B4, one carbon C3 and three silicon sites Si1–Si3. Atomic coordinates, site occupancy and isotropic displacement factors are listed in table Va; 68% of the Y sites are randomly occupied and remaining Y sites are vacant. All boron sites and Si1 and Si2 sites are fully occupied. The C3 and Si3 sites can be occupied by either carbon or silicon atoms (mixed occupancy) with a probability of about 50%. Their separation is only 0.413 Å, and thus either the C3 or Si3 sites, but not both, are occupied. These sites form Si-C pairs, but not Si-Si or C-C pairs. The distances between the C3 and Si3 sites and the surrounding sites for YxB12C0.33Si3.0 are summarized in table Vb and the overall crystal structure is shown in figure 14.
Salvador ''et al.'' reported an isotypic terbium compound
Tb3–xC2Si8(B12)3. Most parts of the crystal structure are the same as those described above; however, its bonding configuration is deduced as (B12)3≡C-C≡(B12)3 instead of (B12)3≡Si-C≡(B12)3. The authors intentionally added carbon to grow single crystals whereas the previous crystals were accidentally contaminated by carbon during their growth. Thus, higher carbon concentration was achieved. Existence of both bonding schemes of (B12)3≡Si-C≡(B12)3 and (B12)3≡C-C≡(B12)3 suggests the occupancy of the carbon sites of 50–100%. On the other hand, (B12)3≡Si-Si≡(B12)3 bonding scheme is unlikely because of too short Si-Si distance, suggesting that the minimum carbon occupancy at the site is 50%. Some B atoms may replace C atoms at the C3 site, as previously assigned to the B site. However, the carbon occupation is more likely because the site is tetrahedrally coordinated whereas the B occupation of the site needs an extra electron to complete tetrahedral bonding. Thus, carbon is indispensable for this group of compounds.
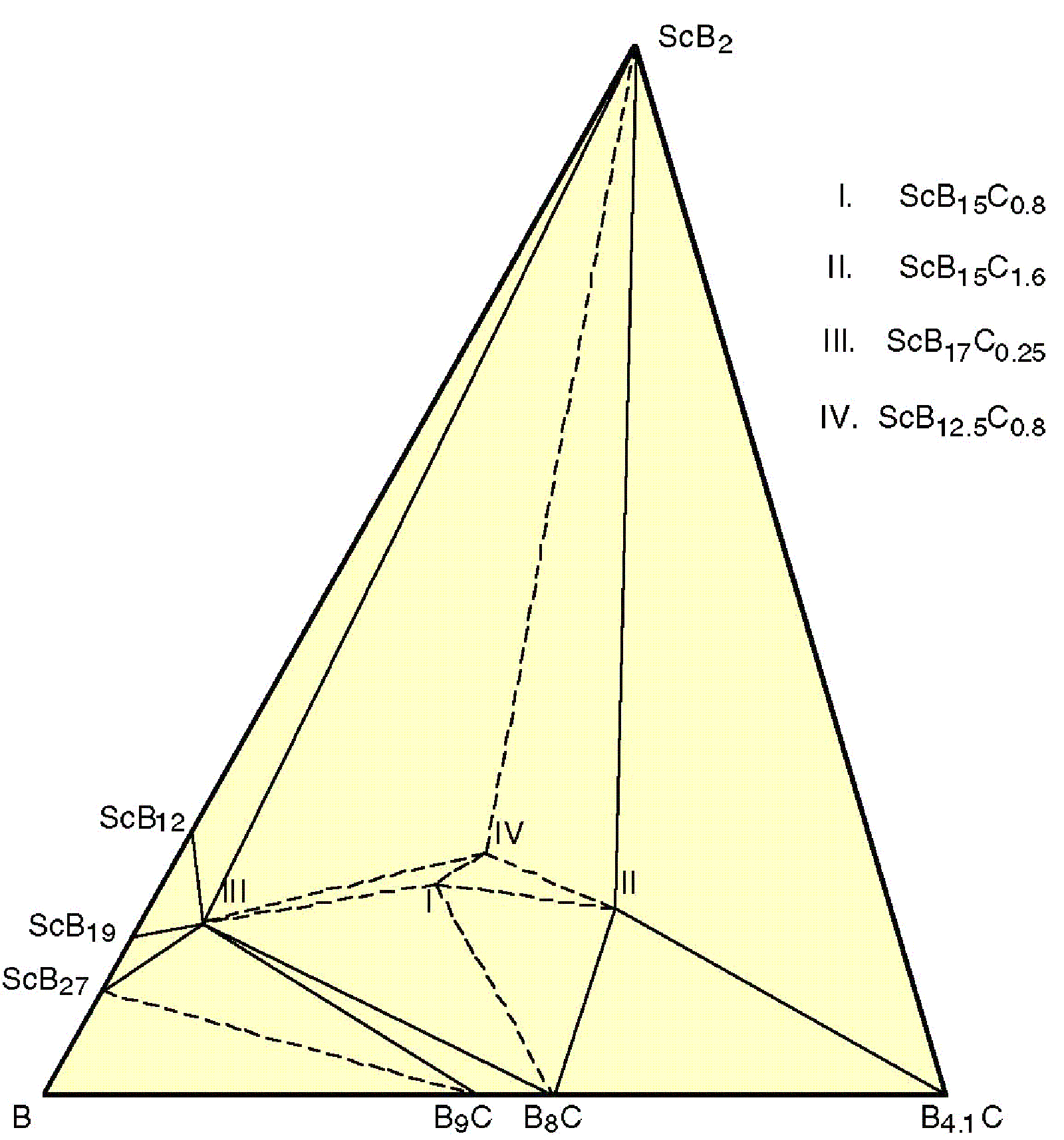 Scandium has the smallest atomic and ionic (3+) radii (1.62 and 0.885 Å, respectively) among the rare-earth elements. It forms several icosahedron-based borides which are not found for other rare-earth elements; however, most of them are ternary Sc-B-C compounds. There are many boron-rich phases in the boron-rich corner of Sc-B-C phase diagram, as shown in figure 17. A slight variation of the composition can produce ScB19, ScB17C0.25, ScB15C0.8 and ScB15C1.6; their crystal structures are unusual for borides and are very different from each other.
Scandium has the smallest atomic and ionic (3+) radii (1.62 and 0.885 Å, respectively) among the rare-earth elements. It forms several icosahedron-based borides which are not found for other rare-earth elements; however, most of them are ternary Sc-B-C compounds. There are many boron-rich phases in the boron-rich corner of Sc-B-C phase diagram, as shown in figure 17. A slight variation of the composition can produce ScB19, ScB17C0.25, ScB15C0.8 and ScB15C1.6; their crystal structures are unusual for borides and are very different from each other.
 Very small amount of carbon is sufficient to stabilize "ScB17C0.25". This compound has a broad composition range, namely ScB16.5+xC0.2+y with x ≤ 2.2 and y ≤ 0.44. ScB17C0.25 has a hexagonal crystal structure with space group ''P6mmm'' (No. 199) and lattice constants ''a, b'' = 1.45501(15) nm and ''c'' = 0.84543(16) nm.
There are 19 atomic sites in the unit cell, which are assigned to one scandium site Sc, 14 boron sites B1–B14 having 100% occupancy, two boron-carbon mixed-occupancy sites B/C15 and B/C16, and two partial-occupancy boron sites B17 and B18. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VII. Although a very small amount of carbon (less than 2 wt%!) plays an important role in the phase stability, carbon does not have its own sites but shares with boron two interstitial sites B/C15 and B/C16.
There are two inequivalent B12 icosahedra, I1 and I2, which are constructed by the B1–B5 and B8–B12 sites, respectively. A "tube" is another characteristic structure unit of ScB17C0.25. It extends along the ''c''-axis and consists of B13, B14, B17 and B18 sites where B13 and B14 form 6-membered rings. B17 and B18 sites also form 6-membered rings; however, their mutual distances (0.985 Å for B17 and 0.955 Å for B18) are too short for a simultaneous occupation of the neighboring sites. Therefore, boron atoms occupy 2nd neighbor site forming a triangle. The occupancies of B17 and B18 sites should be 50%, but the structure analysis suggests larger values. The crystal structure viewed along the ''a''-axis is shown in figure 20, which suggests that the ScB17C0.25 is a layered material. Two layers, respectively constructed by the icosahedra I1 and I2, alternatively stack along the ''c''-axis. However, the ScB17C0.25 crystal is not layered. For example, during arc-melting, ScB17C0.25 needle crystals violently grow along the ''c''-axis – this never happens in layered compounds. The crystal structure viewed along the ''c''-axis is shown in figure 21a. The icosahedra I1 and I2 form a ring centered by the "tube" shown in figure 21b, which probably governs the properties of the ScB17C0.25 crystal. B/C15 and B/C16 mixed-occupancy sites interconnect the rings. A structural similarity can be seen between ScB17C0.25 and BeB3.
Figures 22a and b present HRTEM lattice images and electron diffraction patterns taken along the
Very small amount of carbon is sufficient to stabilize "ScB17C0.25". This compound has a broad composition range, namely ScB16.5+xC0.2+y with x ≤ 2.2 and y ≤ 0.44. ScB17C0.25 has a hexagonal crystal structure with space group ''P6mmm'' (No. 199) and lattice constants ''a, b'' = 1.45501(15) nm and ''c'' = 0.84543(16) nm.
There are 19 atomic sites in the unit cell, which are assigned to one scandium site Sc, 14 boron sites B1–B14 having 100% occupancy, two boron-carbon mixed-occupancy sites B/C15 and B/C16, and two partial-occupancy boron sites B17 and B18. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VII. Although a very small amount of carbon (less than 2 wt%!) plays an important role in the phase stability, carbon does not have its own sites but shares with boron two interstitial sites B/C15 and B/C16.
There are two inequivalent B12 icosahedra, I1 and I2, which are constructed by the B1–B5 and B8–B12 sites, respectively. A "tube" is another characteristic structure unit of ScB17C0.25. It extends along the ''c''-axis and consists of B13, B14, B17 and B18 sites where B13 and B14 form 6-membered rings. B17 and B18 sites also form 6-membered rings; however, their mutual distances (0.985 Å for B17 and 0.955 Å for B18) are too short for a simultaneous occupation of the neighboring sites. Therefore, boron atoms occupy 2nd neighbor site forming a triangle. The occupancies of B17 and B18 sites should be 50%, but the structure analysis suggests larger values. The crystal structure viewed along the ''a''-axis is shown in figure 20, which suggests that the ScB17C0.25 is a layered material. Two layers, respectively constructed by the icosahedra I1 and I2, alternatively stack along the ''c''-axis. However, the ScB17C0.25 crystal is not layered. For example, during arc-melting, ScB17C0.25 needle crystals violently grow along the ''c''-axis – this never happens in layered compounds. The crystal structure viewed along the ''c''-axis is shown in figure 21a. The icosahedra I1 and I2 form a ring centered by the "tube" shown in figure 21b, which probably governs the properties of the ScB17C0.25 crystal. B/C15 and B/C16 mixed-occupancy sites interconnect the rings. A structural similarity can be seen between ScB17C0.25 and BeB3.
Figures 22a and b present HRTEM lattice images and electron diffraction patterns taken along the
 Sc0.83–xB10.0–yC0.17+ySi0.083–z (''x'' = 0.030, ''y'' = 0.36 and ''z'' = 0.026) has a cubic crystal structure with space group ''F3m'' (No. 216) and lattice constant ''a'' = 2.03085(5) nm. This compound was initially identified as ScB15C0.8 (phase I in the Sc-B-C phase diagram of figure 17). A small amount of Si was added into the floating zone crystal growth and thus this phase is a quaternary compound. Its rare cubic structure has 26 sites in the unit cell: three Sc sites, two Si sites, one C site and 20 B sites; 4 out of 20 B sites are boron-carbon mixed-occupancy sites. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VIII.
In the unit cell, there are three independent icosahedra, I1, I2 and I3, and a B10 polyhedron which are formed by the B1–B4, B5–B8, B9–B13 and B14–B17 sites, respectively.There are more than 4 sites in total among, say, B5–B8 sites, but many of them are equivalent by symmetry and thus do not have an individual label. The B10 polyhedron has not been observed previously and it is shown in figure 23. The icosahedron I2 has a boron-carbon mixed-occupancy site B,C6 whose occupancy is B/C=0.58/0.42. Remaining 3 boron-carbon mixed-occupancy sites are bridge sites; C and Si sites are also bridge sites.
More than 1000 atoms are available in the unit cell, which is built up by large structure units such as two supertetrahedra T(1) and T(2) and one superoctahedron O(1). As shown in figure 24a, T(1) consists of 4 icosahedra I(1) which have no direct bonding but are bridged by four B and C20 atoms. These atoms also form tetrahedron centered by the Si2 sites. The supertetrahedron T(2) that consists of 4 icosahedra I(2) is the same as shown in figure 18b; its mixed-occupancy sites B and C6 directly bond with each other. The superoctahedron O(1) consists of 6 icosahedra I(3) and bridge sites B, C18, C1 and Si1; here Si1 and C1 exhibit a tetrahedral arrangement at the center of O(1). The B10 polyhedra also arrange octahedrally, without the central atom, as shown in figure 24c where the B and C19 atoms bridge the B10 polyhedra to form the octahedral supercluster of the B10 polyhedra.
Sc0.83–xB10.0–yC0.17+ySi0.083–z (''x'' = 0.030, ''y'' = 0.36 and ''z'' = 0.026) has a cubic crystal structure with space group ''F3m'' (No. 216) and lattice constant ''a'' = 2.03085(5) nm. This compound was initially identified as ScB15C0.8 (phase I in the Sc-B-C phase diagram of figure 17). A small amount of Si was added into the floating zone crystal growth and thus this phase is a quaternary compound. Its rare cubic structure has 26 sites in the unit cell: three Sc sites, two Si sites, one C site and 20 B sites; 4 out of 20 B sites are boron-carbon mixed-occupancy sites. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VIII.
In the unit cell, there are three independent icosahedra, I1, I2 and I3, and a B10 polyhedron which are formed by the B1–B4, B5–B8, B9–B13 and B14–B17 sites, respectively.There are more than 4 sites in total among, say, B5–B8 sites, but many of them are equivalent by symmetry and thus do not have an individual label. The B10 polyhedron has not been observed previously and it is shown in figure 23. The icosahedron I2 has a boron-carbon mixed-occupancy site B,C6 whose occupancy is B/C=0.58/0.42. Remaining 3 boron-carbon mixed-occupancy sites are bridge sites; C and Si sites are also bridge sites.
More than 1000 atoms are available in the unit cell, which is built up by large structure units such as two supertetrahedra T(1) and T(2) and one superoctahedron O(1). As shown in figure 24a, T(1) consists of 4 icosahedra I(1) which have no direct bonding but are bridged by four B and C20 atoms. These atoms also form tetrahedron centered by the Si2 sites. The supertetrahedron T(2) that consists of 4 icosahedra I(2) is the same as shown in figure 18b; its mixed-occupancy sites B and C6 directly bond with each other. The superoctahedron O(1) consists of 6 icosahedra I(3) and bridge sites B, C18, C1 and Si1; here Si1 and C1 exhibit a tetrahedral arrangement at the center of O(1). The B10 polyhedra also arrange octahedrally, without the central atom, as shown in figure 24c where the B and C19 atoms bridge the B10 polyhedra to form the octahedral supercluster of the B10 polyhedra.
 Using these large polyhedra, the crystal structure of Sc0.83–xB10.0–yC0.17+ySi0.083–z can be described as shown in figure 25. Owing to the crystal symmetry, the tetrahedral coordination between these superstructure units is again a key factor. The supertetrahedron T(1) lies at the body center and at the edge center of the unit cell. The superoctahedra O(1) locate at the body center (0.25, 0.25, 0.25) of the quarter of the unit cell. They coordinate tetrahedrally around T(1) forming a giant tetrahedron. The supertetrahedra T(2) are located at the symmetry-related positions (0.25, 0.25, 0.75); they also form a giant tetrahedron surrounding T(1). Edges of both giant tetrahedra orthogonally cross each other at their centers; at those edge centers, each B10 polyhedron bridges all the super-structure clusters T(1), T(2) and O(1). The superoctahedron built of B10 polyhedra is located at each cubic face center.
Scandium atoms reside in the voids of the boron framework. Four Sc1 atoms form a tetrahedral arrangement inside the B10 polyhedron-based superoctahedron. Sc2 atoms sit between the B10 polyhedron-based superoctahedron and the O(1) superoctahedron. Three Sc3 atoms form a triangle and are surrounded by three B10 polyhedra, a supertetrahedron T(1) and a superoctahedron O(1).
Using these large polyhedra, the crystal structure of Sc0.83–xB10.0–yC0.17+ySi0.083–z can be described as shown in figure 25. Owing to the crystal symmetry, the tetrahedral coordination between these superstructure units is again a key factor. The supertetrahedron T(1) lies at the body center and at the edge center of the unit cell. The superoctahedra O(1) locate at the body center (0.25, 0.25, 0.25) of the quarter of the unit cell. They coordinate tetrahedrally around T(1) forming a giant tetrahedron. The supertetrahedra T(2) are located at the symmetry-related positions (0.25, 0.25, 0.75); they also form a giant tetrahedron surrounding T(1). Edges of both giant tetrahedra orthogonally cross each other at their centers; at those edge centers, each B10 polyhedron bridges all the super-structure clusters T(1), T(2) and O(1). The superoctahedron built of B10 polyhedra is located at each cubic face center.
Scandium atoms reside in the voids of the boron framework. Four Sc1 atoms form a tetrahedral arrangement inside the B10 polyhedron-based superoctahedron. Sc2 atoms sit between the B10 polyhedron-based superoctahedron and the O(1) superoctahedron. Three Sc3 atoms form a triangle and are surrounded by three B10 polyhedra, a supertetrahedron T(1) and a superoctahedron O(1).
 Sc3.67–xB41.4–y–zC0.67+zSi0.33–w (x = 0.52, y = 1.42, z = 1.17 and w = 0.02) has a hexagonal crystal structure with space group ''Pm2'' (No. 187) and lattice constants ''a'' = ''b'' = 1.43055(8) and ''c'' = 2.37477(13) nm. Single crystals of this compound were obtained as an intergrowth phase in a float-zoned single crystal of Sc0.83–xB10.0–yC0.17+ySi0.083–z. This phase is not described in the phase diagram of figure 17 because it is a quaternary compound. Its hexagonal structure is rare and has 79 atomic positions in the unit cell: eight partially occupied Sc sites, 62 B sites, two C sites, two Si sites and six B/C sites. Six B sites and one of the two Si sites have partial occupancies. The associated atomic coordinates, site occupancies and isotropic displacement factors are listed in table X.
There are seven structurally independent icosahedra I1–I7 which are formed by B1–B8, B9–B12, B13–B20, B/C21–B24, B/C25–B29, B30–B37 and B/C38–B42 sites, respectively; B43–B46 sites form the B9 polyhedron and B47–B53 sites construct the B10 polyhedron. B54–B59 sites form the irregularly shaped B16 polyhedron in which only 10.7 boron atoms are available because most of sites are too close to each other to be occupied simultaneously. Ten bridging sites C60–B69 interconnect polyhedron units or other bridging sites to form a 3D boron framework structure. One description of the crystal structure uses three pillar-like units that extend along the ''c''-axis that however results in undesired overlaps between those three pillar-like units. An alternative is to define two pillar-like structure units. Figure 29 shows the boron framework structure of Sc3.67–xB41.4–y–zC0.67+zSi0.33–w viewed along the ''c''-axis, where the pillar-like units P1 and P2 are colored in dark green and light green respectively and are bridged by yellow icosahedra I4 and I7.
These pillar-like units P1 and P2 are shown in figures 30a and b, respectively. P1 consists of icosahedra I1 and I3, an irregularly shaped B16 polyhedron and other bridge site atoms where two supericosahedra can be seen above and below the B16 polyhedron. Each supericosahedron is formed by three icosahedra I1 and three icosahedra I3 and is the same as the supericosahedron O(1) shown in figure 24a.The P2 unit consists of icosahedra I2, I5 and I6, B10 polyhedron and other bridge site atoms. Eight Sc sites with occupancies between 0.49 (Sc8) and 0.98 (Sc1) spread over the boron framework.
As described above, this hexagonal phase originates from a cubic phase, and thus one may expect a similar structural element in these phases. There is an obvious relation between the hexagonal ''ab''-plane and the cubic (111) plane. Figures 31a and b show the hexagonal (001) and the cubic (111) planes, respectively. Both network structures are almost the same that allows intergrowth of the hexagonal phase in the cubic phase.
Sc3.67–xB41.4–y–zC0.67+zSi0.33–w (x = 0.52, y = 1.42, z = 1.17 and w = 0.02) has a hexagonal crystal structure with space group ''Pm2'' (No. 187) and lattice constants ''a'' = ''b'' = 1.43055(8) and ''c'' = 2.37477(13) nm. Single crystals of this compound were obtained as an intergrowth phase in a float-zoned single crystal of Sc0.83–xB10.0–yC0.17+ySi0.083–z. This phase is not described in the phase diagram of figure 17 because it is a quaternary compound. Its hexagonal structure is rare and has 79 atomic positions in the unit cell: eight partially occupied Sc sites, 62 B sites, two C sites, two Si sites and six B/C sites. Six B sites and one of the two Si sites have partial occupancies. The associated atomic coordinates, site occupancies and isotropic displacement factors are listed in table X.
There are seven structurally independent icosahedra I1–I7 which are formed by B1–B8, B9–B12, B13–B20, B/C21–B24, B/C25–B29, B30–B37 and B/C38–B42 sites, respectively; B43–B46 sites form the B9 polyhedron and B47–B53 sites construct the B10 polyhedron. B54–B59 sites form the irregularly shaped B16 polyhedron in which only 10.7 boron atoms are available because most of sites are too close to each other to be occupied simultaneously. Ten bridging sites C60–B69 interconnect polyhedron units or other bridging sites to form a 3D boron framework structure. One description of the crystal structure uses three pillar-like units that extend along the ''c''-axis that however results in undesired overlaps between those three pillar-like units. An alternative is to define two pillar-like structure units. Figure 29 shows the boron framework structure of Sc3.67–xB41.4–y–zC0.67+zSi0.33–w viewed along the ''c''-axis, where the pillar-like units P1 and P2 are colored in dark green and light green respectively and are bridged by yellow icosahedra I4 and I7.
These pillar-like units P1 and P2 are shown in figures 30a and b, respectively. P1 consists of icosahedra I1 and I3, an irregularly shaped B16 polyhedron and other bridge site atoms where two supericosahedra can be seen above and below the B16 polyhedron. Each supericosahedron is formed by three icosahedra I1 and three icosahedra I3 and is the same as the supericosahedron O(1) shown in figure 24a.The P2 unit consists of icosahedra I2, I5 and I6, B10 polyhedron and other bridge site atoms. Eight Sc sites with occupancies between 0.49 (Sc8) and 0.98 (Sc1) spread over the boron framework.
As described above, this hexagonal phase originates from a cubic phase, and thus one may expect a similar structural element in these phases. There is an obvious relation between the hexagonal ''ab''-plane and the cubic (111) plane. Figures 31a and b show the hexagonal (001) and the cubic (111) planes, respectively. Both network structures are almost the same that allows intergrowth of the hexagonal phase in the cubic phase.

Metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
s, and specifically rare-earth elements, form numerous chemical complexes with boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the ''boron group'' it has th ...
. Their crystal structure
In crystallography, crystal structure is a description of the ordered arrangement of atoms, ions or molecules in a crystal, crystalline material. Ordered structures occur from the intrinsic nature of the constituent particles to form symmetric pat ...
and chemical bonding depend strongly on the metal element M and on its atomic ratio to boron. When B/M ratio exceeds 12, boron atoms form B12 icosahedra
In geometry, an icosahedron ( or ) is a polyhedron with 20 faces. The name comes and . The plural can be either "icosahedra" () or "icosahedrons".
There are infinitely many non- similar shapes of icosahedra, some of them being more symmetrica ...
which are linked into a three-dimensional boron framework, and the metal atoms reside in the voids of this framework. Those icosahedra are basic structural units of most allotropes of boron and boron-rich rare-earth borides. In such borides, metal atoms donate electrons to the boron polyhedra, and thus these compounds are regarded as electron-deficient Electron deficiency (and electron-deficient) is jargon that is used in two contexts: species that violate the octet rule because they have too few valence electrons and species that happen to follow the octet rule but have electron-acceptor properti ...
solids.
The crystal structures of many boron-rich borides can be attributed to certain types including MgAlB14, YB66, REB41Si1.2, B4C and other, more complex types such as RExB12C0.33Si3.0. Some of these formulas, for example B4C, YB66 and MgAlB14, historically reflect the idealistic structures, whereas the experimentally determined composition is nonstoichiometric and corresponds to fractional indexes. Boron-rich borides are usually characterized by large and complex unit cells, which can contain more than 1500 atomic sites and feature extended structures shaped as "tubes" and large modular polyhedra ("superpolyhedra"). Many of those sites have partial occupancy, meaning that the probability to find them occupied with a certain atom is smaller than one and thus that only some of them are filled with atoms. Scandium is distinguished among the rare-earth elements by that it forms numerous borides with uncommon structure types; this property of scandium is attributed to its relatively small atomic and ionic radii.
Crystals of the specific rare-earth boride YB66 are used as X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
monochromator
A monochromator is an optical device that transmits a mechanically selectable narrow band of wavelengths of light or other radiation chosen from a wider range of wavelengths available at the input. The name is from the Greek roots ''mono-'', "si ...
s for selecting X-rays with certain energies (in the 1–2 keV range) out of synchrotron
A synchrotron is a particular type of cyclic particle accelerator, descended from the cyclotron, in which the accelerating particle beam travels around a fixed closed-loop path. The magnetic field which bends the particle beam into its closed p ...
radiation. Other rare-earth borides may find application as thermoelectric materials, owing to their low thermal conductivity
The thermal conductivity of a material is a measure of its ability to conduct heat. It is commonly denoted by k, \lambda, or \kappa.
Heat transfer occurs at a lower rate in materials of low thermal conductivity than in materials of high thermal ...
; the latter originates from their complex, "amorphous-like", crystal structure.
Metal borides
 In metal borides, the bonding of boron varies depending on the atomic ratio B/M. Diborides have B/M = 2, as in the well-known superconductor MgB2; they crystallize in a
In metal borides, the bonding of boron varies depending on the atomic ratio B/M. Diborides have B/M = 2, as in the well-known superconductor MgB2; they crystallize in a hexagonal
In geometry, a hexagon (from Greek , , meaning "six", and , , meaning "corner, angle") is a six-sided polygon. The total of the internal angles of any simple (non-self-intersecting) hexagon is 720°.
Regular hexagon
A '' regular hexagon'' has ...
AlB2-type layered structure. Hexaborides have B/M = 6 and form a three-dimensional boron framework based on a boron octahedron (Fig. 1a). Tetraborides, i.e. B/M = 4, are mixtures of diboride and hexaboride structures. Cuboctahedron (Fig. 1b) is the structural unit of dodecaborides, which have a cubic lattice and B/M = 12. When the composition ratio exceeds 12, boron forms B12 icosahedra
In geometry, an icosahedron ( or ) is a polyhedron with 20 faces. The name comes and . The plural can be either "icosahedra" () or "icosahedrons".
There are infinitely many non- similar shapes of icosahedra, some of them being more symmetrica ...
(Fig. 1c) which are linked into a three-dimensional boron framework, and the metal atoms reside in the voids of this framework.
This complex bonding behavior originates from the fact that boron has only three valence electrons; this hinders tetrahedral bonding as in diamond
Diamond is a Allotropes of carbon, solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. Another solid form of carbon known as graphite is the Chemical stability, chemically stable form of car ...
or hexagonal bonding as in graphite
Graphite () is a crystalline form of the element carbon. It consists of stacked layers of graphene. Graphite occurs naturally and is the most stable form of carbon under standard conditions. Synthetic and natural graphite are consumed on large ...
. Instead, boron atoms form polyhedra. For example, three boron atoms make up a triangle where they share two electrons to complete the so-called three-center bonding. Boron polyhedra, such as B6 octahedron, B12 cuboctahedron and B12 icosahedron, lack two valence electrons per polyhedron to complete the polyhedron-based framework structure. Metal atoms need to donate two electrons per boron polyhedron to form boron-rich metal borides. Thus, boron compounds are often regarded as electron-deficient solids. The covalent bonding nature of metal boride compounds also give them their hardness and inert chemical reactivity property.
Icosahedral B12 compounds include α-rhombohedral boron (B13C2), β-rhombohedral boron (MeBx, 23≤x), α-tetragonal boron (B48B2C2), β-tetragonal boron (β-AlB12), AlB10 or AlC4B24, YB25, YB50, YB66, NaB15 or MgAlB14, γ-AlB12, BeB3 and SiB6.
 YB25 and YB50 decompose without melting that hinders their growth as single crystals by the floating zone method. However, addition of a small amount of Si solves this problem and results in single crystals with the stoichiometry of YB41Si1.2. This stabilization technique allowed the synthesis of some other boron-rich rare-earth borides.
Albert and Hillebrecht reviewed binary and selected ternary boron compounds containing main-group elements, namely, borides of the alkali and alkaline-earth metals,
YB25 and YB50 decompose without melting that hinders their growth as single crystals by the floating zone method. However, addition of a small amount of Si solves this problem and results in single crystals with the stoichiometry of YB41Si1.2. This stabilization technique allowed the synthesis of some other boron-rich rare-earth borides.
Albert and Hillebrecht reviewed binary and selected ternary boron compounds containing main-group elements, namely, borides of the alkali and alkaline-earth metals, aluminum
Aluminium (aluminum in American and Canadian English) is a chemical element with the symbol Al and atomic number 13. Aluminium has a density lower than those of other common metals, at approximately one third that of steel. It has ...
borides and compounds of boron and the nonmetals C, Si, Ge, N, P, As, O, S and Se. They, however, excluded the described here icosahedron-based rare-earth borides. Note that rare-earth elements have ''d''- and ''f''-electrons that complicates chemical and physical properties of their borides. Werheit ''et al.'' reviewed Raman spectra of numerous icosahedron-based boron compounds.
Figure 2 shows a relationship between the ionic radius of trivalent rare-earth ions and the composition of some rare-earth borides. Note that scandium
Scandium is a chemical element with the symbol Sc and atomic number 21. It is a silvery-white metallic d-block element. Historically, it has been classified as a rare-earth element, together with yttrium and the Lanthanides. It was discovered in ...
has many unique boron compounds, as shown in figure 2, because of the much smaller ionic radius compared with other rare-earth elements.
In understanding the crystal structures of rare-earth borides, it is important to keep in mind the concept of partial site occupancy, that is, some atoms in the described below unit cells can take several possible positions with a given statistical probability. Thus, with the given statistical probability, some of the partial-occupancy sites in such a unit cell are empty, and the remained sites are occupied.
REAlB14 and REB25
Compounds that were historically given the formulae REAlB14 and REB25 have the MgAlB14 structure with anorthorhombic
In crystallography, the orthorhombic crystal system is one of the 7 crystal systems. Orthorhombic lattices result from stretching a cubic lattice along two of its orthogonal pairs by two different factors, resulting in a rectangular prism with a r ...
symmetry and space group ''Imma'' (No. 74). In this structure, rare-earth atoms enter the Mg site. Aluminium sites are empty for REB25. Both metal sites of REAlB14 structure have partial occupancies of about 60–70%, which shows that the compounds are actually non-stoichiometric. The REB25 formula merely reflects the average atomic ratio E= 25. Yttrium borides
Yttrium boride refers to a crystalline material composed of different proportions of yttrium and boron, such as YB2, YB4, YB6, YB12, YB25, YB50 and YB66. They are all gray-colored, hard solids having high melting temperatures. The most common form ...
form both YAlB14 and YB25 structures. Experiments have confirmed that the borides based on rare-earth elements from Tb to Lu can have the REAlB14 structure. A subset of these borides, which contains rare-earth elements from Gd to Er, can also crystallize in the REB25 structure.
Korsukova ''et al.'' analyzed the YAlB14 crystal structure using a single crystal grown by the high-temperature solution-growth method. The lattice constants were deduced as ''a'' = 0.58212(3), ''b'' = 1.04130(8) and ''c'' = 0.81947(6) nm, and the atomic coordinates and site occupancies are summarized in table I.
 Figure 3 shows the crystal structure of YAlB14 viewed along the ''x''-axis. The large black spheres are Y atoms, the small blue spheres are Al atoms and the small green spheres are the bridging boron sites; B12 clusters are depicted as the green icosahedra. Boron framework of YAlB14 is one of the simplest among icosahedron-based borides – it consists of only one kind of icosahedra and one bridging boron site. The bridging boron site is tetrahedrally coordinated by four boron atoms. Those atoms are another boron atom in the counter bridge site and three equatorial boron atoms of one of three B12 icosahedra. Aluminium atoms are separated by 0.2911 nm and are arranged in lines parallel to the ''x''-axis, whereas yttrium atoms are separated by 0.3405 nm. Both the Y atoms and B12 icosahedra form zigzags along the ''x''-axis. The bridging boron atoms connect three equatorial boron atoms of three icosahedra and those icosahedra make up a network parallel to the (101) crystal plane (''x''-''z'' plane in the figure). The bonding distance between the bridging boron and the equatorial boron atoms is 0.1755 nm, which is typical for the strong covalent B-B bond (bond length 0.17–0.18 nm); thus, the bridging boron atoms strengthen the individual network planes. On the other hand, the large distance between the boron atoms within the bridge (0.2041 nm) suggests weaker interaction, and thus the bridging sites contribute little to the bonding between the network planes.
The boron framework of YAlB14 needs donation of four electrons from metal elements: two electrons for a B12 icosahedron and one electron for each of the two bridging boron atoms – to support their tetrahedral coordination. The actual chemical composition of YAlB14, determined by the structure analysis, is Y0.62Al0.71B14 as described in table I. If both metal elements are trivalent ions then 3.99 electrons can be transferred to the boron framework, which is very close to the required value of 4. However, because the bonding between the bridging boron atoms is weaker than in a typical B-B covalent bond, less than 2 electrons are donated to this bond, and metal atoms need not be trivalent. On the other hand, the electron transfer from metal atoms to the boron framework implies that not only strong covalent B-B bonding within the framework but also ionic interaction between metal atoms and the framework contribute to the YAlB14 phase stabilization.
Figure 3 shows the crystal structure of YAlB14 viewed along the ''x''-axis. The large black spheres are Y atoms, the small blue spheres are Al atoms and the small green spheres are the bridging boron sites; B12 clusters are depicted as the green icosahedra. Boron framework of YAlB14 is one of the simplest among icosahedron-based borides – it consists of only one kind of icosahedra and one bridging boron site. The bridging boron site is tetrahedrally coordinated by four boron atoms. Those atoms are another boron atom in the counter bridge site and three equatorial boron atoms of one of three B12 icosahedra. Aluminium atoms are separated by 0.2911 nm and are arranged in lines parallel to the ''x''-axis, whereas yttrium atoms are separated by 0.3405 nm. Both the Y atoms and B12 icosahedra form zigzags along the ''x''-axis. The bridging boron atoms connect three equatorial boron atoms of three icosahedra and those icosahedra make up a network parallel to the (101) crystal plane (''x''-''z'' plane in the figure). The bonding distance between the bridging boron and the equatorial boron atoms is 0.1755 nm, which is typical for the strong covalent B-B bond (bond length 0.17–0.18 nm); thus, the bridging boron atoms strengthen the individual network planes. On the other hand, the large distance between the boron atoms within the bridge (0.2041 nm) suggests weaker interaction, and thus the bridging sites contribute little to the bonding between the network planes.
The boron framework of YAlB14 needs donation of four electrons from metal elements: two electrons for a B12 icosahedron and one electron for each of the two bridging boron atoms – to support their tetrahedral coordination. The actual chemical composition of YAlB14, determined by the structure analysis, is Y0.62Al0.71B14 as described in table I. If both metal elements are trivalent ions then 3.99 electrons can be transferred to the boron framework, which is very close to the required value of 4. However, because the bonding between the bridging boron atoms is weaker than in a typical B-B covalent bond, less than 2 electrons are donated to this bond, and metal atoms need not be trivalent. On the other hand, the electron transfer from metal atoms to the boron framework implies that not only strong covalent B-B bonding within the framework but also ionic interaction between metal atoms and the framework contribute to the YAlB14 phase stabilization.
REB66-type borides
In addition to yttrium, a wide range of rare-earth elements from Nd to Lu, except for Eu, can form REB66 compounds. Seybolt discovered the compound YB66 in 1960 and its structure was solved by Richards and Kasper in 1969. They reported that YB66 has a face-centered cubic structure with space group ''Fmc'' (No. 226) and lattice constant ''a'' = 2.3440(6) nm. There are 13 boron sites B1–B13 and one yttrium site. The B1 sites form one icosahedron and the B2–B9 sites make up another icosahedron. These icosahedra arrange in a thirteen-icosahedron unit (B12)12B12 which is shown in figure 4a and is called supericosahedron. The icosahedron formed by the B1 site atoms is located at the center of the supericosahedron. The supericosahedron is one of the basic units of the boron framework of YB66. There are two types of supericosahedra: one occupies the cubic face centers and another, which is rotated by 90°, is located at the center of the cell and at the cell edges. Thus, there are eight supericosahedra (1248 boron atoms) in the unit cell.
Coulomb force
Coulomb's inverse-square law, or simply Coulomb's law, is an experimental law of physics that quantifies the amount of force between two stationary, electrically charged particles. The electric force between charged bodies at rest is conventiona ...
. To clarify this point, split Y sites were introduced in the structure analysis resulting in a better agreement with the experiment. The Y site distances and occupancies are presented in the left table.
There are twenty Y pair sites with one Y atom and three pairs with two Y atoms; there is also one empty Y pair (partial occupancy = 0). The separation 0.340 nm for the Y2 pair site (two Y atoms in the pair site) is much larger than the separation 0.254 nm for the Y1 pair site (one Y atom in the pair site), as expected. The total number of Y atoms in the unit cell is 26.3, exactly as measured. Both cases are compared in figure 7. The larger separation for the Y2 pair site is clear as compared with that for the Y1 pair site. In case of the Y2 pair, some neighboring boron sites that belong to the B80 cluster must be unoccupied because they are too close to the Y2 site.
Splitting the Y site yields right number of Y atoms in the unit cell, but not B atoms. Not only the occupation of the B sites in the B80 cluster must be strongly dependent on whether or not the Y site is the Y1 state or the Y2 state, but also the position of the occupied B sites must be affected by the state of the Y site. Atomic coordinates and site occupancies are summarized in table II.
REB41Si1.2
Similar to yttrium, rare-earth metals from Gd to Lu can form REB41Si1.2-type boride. The first such compound was synthesized by solid-state reaction and its structure was deduced as YB50. X-ray powder diffraction (XRD) andelectron diffraction
Electron diffraction refers to the bending of electron beams around atomic structures. This behaviour, typical for waves, is applicable to electrons due to the wave–particle duality stating that electrons behave as both particles and waves. Si ...
indicated that YB50 has an orthorhombic structure with lattice constants ''a'' = 1.66251(9), ''b'' = 1.76198 and ''c'' = 0.94797(3) nm. The space group was assigned as ''P''21212. Because of the close similarity in lattice constants and space group, one might expect that YB50 has the γ-AlB12-type orthorhombic structure whose lattice constants and space group are ''a'' = 1.6573(4), ''b'' = 1.7510(3) and ''c'' = 1.0144(1) nm and ''P2''1212. YB50 decomposes at ~1750 °C without melting that hinders growth of single crystals from the melt. Small addition of silicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic tab ...
made YB50 to melt without decomposition, and so enabled single-crystal growth from the melt and single-crystal structure analysis.
The structure analysis indicated that YB41Si1.2 has not the γ-AlB12-type lattice but a rare orthorhombic crystal structure (space group: ''Pbam'', No. 55) with lattice constants of ''a'' = 1.674(1) nm, ''b'' = 1.7667(1) nm and ''c'' = 0.9511(7) nm. There are 58 independent atomic sites in the unit cell. Three of them are occupied by either B or Si atoms (mixed-occupancy sites), one is a Si bridge site and one is Y site. From the remaining 53 boron sites, 48 form icosahedra and 5 are bridging sites. Atomic coordinates and site occupancies are summarized in table III.
The boron framework of YB41Si1.2 consists of five B12 icosahedra (I1–I5) and a B12Si3 polyhedron shown in figure 8a. An unusual linkage is depicted in figure 8b, where two B12-I5 icosahedra connect via two B atoms of each icosahedron forming an imperfect square. The boron framework of YB41Si1.2 can be described as a layered structure where two boron networks (figures 9a,b) stack along the ''z''-axis. One boron network consists of 3 icosahedra I1, I2 and I3 and is located in the ''z'' = 0 plane; another network consists of the icosahedron I5 and the B12Si3 polyhedron and lies at ''z'' = 0.5. The icosahedron I4 bridges these networks, and thus its height along the ''z''-axis is 0.25.
The I4 icosahedra link two networks along the ''c''-axis and therefore form an infinite chain of icosahedra along this axis as shown in figure 10. The unusually short distances (0.4733 and 0.4788 nm) between the neighboring icosahedra in this direction result in the relatively small ''c''-axis lattice constant of 0.95110(7) nm in this compound – other borides with a similar icosahedral chain have this value larger than 1.0 nm. However, the bonding distances between the apex B atoms (0.1619 and 0.1674 nm) of neighboring I4 icosahedra are usual for the considered metal borides.
Another unusual feature of YB41Si1.2 is the 100% occupancy of the Y site. In most icosahedron-based metal borides, metal sites have rather low site occupancy, for example, about 50% for YB66 and 60–70% for REAlB14. When the Y site is replaced by rare-earth elements, REB41Si1.2 can have an antiferromagnetic-like ordering because of this high site occupancy.
Homologous icosahedron-based rare-earth borides
Rare-earth borides REB15.5CN, REB22C2N and REB28.5C4 are homologous, i.e. have a similar crystal structure, to B4C. The latter has a structure typical of icosahedron-based borides, as shown in figure 11a. There, B12 icosahedra form a rhombohedral lattice unit (space group: ''Rm'' (No. 166), lattice constants: ''a'' = 0.56 nm and ''c'' = 1.212 nm) surrounding a C-B-C chain that resides at the center of the lattice unit, and both C atoms bridge the neighboring three icosahedra. This structure is layered: as shown in figure 11b, B12 icosahedra and bridgingcarbons
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes up o ...
form a network plane that spreads parallel to the ''c''-plane and stacks along the ''c''-axis.
These homologous compounds have two basic structure units – the B12 icosahedron and the B6 octahedron. The network plane of B4C structure can be periodically replaced by a B6 octahedron layer so that replacement of
every third, fourth and fifth layer would correspond to REB15.5CN, REB22C2N and REB28.5C4, respectively. The B6 octahedron is smaller than the B12 icosahedron; therefore, rare-earth elements can reside in the space created by the replacement. The stacking sequences of B4C, REB15.5CN, REB22C2N and REB28.5C4 are shown in figures 12a, b, c and d, respectively. High-resolution transmission electron microscopy
Transmission electron microscopy (TEM) is a microscopy technique in which a beam of electrons is transmitted through a specimen to form an image. The specimen is most often an ultrathin section less than 100 nm thick or a suspension on a g ...
(HRTEM) lattice images of the latter three compounds, added to Fig. 12, do confirm the stacking sequence of each compound. The symbols 3T, 12R and 15R in brackets indicate the number of layers necessary to complete the stacking sequence, and T and R refer to trigonal and rhombohedral. Thus, REB22C2N and REB28.5C4 have rather large ''c''-lattice constants.
Because of the small size of the B6 octahedra, they cannot interconnect. Instead, they bond to the B12 icosahedra in the neighboring layer, and this decreases bonding strength in the ''c''-plane. Nitrogen atoms strengthen the bonding in the ''c''-plane by bridging three icosahedra, like C atoms in the C-B-C chain. Figure 13 depicts the ''c''-plane network revealing the alternate bridging of the boron icosahedra by N and C atoms. Decreasing the number of the B6 octahedra diminishes the role of nitrogen because the C-B-C chains start bridging the icosahedra. On the other hand, in MgB9N the B6 octahedron layer and the B12 icosahedron layer stack alternatively and there is no C-B-C chains; thus only N atoms bridge the B12 icosahedra. However, REB9N compounds have not been identified yet.
Sc, Y, Ho, Er, Tm and Lu are confirmed to form REB15.5CN-type compounds. Single-crystal structure analysis yielded trigonal symmetry for ScB15.5CN (space group ''Pm1'' (No.164) with ''a'' = 0.5568(2) and ''c'' = 1.0756(2) nm), and the deduced atomic coordinates are summarized in table IVa.
REB22C2N was synthesized for Y, Ho, Er, Tm and Lu. The crystal structure, solved for a representative compound YB22C2N, belongs to the trigonal with space group ''Rm'' (No.166); it has six formula units in the unit cell and lattice constants ''a'' = ''b'' = 0.5623(0) nm and ''c'' = 4.4785(3) nm. Atomic coordinates of YB22C2N are summarized in table IVb.
Y, Ho, Er, Tm and Lu also form REB28.5C4 which has a trigonal crystal structure with space group ''Rm''
(No. 166). Lattice constants of the representative compound YB28.5C4 are ''a'' = ''b'' = 0.56457(9) nm and ''c'' = 5.68873(13) nm and there are six formula units in the unit cell. Structure data of YB28.5C4 are
summarized in table IVc.
RExB12C0.33Si3.0
 Initially these were described as ternary RE-B-Si compounds, but later carbon was included to improve the structure description that resulted in a quaternary RE-B-C-Si composition. RExB12C0.33Si3.0 (RE=Y and Gd–Lu) have a unique crystal structure with two units – a cluster of B12 icosahedra and a Si8 ethane-like complex – and one bonding configuration (B12)3≡Si-C≡(B12)3. A representative compound of this group is YxB12C0.33Si3.0 (x=0.68). It has a trigonal crystal structure with space group ''Rm'' (No. 166) and lattice constants ''a'' = ''b'' = 1.00841(4) nm, ''c'' = 1.64714(5) nm, α = β = 90° and γ = 120°.
Initially these were described as ternary RE-B-Si compounds, but later carbon was included to improve the structure description that resulted in a quaternary RE-B-C-Si composition. RExB12C0.33Si3.0 (RE=Y and Gd–Lu) have a unique crystal structure with two units – a cluster of B12 icosahedra and a Si8 ethane-like complex – and one bonding configuration (B12)3≡Si-C≡(B12)3. A representative compound of this group is YxB12C0.33Si3.0 (x=0.68). It has a trigonal crystal structure with space group ''Rm'' (No. 166) and lattice constants ''a'' = ''b'' = 1.00841(4) nm, ''c'' = 1.64714(5) nm, α = β = 90° and γ = 120°.
 The crystal has layered structure. Figure 15 shows a network of boron icosahedra that spreads parallel to the (001) plane, connecting with four neighbors through B1–B1 bonds. The C3 and Si3 site atoms strengthen the network by bridging the boron icosahedra. Contrary to other boron-rich icosahedral compounds, the boron icosahedra from different layers are not directly bonded. The icosahedra within one layer are linked through Si8 ethane-like clusters with (B12)3≡Si-C≡(B12)3 bonds, as shown in figures 16a and b.
There are eight atomic sites in the unit cell: one yttrium Y, four boron B1–B4, one carbon C3 and three silicon sites Si1–Si3. Atomic coordinates, site occupancy and isotropic displacement factors are listed in table Va; 68% of the Y sites are randomly occupied and remaining Y sites are vacant. All boron sites and Si1 and Si2 sites are fully occupied. The C3 and Si3 sites can be occupied by either carbon or silicon atoms (mixed occupancy) with a probability of about 50%. Their separation is only 0.413 Å, and thus either the C3 or Si3 sites, but not both, are occupied. These sites form Si-C pairs, but not Si-Si or C-C pairs. The distances between the C3 and Si3 sites and the surrounding sites for YxB12C0.33Si3.0 are summarized in table Vb and the overall crystal structure is shown in figure 14.
Salvador ''et al.'' reported an isotypic terbium compound
Tb3–xC2Si8(B12)3. Most parts of the crystal structure are the same as those described above; however, its bonding configuration is deduced as (B12)3≡C-C≡(B12)3 instead of (B12)3≡Si-C≡(B12)3. The authors intentionally added carbon to grow single crystals whereas the previous crystals were accidentally contaminated by carbon during their growth. Thus, higher carbon concentration was achieved. Existence of both bonding schemes of (B12)3≡Si-C≡(B12)3 and (B12)3≡C-C≡(B12)3 suggests the occupancy of the carbon sites of 50–100%. On the other hand, (B12)3≡Si-Si≡(B12)3 bonding scheme is unlikely because of too short Si-Si distance, suggesting that the minimum carbon occupancy at the site is 50%. Some B atoms may replace C atoms at the C3 site, as previously assigned to the B site. However, the carbon occupation is more likely because the site is tetrahedrally coordinated whereas the B occupation of the site needs an extra electron to complete tetrahedral bonding. Thus, carbon is indispensable for this group of compounds.
The crystal has layered structure. Figure 15 shows a network of boron icosahedra that spreads parallel to the (001) plane, connecting with four neighbors through B1–B1 bonds. The C3 and Si3 site atoms strengthen the network by bridging the boron icosahedra. Contrary to other boron-rich icosahedral compounds, the boron icosahedra from different layers are not directly bonded. The icosahedra within one layer are linked through Si8 ethane-like clusters with (B12)3≡Si-C≡(B12)3 bonds, as shown in figures 16a and b.
There are eight atomic sites in the unit cell: one yttrium Y, four boron B1–B4, one carbon C3 and three silicon sites Si1–Si3. Atomic coordinates, site occupancy and isotropic displacement factors are listed in table Va; 68% of the Y sites are randomly occupied and remaining Y sites are vacant. All boron sites and Si1 and Si2 sites are fully occupied. The C3 and Si3 sites can be occupied by either carbon or silicon atoms (mixed occupancy) with a probability of about 50%. Their separation is only 0.413 Å, and thus either the C3 or Si3 sites, but not both, are occupied. These sites form Si-C pairs, but not Si-Si or C-C pairs. The distances between the C3 and Si3 sites and the surrounding sites for YxB12C0.33Si3.0 are summarized in table Vb and the overall crystal structure is shown in figure 14.
Salvador ''et al.'' reported an isotypic terbium compound
Tb3–xC2Si8(B12)3. Most parts of the crystal structure are the same as those described above; however, its bonding configuration is deduced as (B12)3≡C-C≡(B12)3 instead of (B12)3≡Si-C≡(B12)3. The authors intentionally added carbon to grow single crystals whereas the previous crystals were accidentally contaminated by carbon during their growth. Thus, higher carbon concentration was achieved. Existence of both bonding schemes of (B12)3≡Si-C≡(B12)3 and (B12)3≡C-C≡(B12)3 suggests the occupancy of the carbon sites of 50–100%. On the other hand, (B12)3≡Si-Si≡(B12)3 bonding scheme is unlikely because of too short Si-Si distance, suggesting that the minimum carbon occupancy at the site is 50%. Some B atoms may replace C atoms at the C3 site, as previously assigned to the B site. However, the carbon occupation is more likely because the site is tetrahedrally coordinated whereas the B occupation of the site needs an extra electron to complete tetrahedral bonding. Thus, carbon is indispensable for this group of compounds.
Scandium compounds
 Scandium has the smallest atomic and ionic (3+) radii (1.62 and 0.885 Å, respectively) among the rare-earth elements. It forms several icosahedron-based borides which are not found for other rare-earth elements; however, most of them are ternary Sc-B-C compounds. There are many boron-rich phases in the boron-rich corner of Sc-B-C phase diagram, as shown in figure 17. A slight variation of the composition can produce ScB19, ScB17C0.25, ScB15C0.8 and ScB15C1.6; their crystal structures are unusual for borides and are very different from each other.
Scandium has the smallest atomic and ionic (3+) radii (1.62 and 0.885 Å, respectively) among the rare-earth elements. It forms several icosahedron-based borides which are not found for other rare-earth elements; however, most of them are ternary Sc-B-C compounds. There are many boron-rich phases in the boron-rich corner of Sc-B-C phase diagram, as shown in figure 17. A slight variation of the composition can produce ScB19, ScB17C0.25, ScB15C0.8 and ScB15C1.6; their crystal structures are unusual for borides and are very different from each other.
ScB19+xSiy
ScB19+xSiy has a tetragonal crystal structure with space group ''P''41212 (No. 92) or ''P''43212 and lattice constants of ''a'', ''b'' = 1.03081(2) and ''c'' = 1.42589(3) nm; it is isotypic to the α-AlB12 structure type. There are 28 atomic sites in the unit cell, which are assigned to 3 scandium atoms, 24 boron atoms and one silicon atom. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VI. The boron framework of ScB19+xSiy is based on one B12 icosahedron and one B22 unit. This unit can be observed in β-tetragonal boron and is a modification of the B20 unit of α-AlB12 (or B19 unit in early reports). The B20 unit is a twinned icosahedron made from B13 to B22 sites with two vacant sites and one B atom (B23) bridging both sides of the unit. The twinned icosahedron is shown in figure 18a. B23 was treated as an isolated atom in the early reports; it is bonded to each twinned icosahedra through B18 and to another icosahedron through B5 site. If the twinned icosahedra were independent without twinning then B23 would be a bridge site linking three icosahedra. However, because of twinning, B23 shifts closer to the twinned icosahedra than another icosahedron; thus B23 is currently treated as a member of the twinned icosahedra. In ScB19+xSiy, the two B24 sites which correspond to the vacant sites in the B20 unit are partially occupied; thus, the unit should be referred to as a B22 cluster which is occupied by about 20.6 boron atoms. Scandium atoms occupy 3 of 5 Al sites of α-AlB12, that is Sc1, Sc2 and Sc3 correspond to Al4, Al1 and Al2 sites of α-AlB12, respectively. The Al3 and Al5 sites are empty for ScB19+xSiy, and the Si site links two B22 units. This phase also exists without silicon. Figure 19a shows the network of boron icosahedra in the boron framework of ScB19+xSiy. In this network, 4 icosahedra form a supertetrahedron
In geometry, a tetrahedron (plural: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular faces, six straight edges, and four vertex corners. The tetrahedron is the simplest of all the o ...
(figure 18b); its one edge is parallel to the ''a''-axis, and the icosahedra on this edge make up a chain along the ''a''-axis. The opposite edge of the supertetrahedron is parallel to the ''b''-axis and the icosahedra on this edge form a chain along the ''b''-axis. As shown in figure 19, there are wide tunnels surrounded by the icosahedron arrangement along the ''a''- and ''b''-axes. The tunnels are filled by the B22 units which strongly bond to the surrounding icosahedra; the connection of the B22 units is helical and it runs along the ''c''-axis as shown in figure 19b. Scandium atoms occupy the voids in the boron network as shown in figure 19c, and the Si atoms bridge the B22 units.
ScB17C0.25
 Very small amount of carbon is sufficient to stabilize "ScB17C0.25". This compound has a broad composition range, namely ScB16.5+xC0.2+y with x ≤ 2.2 and y ≤ 0.44. ScB17C0.25 has a hexagonal crystal structure with space group ''P6mmm'' (No. 199) and lattice constants ''a, b'' = 1.45501(15) nm and ''c'' = 0.84543(16) nm.
There are 19 atomic sites in the unit cell, which are assigned to one scandium site Sc, 14 boron sites B1–B14 having 100% occupancy, two boron-carbon mixed-occupancy sites B/C15 and B/C16, and two partial-occupancy boron sites B17 and B18. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VII. Although a very small amount of carbon (less than 2 wt%!) plays an important role in the phase stability, carbon does not have its own sites but shares with boron two interstitial sites B/C15 and B/C16.
There are two inequivalent B12 icosahedra, I1 and I2, which are constructed by the B1–B5 and B8–B12 sites, respectively. A "tube" is another characteristic structure unit of ScB17C0.25. It extends along the ''c''-axis and consists of B13, B14, B17 and B18 sites where B13 and B14 form 6-membered rings. B17 and B18 sites also form 6-membered rings; however, their mutual distances (0.985 Å for B17 and 0.955 Å for B18) are too short for a simultaneous occupation of the neighboring sites. Therefore, boron atoms occupy 2nd neighbor site forming a triangle. The occupancies of B17 and B18 sites should be 50%, but the structure analysis suggests larger values. The crystal structure viewed along the ''a''-axis is shown in figure 20, which suggests that the ScB17C0.25 is a layered material. Two layers, respectively constructed by the icosahedra I1 and I2, alternatively stack along the ''c''-axis. However, the ScB17C0.25 crystal is not layered. For example, during arc-melting, ScB17C0.25 needle crystals violently grow along the ''c''-axis – this never happens in layered compounds. The crystal structure viewed along the ''c''-axis is shown in figure 21a. The icosahedra I1 and I2 form a ring centered by the "tube" shown in figure 21b, which probably governs the properties of the ScB17C0.25 crystal. B/C15 and B/C16 mixed-occupancy sites interconnect the rings. A structural similarity can be seen between ScB17C0.25 and BeB3.
Figures 22a and b present HRTEM lattice images and electron diffraction patterns taken along the
Very small amount of carbon is sufficient to stabilize "ScB17C0.25". This compound has a broad composition range, namely ScB16.5+xC0.2+y with x ≤ 2.2 and y ≤ 0.44. ScB17C0.25 has a hexagonal crystal structure with space group ''P6mmm'' (No. 199) and lattice constants ''a, b'' = 1.45501(15) nm and ''c'' = 0.84543(16) nm.
There are 19 atomic sites in the unit cell, which are assigned to one scandium site Sc, 14 boron sites B1–B14 having 100% occupancy, two boron-carbon mixed-occupancy sites B/C15 and B/C16, and two partial-occupancy boron sites B17 and B18. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VII. Although a very small amount of carbon (less than 2 wt%!) plays an important role in the phase stability, carbon does not have its own sites but shares with boron two interstitial sites B/C15 and B/C16.
There are two inequivalent B12 icosahedra, I1 and I2, which are constructed by the B1–B5 and B8–B12 sites, respectively. A "tube" is another characteristic structure unit of ScB17C0.25. It extends along the ''c''-axis and consists of B13, B14, B17 and B18 sites where B13 and B14 form 6-membered rings. B17 and B18 sites also form 6-membered rings; however, their mutual distances (0.985 Å for B17 and 0.955 Å for B18) are too short for a simultaneous occupation of the neighboring sites. Therefore, boron atoms occupy 2nd neighbor site forming a triangle. The occupancies of B17 and B18 sites should be 50%, but the structure analysis suggests larger values. The crystal structure viewed along the ''a''-axis is shown in figure 20, which suggests that the ScB17C0.25 is a layered material. Two layers, respectively constructed by the icosahedra I1 and I2, alternatively stack along the ''c''-axis. However, the ScB17C0.25 crystal is not layered. For example, during arc-melting, ScB17C0.25 needle crystals violently grow along the ''c''-axis – this never happens in layered compounds. The crystal structure viewed along the ''c''-axis is shown in figure 21a. The icosahedra I1 and I2 form a ring centered by the "tube" shown in figure 21b, which probably governs the properties of the ScB17C0.25 crystal. B/C15 and B/C16 mixed-occupancy sites interconnect the rings. A structural similarity can be seen between ScB17C0.25 and BeB3.
Figures 22a and b present HRTEM lattice images and electron diffraction patterns taken along the 001 001, O01, or OO1 may refer to:
*1 (number), a number, a numeral
*001, fictional British agent, see 00 Agent
*001, former emergency telephone number for the Norwegian fire brigade (until 1986)
*AM-RB 001, the code-name for the Aston Martin Valkyrie ...
and 10crystalline directions, respectively. The HRTEM lattice image of figure 22a reproduces well the (''a, b'') plane of the crystal structure shown in figure 21a, with the clearly visible rings membered by icosahedra I1 and I2 and centered by the "tube". Figure 22b proves that ScB17C0.25 does not have layered character but its ''c''-axis direction is built up by the ring-like structure and tubular structures.
Sc0.83–xB10.0–yC0.17+ySi0.083–z
 Sc0.83–xB10.0–yC0.17+ySi0.083–z (''x'' = 0.030, ''y'' = 0.36 and ''z'' = 0.026) has a cubic crystal structure with space group ''F3m'' (No. 216) and lattice constant ''a'' = 2.03085(5) nm. This compound was initially identified as ScB15C0.8 (phase I in the Sc-B-C phase diagram of figure 17). A small amount of Si was added into the floating zone crystal growth and thus this phase is a quaternary compound. Its rare cubic structure has 26 sites in the unit cell: three Sc sites, two Si sites, one C site and 20 B sites; 4 out of 20 B sites are boron-carbon mixed-occupancy sites. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VIII.
In the unit cell, there are three independent icosahedra, I1, I2 and I3, and a B10 polyhedron which are formed by the B1–B4, B5–B8, B9–B13 and B14–B17 sites, respectively.There are more than 4 sites in total among, say, B5–B8 sites, but many of them are equivalent by symmetry and thus do not have an individual label. The B10 polyhedron has not been observed previously and it is shown in figure 23. The icosahedron I2 has a boron-carbon mixed-occupancy site B,C6 whose occupancy is B/C=0.58/0.42. Remaining 3 boron-carbon mixed-occupancy sites are bridge sites; C and Si sites are also bridge sites.
More than 1000 atoms are available in the unit cell, which is built up by large structure units such as two supertetrahedra T(1) and T(2) and one superoctahedron O(1). As shown in figure 24a, T(1) consists of 4 icosahedra I(1) which have no direct bonding but are bridged by four B and C20 atoms. These atoms also form tetrahedron centered by the Si2 sites. The supertetrahedron T(2) that consists of 4 icosahedra I(2) is the same as shown in figure 18b; its mixed-occupancy sites B and C6 directly bond with each other. The superoctahedron O(1) consists of 6 icosahedra I(3) and bridge sites B, C18, C1 and Si1; here Si1 and C1 exhibit a tetrahedral arrangement at the center of O(1). The B10 polyhedra also arrange octahedrally, without the central atom, as shown in figure 24c where the B and C19 atoms bridge the B10 polyhedra to form the octahedral supercluster of the B10 polyhedra.
Sc0.83–xB10.0–yC0.17+ySi0.083–z (''x'' = 0.030, ''y'' = 0.36 and ''z'' = 0.026) has a cubic crystal structure with space group ''F3m'' (No. 216) and lattice constant ''a'' = 2.03085(5) nm. This compound was initially identified as ScB15C0.8 (phase I in the Sc-B-C phase diagram of figure 17). A small amount of Si was added into the floating zone crystal growth and thus this phase is a quaternary compound. Its rare cubic structure has 26 sites in the unit cell: three Sc sites, two Si sites, one C site and 20 B sites; 4 out of 20 B sites are boron-carbon mixed-occupancy sites. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table VIII.
In the unit cell, there are three independent icosahedra, I1, I2 and I3, and a B10 polyhedron which are formed by the B1–B4, B5–B8, B9–B13 and B14–B17 sites, respectively.There are more than 4 sites in total among, say, B5–B8 sites, but many of them are equivalent by symmetry and thus do not have an individual label. The B10 polyhedron has not been observed previously and it is shown in figure 23. The icosahedron I2 has a boron-carbon mixed-occupancy site B,C6 whose occupancy is B/C=0.58/0.42. Remaining 3 boron-carbon mixed-occupancy sites are bridge sites; C and Si sites are also bridge sites.
More than 1000 atoms are available in the unit cell, which is built up by large structure units such as two supertetrahedra T(1) and T(2) and one superoctahedron O(1). As shown in figure 24a, T(1) consists of 4 icosahedra I(1) which have no direct bonding but are bridged by four B and C20 atoms. These atoms also form tetrahedron centered by the Si2 sites. The supertetrahedron T(2) that consists of 4 icosahedra I(2) is the same as shown in figure 18b; its mixed-occupancy sites B and C6 directly bond with each other. The superoctahedron O(1) consists of 6 icosahedra I(3) and bridge sites B, C18, C1 and Si1; here Si1 and C1 exhibit a tetrahedral arrangement at the center of O(1). The B10 polyhedra also arrange octahedrally, without the central atom, as shown in figure 24c where the B and C19 atoms bridge the B10 polyhedra to form the octahedral supercluster of the B10 polyhedra.
 Using these large polyhedra, the crystal structure of Sc0.83–xB10.0–yC0.17+ySi0.083–z can be described as shown in figure 25. Owing to the crystal symmetry, the tetrahedral coordination between these superstructure units is again a key factor. The supertetrahedron T(1) lies at the body center and at the edge center of the unit cell. The superoctahedra O(1) locate at the body center (0.25, 0.25, 0.25) of the quarter of the unit cell. They coordinate tetrahedrally around T(1) forming a giant tetrahedron. The supertetrahedra T(2) are located at the symmetry-related positions (0.25, 0.25, 0.75); they also form a giant tetrahedron surrounding T(1). Edges of both giant tetrahedra orthogonally cross each other at their centers; at those edge centers, each B10 polyhedron bridges all the super-structure clusters T(1), T(2) and O(1). The superoctahedron built of B10 polyhedra is located at each cubic face center.
Scandium atoms reside in the voids of the boron framework. Four Sc1 atoms form a tetrahedral arrangement inside the B10 polyhedron-based superoctahedron. Sc2 atoms sit between the B10 polyhedron-based superoctahedron and the O(1) superoctahedron. Three Sc3 atoms form a triangle and are surrounded by three B10 polyhedra, a supertetrahedron T(1) and a superoctahedron O(1).
Using these large polyhedra, the crystal structure of Sc0.83–xB10.0–yC0.17+ySi0.083–z can be described as shown in figure 25. Owing to the crystal symmetry, the tetrahedral coordination between these superstructure units is again a key factor. The supertetrahedron T(1) lies at the body center and at the edge center of the unit cell. The superoctahedra O(1) locate at the body center (0.25, 0.25, 0.25) of the quarter of the unit cell. They coordinate tetrahedrally around T(1) forming a giant tetrahedron. The supertetrahedra T(2) are located at the symmetry-related positions (0.25, 0.25, 0.75); they also form a giant tetrahedron surrounding T(1). Edges of both giant tetrahedra orthogonally cross each other at their centers; at those edge centers, each B10 polyhedron bridges all the super-structure clusters T(1), T(2) and O(1). The superoctahedron built of B10 polyhedra is located at each cubic face center.
Scandium atoms reside in the voids of the boron framework. Four Sc1 atoms form a tetrahedral arrangement inside the B10 polyhedron-based superoctahedron. Sc2 atoms sit between the B10 polyhedron-based superoctahedron and the O(1) superoctahedron. Three Sc3 atoms form a triangle and are surrounded by three B10 polyhedra, a supertetrahedron T(1) and a superoctahedron O(1).
ScB14–xCx (x = 1.1) and ScB15C1.6
ScB14–xCx has an orthorhombic crystal structure with space group ''Imma'' (No. 74) and lattice constants of ''a'' = 0.56829(2), ''b'' = 0.80375(3) and ''c'' = 1.00488(4) nm. The crystal structure of ScB14–xCx is isotypic to that of MgAlB14 where Sc occupies the Mg site, the Al site is empty and the boron bridge site is a B/C mixed-occupancy site with the occupancy of B/C = 0.45/0.55. The occupancy of the Sc site in flux-grown single crystals is 0.964(4), i.e. almost 1. Solid-state powder-reaction growth resulted in lower Sc site occupancy and in the resulting chemical composition ScB15C1.6. The B-C bonding distance 0.1796(3) nm between the B/C bridge sites is rather long as compared with that (0.15–0.16 nm) of an ordinary B-C covalent bond, that suggests weak bonding between the B/C bridge sites.Sc4.5–xB57–y+zC3.5–z
Sc4.5–xB57–y+zC3.5–z (x = 0.27, y = 1.1, z = 0.2) has an orthorhombic crystal structure with space group ''Pbam'' (No. 55) and lattice constants of ''a'' = 1.73040(6), ''b'' = 1.60738(6) and ''c'' = 1.44829(6) nm. This phase is indicated as ScB12.5C0.8 (phase IV) in the phase diagram of figure 17. This rare orthorhombic structure has 78 atomic positions in the unit cell: seven partially occupied Sc sites, four C sites, 66 B sites including three partially occupied sites and one B/C mixed-occupancy site. Atomic coordinates, site occupancies and isotropic displacement factors are listed in table IX. More than 500 atoms are available in the unit cell. In the crystal structure, there are six structurally independent icosahedra I1–I6, which are constructed from B1–B12, B13–B24, B25–B32, B33–B40, B41–B44 and B45–B56 sites, respectively; B57–B62 sites form a B8 polyhedron. The Sc4.5–xB57–y+zC3.5–z crystal structure is layered, as shown in figure 26. This structure has been described in terms of two kinds of boron icosahedron layers, L1 and L2. L1 consists of the icosahedra I3, I4 and I5 and the C65 "dimer", and L2 consists of the icosahedra I2 and I6. I1 is sandwiched by L1 and L2 and the B8 polyhedron is sandwiched by L2. An alternative description is based on the same B12(B12)12supericosahedron as in the YB66 structure. In the YB66 crystal structure, the supericosahedra form 3-dimensional boron framework as shown in figure 5. In this framework, the neighboring supericosahedra are rotated 90° with respect to each other. On the contrary, in Sc4.5–xB57–y+zC3.5–z the supericosahedra form a 2-dimensional network where the 90° rotation relation is broken because of the orthorhombic symmetry. The planar projections of the supericosahedron connection in Sc4.5–xB57–y+zC3.5–z and YB66 are shown in figures 27a and b, respectively. In the YB66 crystal structure, the neighboring 2-dimensional supericosahedron connections are out-of-phase for the rotational relation of the supericosahedron. This allows 3-dimensional stacking of the 2-dimensional supericosahedron connection while maintaining the cubic symmetry. The B80 boron cluster occupies the large space between four supericosahedra as described in the REB66 section. On the other hand, the 2-dimensional supericosahedron networks in the Sc4.5–xB57–y+zC3.5–z crystal structure stack in-phase along the ''z''-axis. Instead of the B80 cluster, a pair of the I2 icosahedra fills the open space staying within the supericosahedron network, as shown in figure 28 where the icosahedron I2 is colored in yellow. All Sc atoms except for Sc3 reside in large spaces between the supericosahedron networks, and the Sc3 atom occupies a void in the network as shown in figure 26. Because of the small size of Sc atom, the occupancies of the Sc1–Sc5 sites exceed 95%, and those of Sc6 and Sc7 sites are approximately 90% and 61%, respectively (see table IX).Sc3.67–xB41.4–y–zC0.67+zSi0.33–w
 Sc3.67–xB41.4–y–zC0.67+zSi0.33–w (x = 0.52, y = 1.42, z = 1.17 and w = 0.02) has a hexagonal crystal structure with space group ''Pm2'' (No. 187) and lattice constants ''a'' = ''b'' = 1.43055(8) and ''c'' = 2.37477(13) nm. Single crystals of this compound were obtained as an intergrowth phase in a float-zoned single crystal of Sc0.83–xB10.0–yC0.17+ySi0.083–z. This phase is not described in the phase diagram of figure 17 because it is a quaternary compound. Its hexagonal structure is rare and has 79 atomic positions in the unit cell: eight partially occupied Sc sites, 62 B sites, two C sites, two Si sites and six B/C sites. Six B sites and one of the two Si sites have partial occupancies. The associated atomic coordinates, site occupancies and isotropic displacement factors are listed in table X.
There are seven structurally independent icosahedra I1–I7 which are formed by B1–B8, B9–B12, B13–B20, B/C21–B24, B/C25–B29, B30–B37 and B/C38–B42 sites, respectively; B43–B46 sites form the B9 polyhedron and B47–B53 sites construct the B10 polyhedron. B54–B59 sites form the irregularly shaped B16 polyhedron in which only 10.7 boron atoms are available because most of sites are too close to each other to be occupied simultaneously. Ten bridging sites C60–B69 interconnect polyhedron units or other bridging sites to form a 3D boron framework structure. One description of the crystal structure uses three pillar-like units that extend along the ''c''-axis that however results in undesired overlaps between those three pillar-like units. An alternative is to define two pillar-like structure units. Figure 29 shows the boron framework structure of Sc3.67–xB41.4–y–zC0.67+zSi0.33–w viewed along the ''c''-axis, where the pillar-like units P1 and P2 are colored in dark green and light green respectively and are bridged by yellow icosahedra I4 and I7.
These pillar-like units P1 and P2 are shown in figures 30a and b, respectively. P1 consists of icosahedra I1 and I3, an irregularly shaped B16 polyhedron and other bridge site atoms where two supericosahedra can be seen above and below the B16 polyhedron. Each supericosahedron is formed by three icosahedra I1 and three icosahedra I3 and is the same as the supericosahedron O(1) shown in figure 24a.The P2 unit consists of icosahedra I2, I5 and I6, B10 polyhedron and other bridge site atoms. Eight Sc sites with occupancies between 0.49 (Sc8) and 0.98 (Sc1) spread over the boron framework.
As described above, this hexagonal phase originates from a cubic phase, and thus one may expect a similar structural element in these phases. There is an obvious relation between the hexagonal ''ab''-plane and the cubic (111) plane. Figures 31a and b show the hexagonal (001) and the cubic (111) planes, respectively. Both network structures are almost the same that allows intergrowth of the hexagonal phase in the cubic phase.
Sc3.67–xB41.4–y–zC0.67+zSi0.33–w (x = 0.52, y = 1.42, z = 1.17 and w = 0.02) has a hexagonal crystal structure with space group ''Pm2'' (No. 187) and lattice constants ''a'' = ''b'' = 1.43055(8) and ''c'' = 2.37477(13) nm. Single crystals of this compound were obtained as an intergrowth phase in a float-zoned single crystal of Sc0.83–xB10.0–yC0.17+ySi0.083–z. This phase is not described in the phase diagram of figure 17 because it is a quaternary compound. Its hexagonal structure is rare and has 79 atomic positions in the unit cell: eight partially occupied Sc sites, 62 B sites, two C sites, two Si sites and six B/C sites. Six B sites and one of the two Si sites have partial occupancies. The associated atomic coordinates, site occupancies and isotropic displacement factors are listed in table X.
There are seven structurally independent icosahedra I1–I7 which are formed by B1–B8, B9–B12, B13–B20, B/C21–B24, B/C25–B29, B30–B37 and B/C38–B42 sites, respectively; B43–B46 sites form the B9 polyhedron and B47–B53 sites construct the B10 polyhedron. B54–B59 sites form the irregularly shaped B16 polyhedron in which only 10.7 boron atoms are available because most of sites are too close to each other to be occupied simultaneously. Ten bridging sites C60–B69 interconnect polyhedron units or other bridging sites to form a 3D boron framework structure. One description of the crystal structure uses three pillar-like units that extend along the ''c''-axis that however results in undesired overlaps between those three pillar-like units. An alternative is to define two pillar-like structure units. Figure 29 shows the boron framework structure of Sc3.67–xB41.4–y–zC0.67+zSi0.33–w viewed along the ''c''-axis, where the pillar-like units P1 and P2 are colored in dark green and light green respectively and are bridged by yellow icosahedra I4 and I7.
These pillar-like units P1 and P2 are shown in figures 30a and b, respectively. P1 consists of icosahedra I1 and I3, an irregularly shaped B16 polyhedron and other bridge site atoms where two supericosahedra can be seen above and below the B16 polyhedron. Each supericosahedron is formed by three icosahedra I1 and three icosahedra I3 and is the same as the supericosahedron O(1) shown in figure 24a.The P2 unit consists of icosahedra I2, I5 and I6, B10 polyhedron and other bridge site atoms. Eight Sc sites with occupancies between 0.49 (Sc8) and 0.98 (Sc1) spread over the boron framework.
As described above, this hexagonal phase originates from a cubic phase, and thus one may expect a similar structural element in these phases. There is an obvious relation between the hexagonal ''ab''-plane and the cubic (111) plane. Figures 31a and b show the hexagonal (001) and the cubic (111) planes, respectively. Both network structures are almost the same that allows intergrowth of the hexagonal phase in the cubic phase.
Applications
The diversity of the crystal structures of rare-earth borides results in unusual physical properties and potential applications inthermopower
The Seebeck coefficient (also known as thermopower, thermoelectric power, and thermoelectric sensitivity) of a material is a measure of the magnitude of an induced thermoelectric voltage in response to a temperature difference across that material ...
generation. Thermal conductivity
The thermal conductivity of a material is a measure of its ability to conduct heat. It is commonly denoted by k, \lambda, or \kappa.
Heat transfer occurs at a lower rate in materials of low thermal conductivity than in materials of high thermal ...
of boron icosahedra based compounds is low because of their complex crystal structure; this property is favored for thermoelectric materials. On the other hand, these compounds exhibit very low (variable range hopping Variable-range hopping is a model used to describe carrier transport in a disordered semiconductor or in amorphous solid by hopping in an extended temperature range. It has a characteristic temperature dependence of
:\sigma= \sigma_0e^
where \sigm ...
type) p-type electrical conductivity
Electrical resistivity (also called specific electrical resistance or volume resistivity) is a fundamental property of a material that measures how strongly it resists electric current. A low resistivity indicates a material that readily allow ...
. Increasing the conductivity is a key issue for thermoelectric applications of these borides.
YB66 is used as a soft-X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
monochromator
A monochromator is an optical device that transmits a mechanically selectable narrow band of wavelengths of light or other radiation chosen from a wider range of wavelengths available at the input. The name is from the Greek roots ''mono-'', "si ...
for dispersing 1–2 keV synchrotron
A synchrotron is a particular type of cyclic particle accelerator, descended from the cyclotron, in which the accelerating particle beam travels around a fixed closed-loop path. The magnetic field which bends the particle beam into its closed p ...
radiation at some synchrotron radiation facilities. Contrary to thermoelectric applications, high thermal conductivity is desirable for synchrotron radiation monochromators. YB66 exhibits low, amorphous-like thermal conductivity. However, transition metal doping increases the thermal conductivity twice in YNb0.3B62 as compared to undoped YB66.
Notes
References
{{reflist, 35em, refs= {{cite journal, author=Chan J Y, Fronczek F R, Young D P, DiTusa J F and Adams P W 2002, title=Synthesis, Structure, and Superconductivity in Be1.09B3, journal=J. Solid State Chem., volume=163, page=385, year=2002, doi=10.1006/jssc.2001.9374, bibcode = 2002JSSCh.163..385C, issue=2 {{cite journal, vauthors=Vlasse M, Slack GA, Garbauskas M, Kasper JS, Viala JC , year=1986, journal=J. Solid State Chem., volume=63, issue=1, page=31, doi=10.1016/0022-4596(86)90149-0, title=The crystal structure of SiB6, bibcode = 1986JSSCh..63...31V {{cite journal, vauthors=Tanaka T, Okada S, Ishizawa Y , year=1997, title=Single Crystal Growth of a New YB50 Family Compound: YB44Si1.0, journal=J. Solid State Chem., volume=133, issue=1, page=55, doi=10.1006/jssc.1997.7317, bibcode = 1997JSSCh.133...55T {{cite journal, vauthors=Higashi I, Tanaka T, Kobayashi K, Ishizawa Y, Takami M , year=1997, title=Crystal Structure of YB41Si1.2, journal=J. Solid State Chem., volume=133, issue=1, page=11, doi=10.1006/jssc.1997.7307, bibcode = 1997JSSCh.133...11H {{cite journal, vauthors=Albert B, Hillebrecht H, year=2009, title=Boron: Elementary Challenge for Experimenters and Theoreticians, journal=Angew. Chem. Int. Ed., volume=48, issue=46, doi=10.1002/anie.200903246, pages=8640–68, pmid=19830749 {{cite journal, vauthors=Werheit H, Filipov V, Kuhlmann U, Schwarz U, Ambruster M, Leithe-Jasper A, Tanaka T, Higashi I, Lundstrom T, Gurin VN, Korusukova MM , year=2010, pages=023001, volume=11, journal=Sci. Technol. Adv. Mater., title= Raman effect in icosahedral boron-rich solids, doi= 10.1088/1468-6996/11/2/023001, pmid=27877328, pmc=5090270, bibcode = 2010STAdM..11b3001W, issue=2 {{cite journal, vauthors=Brandt NB, Gippius AA, Moshchalkov VV, Nyan KK, Gurin VN, Korsukova MM, Kuz'ma YB, year=1988, journal=Soviet Physics - Solid State, volume=30, issue=5, page=1380, url=http://oldjournals.ioffe.ru/ftt/1988/05/p1380-1383.pdf, title=Электрические и магнитные свойства соединений LnAlB14 (Ln = Tb, Dy, Но, Er, Lu){{Dead link, date=July 2019 , bot=InternetArchiveBot , fix-attempted=yes {{cite journal, vauthors=Korsukova MM, Gurin VN, Kuz'ma YB, Chaban NF, Chikhrij SI, Moshchalkov VV, Braudt NB, Gippius AA, Nyan KK , year=1989, title=Crystal Structure, Electrical, and Magnetic Properties of the New Ternary Compounds LnAIB14, journal=Physica Status Solidi A, volume=114, issue=1, page=265, doi=10.1002/pssa.2211140126, bibcode = 1989PSSAR.114..265K {{cite journal, authors=Korsukova MM, Gurin VN, Yu Y, Tergenius L-E and Lundstrom, year=1993, title=Crystal structural refinement of the new compound TmAlB14, journal=Journal of Alloys and Compounds, doi=10.1016/0925-8388(93)90397-6, volume=190, page=185, issue=2 {{cite journal, vauthors=Tanaka T, Okada S, Yu Y, Ishizawa Y , year=1997, title=A New Yttrium Boride: YB25, journal=J. Solid State Chem., volume=133, issue=1, page=122, doi=10.1006/jssc.1997.7328, bibcode = 1997JSSCh.133..122T {{cite book, author=Spear K E, editor=Alper, A. M., year=1976, title=Phase diagrams: materials science and technology Vol. IV, publisher=Academic Press, Inc., New York, page= 91, isbn=978-0-12-053204-9 {{cite journal, author=Seybolt A U, year=1960, journal=Trans. Am. Soc. Metals, volume=52, pages=971–989, title=An Exploration of High Boron Alloys {{cite journal, vauthors=Richards SM, Kasper JS, year=1969, title=The crystal structure of YB66, journal=Acta Crystallogr. B, volume=25, page=237, doi=10.1107/S056774086900207X, issue=2, doi-access=free {{cite journal, vauthors=Higashi I, Kobayashi K, Tanaka T, Ishizawa Y , year=1997, title=Structure Refinement of YB62 and YB56 of the YB66-Type Structure, journal=J. Solid State Chem., volume=133, issue=1, page=16, doi=10.1006/jssc.1997.7308, bibcode = 1997JSSCh.133...16H {{cite journal, vauthors=Slack GA, Oliver DW, Brower GD, Young JD , year=1977, title=Properties of melt-grown single crystals of "YB68", journal=J. Phys. Chem. Solids, volume=38, issue=1, page=45, doi=10.1016/0022-3697(77)90144-5, bibcode = 1977JPCS...38...45S {{cite journal, vauthors=Tanaka T, Kamiya K, Numazawa T, Sato A, Takenouchi S, year=2006, journal=Z. Kristallogr., volume=221, page=472, title=The effect of transition metal doping on thermal conductivity of YB66, doi=10.1524/zkri.2006.221.5-7.472, issue=5–7_2006, bibcode = 2006ZK....221..472T , s2cid=94723178 {{cite journal, vauthors=Tanaka T, Okada S, Ishizawa Y , year=1994, title=A new yttrium higher boride: YB50, journal=J. Alloys Compd., volume=205, issue=1–2, page=281, doi=10.1016/0925-8388(94)90802-8 {{cite journal, author=Higashi I, year=1983, title=Aluminum distribution in the boron framework of γ-AlB12, journal=J. Solid State Chem., volume=47, page=333, doi=10.1016/0022-4596(83)90027-0, bibcode = 1983JSSCh..47..333H, issue=3 {{cite journal, vauthors=Mori T, Tanaka T, year=1999, title=Magnetic Properties of Terbium B12 Icosahedral Boron-Rich Compounds, journal=J. Phys. Soc. Jpn., doi=10.1143/JPSJ.68.2033 , volume=68, page=2033, bibcode = 1999JPSJ...68.2033M, issue=6 {{cite journal, vauthors=Mori T, Tanaka T, year=1999, title=Magnetic transitions in B12 icosahedral boron-rich compounds TbB50 and TbB41Si1.2: Lattice constant dependence of the transition, journal=J. Alloys Compd., volume=288, issue=1–2, page=32, doi=10.1016/S0925-8388(99)00078-X {{cite journal, vauthors=Mori T, Tanaka T, year=2000, title=Magnetic Transitions in B12 Icosahedral Cluster Compounds REB50 (RE=Tb, Dy, Ho, Er), journal=J. Phys. Soc. Jpn., volume=69, page=579, doi=10.1143/JPSJ.69.579, bibcode = 2000JPSJ...69..579M, issue=2 {{cite journal, vauthors=Mironov A, Kazakov S, Jun J, Kapinski J , year=2002, title=MgNB9, a new magnesium nitridoboride, journal=Acta Crystallogr. C , volume= 58, doi=10.1107/S0108270102009253, pmid=12094025, issue=7, pages=i95-7 {{cite journal, vauthors=Leithe-Jasper A, Tanaka T, Bourgeois L, Mori T, Michiue Y , year=2004, title=New quaternary carbon and nitrogen stabilized polyborides: REB15.5CN (RE: Sc, Y, Ho, Er, Tm, Lu), crystal structure and compound formation, journal=J. Solid State Chem., volume=177, page=431, doi=10.1016/j.jssc.2003.02.003, bibcode = 2004JSSCh.177..431L, issue=2 {{cite journal, vauthors=Zhang FX, Leithe-Jasper A, Xu J, Matsui Y, Tanaka T, Okada S , year=2001, title=Novel Rare Earth Boron-Rich Solids, journal=J. Solid State Chem., volume=159, issue=1, page=174, doi=10.1006/jssc.2001.9147, bibcode = 2001JSSCh.159..174Z {{cite journal, vauthors=Zhang FX, Xu FF, Mori T, Liu QL, Sato A, Tanaka T , year=2001, title=Crystal structure of new rare-earth boron-rich solids: REB28.5C4, journal=J. Alloys Compd., volume=329, issue=1–2, page=168, doi=10.1016/S0925-8388(01)01581-X {{cite journal, vauthors=Zhang FZ, Xu FF, Leithe-Jasper A, Mori T, Tanaka T, Xu J, Sato A, Bando Y, Matsui Y , year=2001, title=Homologous Phases Built by Boron Clusters and Their Vibrational Properties, journal=Inorg. Chem., volume=40, doi=10.1021/ic010527s, pmid=11754276, issue=27, pages=6948–51 {{cite journal, vauthors=Zhang FX, Sato A, Tanaka T , year=2002, title=A New Boron-Rich Compound in the Y–B–Si Ternary System, journal=J. Solid State Chem., volume=164, page=361, doi=10.1006/jssc.2001.9508, bibcode = 2002JSSCh.164..361Z, issue=2 {{cite journal, vauthors=Zhang FX, Xu FF, Mori T, Liu QL, Tanaka T , year=2003, title=Novel rare-earth borosilicide RE1−xB12Si3.3−δ (RE=Y, Gd–Lu) (0≤x≤0.5, δ≈0.3): synthesis, crystal growth, structure analysis and properties, journal=J. Solid State Chem., volume=170, issue=1, page=75, doi=10.1016/S0022-4596(02)00025-7, bibcode = 2003JSSCh.170...75Z {{cite journal, vauthors=Zhang FX, Tanaka T, year=2003, journal=Z. Kristallogr. - New Cryst. Struct., volume=218, page=26, title=Crystal structure of dysprosium borosilicide, Dy0.7B12.33Si3, doi=10.1524/ncrs.2003.218.1.26, s2cid=96554379, doi-access=free {{cite journal, vauthors=Tanaka T, Sato A, Zhang FX , year=2009, title=Structure refinement of quaternary RE-B-C-Si compounds: Y3−x(B12)3(CSi)Si8 (x ≈ 0.96) and Dy3−x(B12)3(CSi)Si8 (x ≈ 0.90), journal=J. Phys.: Conf. Ser., volume=176, issue=1, page=012015, doi=10.1088/1742-6596/176/1/012015, bibcode = 2009JPhCS.176a2015T, format=free download , doi-access=free {{cite journal, vauthors=Salvador JR, Bilc D, Mahanti SD, Kanatzidis MG, year=2002, title=Gallium Flux Synthesis of Tb3−xC2Si8(B12)3: A Novel Quaternary Boron-Rich Phase Containing B12 Icosahedra, journal=Angew. Chem. Int. Ed., volume=41, doi=10.1002/1521-3773(20020301)41:5<844::AID-ANIE844>3.0.CO;2-R, pages=844–6, issue=5, pmid=12491355, url=http://www.cem.msu.edu/~kanatzid/Reprints/Tb3-xC2Si8%28B-12%293.pdf, access-date=2013-11-05, archive-url=https://web.archive.org/web/20131105021931/http://www.cem.msu.edu/~kanatzid/Reprints/Tb3-xC2Si8%28B-12%293.pdf, archive-date=2013-11-05, url-status=dead {{cite journal, vauthors=Tanaka T, Sato A, year=2001, journal=J. Solid State Chem., volume=160, page=394, title=Floating Zone Crystal Growth and Structure Analysis of a Novel ScB19 Family Compound, ScB19+xSiy, url=http://pubman.nims.go.jp/pubman/item/escidoc:458051:1/component/escidoc:458049/Floating+Zone+Crystal+Growth+and+Structure+Analysis+of+a+Novel+ScB19+Family+Compound.pdf, doi=10.1006/jssc.2001.9253, bibcode=2001JSSCh.160..394T, issue=2 {{cite journal, vauthors=Vlasse M, Naslain R, Kasper JS, Ploog K , year=1979, title=The crystal structure of tetragonal boron, journal=J. Less-Common Met., doi=10.1016/0022-5088(79)90067-5, volume=67, page=1 {{cite journal, author=Higashi I, year=2000, title=Crystal Chemistry of α-AlB12 and γ-AlB12, journal=J. Solid State Chem., volume=154, issue=1, page=168, doi=10.1006/jssc.2000.8831, bibcode = 2000JSSCh.154..168H {{cite journal, vauthors=Higashi I, Sakurai T, Atoda T , year=1977, title=Crystal structure of α-AlB12, journal=J. Solid State Chem., volume=20, issue=1, page=67, doi=10.1016/0022-4596(77)90052-4, bibcode = 1977JSSCh..20...67H {{cite journal, vauthors=Kasper JS, Vlasse M, Naslain R , year=1977, title=The α-AlB12 structure, journal=J. Solid State Chem., volume=20, page=281, doi=10.1016/0022-4596(77)90164-5, bibcode = 1977JSSCh..20..281K, issue=3 {{cite journal, vauthors=Tanaka T, Okada S, Gurin VN , year=1998, title=A new scandium boride: ScB19, journal=J. Alloys Compd., volume=267, issue=1–2, page=211, doi=10.1016/S0925-8388(97)00490-8 {{cite journal, vauthors=Shi Y, Leithe-Jasper A, Tanaka T , year=1999, title=New Ternary Compounds Sc3B0.75C3, Sc2B1.1C3.2, ScB15C1.60 and Subsolidus Phase Relations in the Sc–B–C System at 1700 °C, journal=J. Solid State Chem., volume=148, page=250, doi=10.1006/jssc.1999.8446, bibcode = 1999JSSCh.148..250S, issue=2 {{cite journal, vauthors=Leithe-Jasper A, Bourgeois L, Michiue Y, Shi Y, Tanaka T , year=2000, title=A Single-Crystal XRD and TEM Study of "ScB17C0.25", journal=J. Solid State Chem., volume=154, issue=1, page=130, doi=10.1006/jssc.2000.8822, bibcode = 2000JSSCh.154..130L {{cite journal, vauthors=Tanaka T, Sato A, year=2002, title=A Novel Boron-rich Scandium Borocarbosilicide; Sc0.83−xB10.0−yC0.17+ySi0.083−z (x=0.030, y=0.36 and z=0.026): Floating Zone Crystal Growth and Structure Analysis, journal=J. Solid State Chem., volume=165, issue=1, page=148, doi=10.1006/jssc.2002.9524, bibcode = 2002JSSCh.165..148T {{cite journal, vauthors=Leithe-Jasper A, Sato A, Tanaka T, year=2002, journal=Z. Kristallogr. - New Cryst. Struct., volume=217, page=319, title=Refinement of the crystal structure of zirconium dodecaboride, ZrB12, at 140 K and 293 K, doi=10.1524/ncrs.2002.217.jg.319, s2cid=94997047, doi-access=free {{cite journal, vauthors=Tanaka T, Yamamoto A, Sato A , year=2002, title=A Novel Boron-Rich Scandium Borocarbide; Sc4.5−xB57−y+zC3.5−z (x=0.27, y=1.1, z=0.2), journal=J. Solid State Chem., volume=168, issue=1, page=192, doi=10.1006/jssc.2002.9709, bibcode = 2002JSSCh.168..192T {{cite journal, vauthors=Tanaka T, Yamamoto A, Sato A , year=2002, title=A novel boron-rich quaternary scandium borocarbosilicide Sc3.67−xB41.4−y−zC0.67+zSi0.33−w, journal=J. Solid State Chem., volume=177, page=476, doi=10.1016/j.jssc.2003.02.006, bibcode = 2004JSSCh.177..476T, issue=2 {{cite journal, author=Mori T, year=2009, title=Novel physical properties of rare earth higher borides, journal=J. Phys.: Conf. Ser., format=free download, volume=176, issue=1, page=012036, doi=10.1088/1742-6596/176/1/012036, bibcode = 2009JPhCS.176a2036M, doi-access=free {{cite journal, vauthors=Wong J, Tanaka T, Rowen M, Schafers F, Muler BR, Rek ZU , year=1999, title=YB66 – a new soft X-ray monochromator for synchrotron radiation. II. Characterization, journal= Journal of Synchrotron Radiation, volume=6, page=1086, doi=10.1107/S0909049599009000, issue=6, doi-access=free Borides Crystallography