Boyer Reaction on:
[Wikipedia]
[Google]
[Amazon]
 In
In 
organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; ...
, the Schmidt reaction is an organic reaction
Organic reactions are chemical reactions involving organic compounds. The basic organic chemistry reaction types are addition reactions, elimination reactions, substitution reactions, pericyclic reactions, rearrangement reactions, Mechanistic Organ ...
in which an azide
In chemistry, azide is a linear, polyatomic anion with the formula and structure . It is the conjugate base of hydrazoic acid . Organic azides are organic compounds with the formula , containing the azide functional group. The dominant applic ...
reacts with a carbonyl derivative, usually an aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
, ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bo ...
, or carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
, under acidic conditions to give an amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituen ...
or amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it is ...
, with expulsion of nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
. It is named after Karl Friedrich Schmidt (1887–1971), who first reported it in 1924 by successfully converting benzophenone and hydrazoic acid
Hydrazoic acid, also known as hydrogen azide or azoimide, This also contains a detailed description of the contemporaneous production process. is a compound with the chemical formula . It is a colorless, volatile, and explosive liquid at room tem ...
to benzanilide
Benzanilide is the organic compound with the formula C6H5C(O)NHC6H5. It is a white solid. Commercially available, it may be prepared by treating benzoic acid with aniline
Aniline is an organic compound with the formula C6 H5 NH2. Consisting ...
. The intramolecular reaction Intramolecular in chemistry describes a process or characteristic limited within the structure of a single molecule, a property or phenomenon limited to the extent of a single molecule.
Examples
* intramolecular hydride transfer (transfer of a hy ...
was not reported until 1991 but has become important in the synthesis of natural products.
The reaction is effective with carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
s to give amines (above), and with ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bo ...
s to give amides (below).
Reaction mechanism
The reaction is closely related to theCurtius rearrangement
The Curtius rearrangement (or Curtius reaction or Curtius degradation), first defined by Theodor Curtius in 1885, is the thermal decomposition of an acyl azide to an isocyanate with loss of nitrogen gas. The isocyanate then undergoes attack by a va ...
except that in this reaction the acyl azide is produced by reaction of the carboxylic acid with hydrazoic acid via the protonated carboxylic acid, in a process akin to a Fischer esterification
Fischer is a German occupational surname, meaning fisherman. The name Fischer is the fourth most common German surname. The English version is Fisher.
People with the surname A
* Abraham Fischer (1850–1913) South African public official
* Ad ...
. An alternative, involving the formation of an acylium ion, becomes more important when the reaction takes place in concentrated acid (>90% sulfuric acid). (In the Curtius rearrangement, sodium azide and an acyl chloride are combined to quantitatively generate the acyl azide intermediate, and the rest of the reaction takes place under neutral conditions.)
The carboxylic acid Schmidt reaction starts with acylium
In chemistry, an acyl group is a moiety derived by the removal of one or more hydroxyl groups from an oxoacid, including inorganic acids. It contains a double-bonded oxygen atom and an alkyl group (). In organic chemistry, the acyl group (IUPAC n ...
ion 1 obtained from protonation
In chemistry, protonation (or hydronation) is the adding of a proton (or hydron, or hydrogen cation), (H+) to an atom, molecule, or ion, forming a conjugate acid. (The complementary process, when a proton is removed from a Brønsted–Lowry acid, ...
and loss of water. Reaction with hydrazoic acid
Hydrazoic acid, also known as hydrogen azide or azoimide, This also contains a detailed description of the contemporaneous production process. is a compound with the chemical formula . It is a colorless, volatile, and explosive liquid at room tem ...
forms the protonated azido ketone 2, which goes through a rearrangement reaction
In organic chemistry, a rearrangement reaction is a broad class of organic reactions where the carbon skeleton of a molecule is rearranged to give a structural isomer of the original molecule. Often a substituent moves from one atom to another ...
with the alkyl group R, migrating over the C-N bond with expulsion of nitrogen. The protonated isocyanate is attacked by water forming carbamate
In organic chemistry, a carbamate is a category of organic compounds with the general formula and structure , which are formally derived from carbamic acid (). The term includes organic compounds (e.g., the ester ethyl carbamate), formally o ...
4, which after deprotonation loses carbon dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transpar ...
to the amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituen ...
.
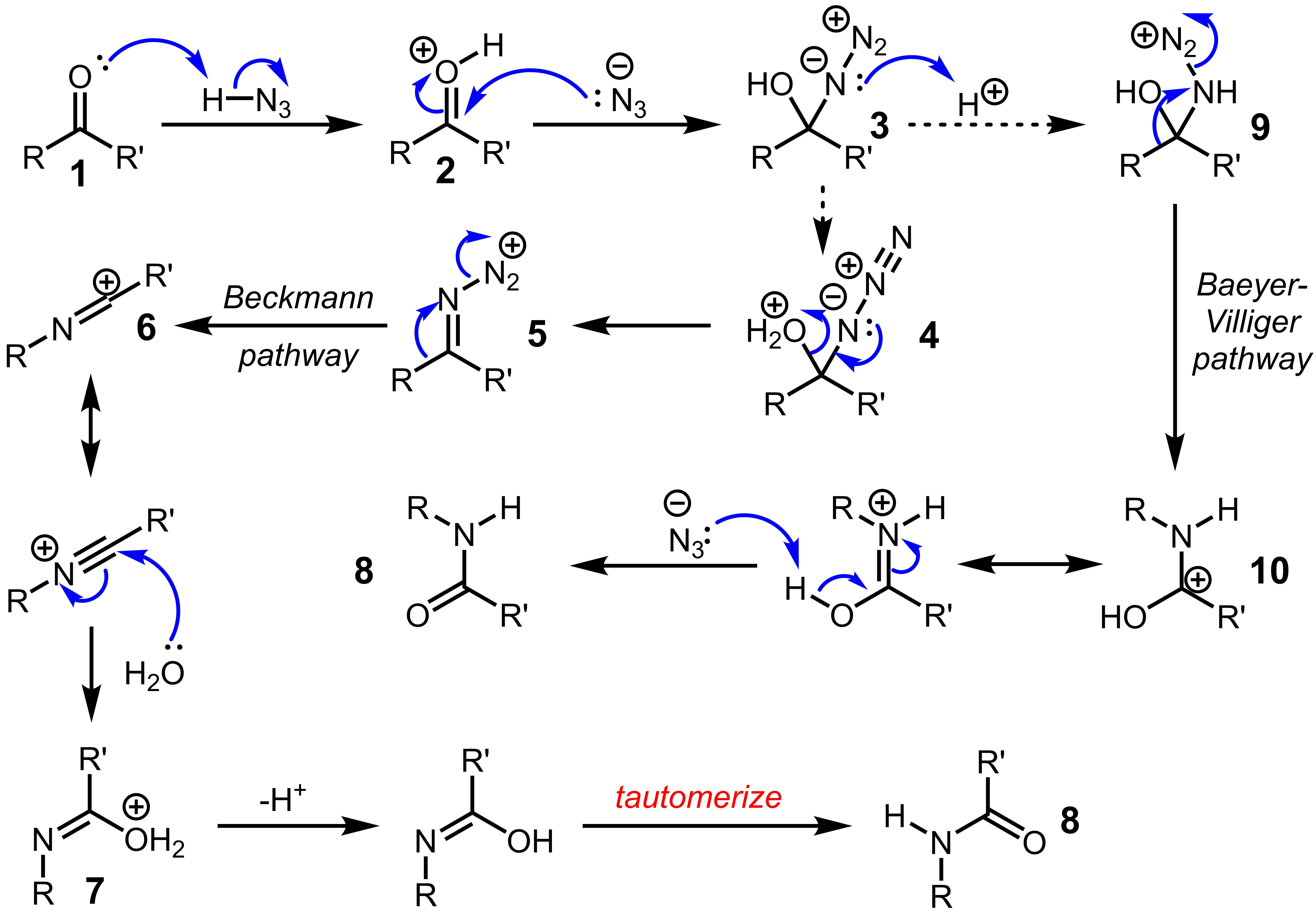
In the reaction mechanism for the Schmidt reaction of ketones
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bon ...
, the carbonyl group is activated by protonation for nucleophilic addition
In organic chemistry, a nucleophilic addition reaction is an addition reaction where a chemical compound with an electrophilic double or triple bond reacts with a nucleophile, such that the double or triple bond is broken. Nucleophilic additions di ...
by the azide, forming azidohydrin 3, which loses water in an elimination reaction to diazoiminium 5. One of the alkyl or aryl groups migrates from carbon to nitrogen with loss of nitrogen to give a nitrilium intermediate 6, as in the Beckmann rearrangement. Attack by water converts 6 to protonated imidic acid 7, which undergoes loss of proton to arrive at the imidic acid tautomer
Tautomers () are structural isomers (constitutional isomers) of chemical compounds that readily interconvert.
The chemical reaction interconverting the two is called tautomerization. This conversion commonly results from the relocation of a hydr ...
of the final amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent organic groups or hydrogen atoms. The amide group is called a peptide bond when it is ...
. In an alternative mechanism, the migration occurs at 9, directly after protonation of intermediate 3, in a manner similar to the Baeyer-Villiger reaction to give protonated amide 10. Loss of a proton again furnishes the amide. It has been proposed that the dehydration to 3 to give 5 (and, hence, the Beckmann pathway) is favored by nonaqueous acids like conc. H2SO4, while aqueous acids like conc. HCl favor migration from 9 (the Baeyer-Villiger pathway). These possibilities have been used to account for the fact that, for certain substrates like α-tetralone, the group that migrates can sometimes change, depending on the conditions used, to deliver either of the two possible amides.

Reactions involving alkyl azides
The scope of this reaction has been extended to reactions of carbonyls with alkylazides
In chemistry, azide is a linear, polyatomic anion with the formula and structure . It is the conjugate base of hydrazoic acid . Organic azides are organic compounds with the formula , containing the azide functional group. The dominant applicat ...
R-N3. This extension was first reported by J.H. Boyer in 1955 (hence the name Boyer reaction), for example, the reaction of m-nitrobenzaldehyde with β-azido-ethanol:
Variations involving intramolecular Schmidt reactions have been known since 1991. These are annulation reactions and have some utility in the synthesis of natural products; such as lactam
A lactam is a cyclic amide, formally derived from an amino alkanoic acid. The term is a portmanteau of the words ''lactone'' + ''amide''.
Nomenclature
Greek prefixes in alphabetical order indicate ring size:
* α-Lactam (3-atom rings)
* β-Lacta ...
s and alkaloids
Alkaloids are a class of basic, naturally occurring organic compounds that contain at least one nitrogen atom. This group also includes some related compounds with neutral and even weakly acidic properties. Some synthetic compounds of similar st ...
.
See also
*Curtius rearrangement
The Curtius rearrangement (or Curtius reaction or Curtius degradation), first defined by Theodor Curtius in 1885, is the thermal decomposition of an acyl azide to an isocyanate with loss of nitrogen gas. The isocyanate then undergoes attack by a va ...
* Hofmann rearrangement
*Lossen rearrangement
The Lossen rearrangement is the conversion of a hydroxamate ester to an isocyanate. Typically O-acyl, sulfonyl, or phosphoryl O-derivative are employed. The isocyanate can be used further to generate ureas in the presence of amines or generate ...
References
{{Organic reactions Rearrangement reactions Name reactions Amide synthesis reactions