|
Thiolactone
Thiolactones are a class of heterocyclic compounds in organic chemistry. They are analogs of the more common lactones in which an oxygen atom is replaced with a sulfur atom. The sulfur atom is within the ring system and adjacent to a carbonyl group. Chemistry Thiolactones can be prepared by dehydration of thiol-containing carboxylic acids. Thiolactones can be hydrolyzed back to the thiol acids under basic conditions. β-Thiolactones can be opened by reaction at the 4-position via SN2 nucleophilic reactions. Occurrence The most common thiolactone, homocysteine thiolactone is produced biochemically from homocysteine and it may play a role in protein damage. The thiolactone functional group is also present in some pharmaceutical drugs such as citiolone and erdosteine. Thiolactone rings can also be found in peptides synthesized by bacteria such as ''Staphylococcus aureus'' in order to regulate their quorum-sensing system. See also *Lactone *Lactam A lactam is a cyclic amide, form ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiolactone Types
Thiolactones are a class of heterocyclic compounds in organic chemistry. They are analogs of the more common lactones in which an oxygen atom is replaced with a sulfur atom. The sulfur atom is within the ring system and adjacent to a carbonyl group. Chemistry Thiolactones can be prepared by dehydration reaction, dehydration of thiol-containing carboxylic acids. Thiolactones can be hydrolysis, hydrolyzed back to the thiol acids under basic conditions. β-Thiolactones can be opened by reaction at the 4-position via SN2 reaction, SN2 nucleophilic reactions. Occurrence The most common thiolactone, homocysteine thiolactone is produced biochemically from homocysteine and it may play a role in protein damage. The thiolactone functional group is also present in some pharmaceutical drugs such as citiolone and erdosteine. Thiolactone rings can also be found in peptides synthesized by bacteria such as ''Staphylococcus aureus'' in order to regulate their quorum-sensing system. See also *La ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homocysteine
Homocysteine is a non-proteinogenic α-amino acid. It is a homologue of the amino acid cysteine, differing by an additional methylene bridge (-CH2-). It is biosynthesized from methionine by the removal of its terminal Cε methyl group. In the body, homocysteine can be recycled into methionine or converted into cysteine with the aid of certain B-vitamins. High levels of homocysteine in the blood (hyperhomocysteinemia) is regarded as a marker of cardiovascular disease, likely working through atherogenesis, which can result in ischemic injury. Therefore, hyperhomocysteinemia is a possible risk factor for coronary artery disease. Coronary artery disease occurs when an atherosclerotic plaque blocks blood flow to the coronary arteries, which supply the heart with oxygenated blood. Hyperhomocysteinemia has been correlated with the occurrence of blood clots, heart attacks, and strokes, although it is unclear whether hyperhomocysteinemia is an independent risk factor for these conditi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homocysteine Thiolactone
Homocysteine is a non-proteinogenic α-amino acid. It is a homologue of the amino acid cysteine, differing by an additional methylene bridge (-CH2-). It is biosynthesized from methionine by the removal of its terminal Cε methyl group. In the body, homocysteine can be recycled into methionine or converted into cysteine with the aid of certain B-vitamins. High levels of homocysteine in the blood (hyperhomocysteinemia) is regarded as a marker of cardiovascular disease, likely working through atherogenesis, which can result in ischemic injury. Therefore, hyperhomocysteinemia is a possible risk factor for coronary artery disease. Coronary artery disease occurs when an atherosclerotic plaque blocks blood flow to the coronary arteries, which supply the heart with oxygenated blood. Hyperhomocysteinemia has been correlated with the occurrence of blood clots, heart attacks, and strokes, although it is unclear whether hyperhomocysteinemia is an independent risk factor for these con ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Erdosteine
Erdosteine is a molecule with mucolytic activity. Structurally it is a thiol derivative characterized by the presence of two thiol groups. These two functional sulfhydryl groups contained in the molecule are released following first-pass metabolism with the conversion of erdosteine into its pharmacologically active metabolite Met-I. The molecule has been discovered and developed in Italy by Edmond Pharma, today it is prescribed for chronic and acute respiratory disorders in more than 40 countries worldwide. The drug is sold under several commercial names (Esteclin, Erdomed, Erdos, Erdotin etc.), as hard capsules 300 mg, dispersible tablets 300 mg, granulates for oral suspension 225 mg and powder for oral suspension 175 mg/5ml. Pharmacodynamics Erdosteine is an oral mucoactive anti-oxidant molecule, characterized by a multi-faceted pharmacological profile that may positively interfere in more than one of the pathological processes ongoing in all respiratory ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
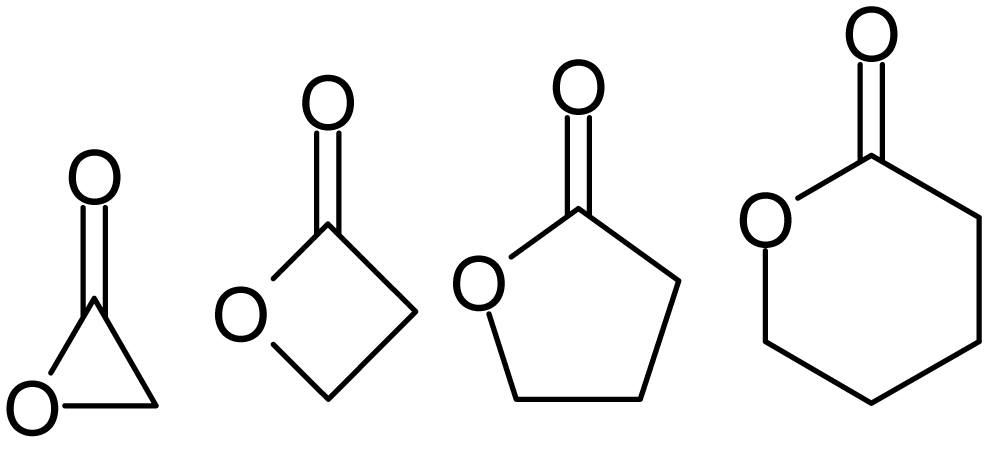
Lactone
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring. Lactones are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids, which takes place spontaneously when the ring that is formed is five- or six-membered. Lactones with three- or four-membered rings (α-lactones and β-lactones) are very reactive, making their isolation difficult. Special methods are normally required for the laboratory synthesis of small-ring lactones as well as those that contain rings larger than six-membered. Nomenclature Lactones are usually named according to the precursor acid molecule (''aceto'' = 2 carbon atoms, ''propio'' = 3, ''butyro'' = 4, ''valero'' = 5, ''capro'' = 6, etc.), with a ''-lactone'' suffix and a Greek letter prefix that specifies the number of carbon atoms in the heterocycle — that is, the distance between the relevant -OH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbonyl Group
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a carbonyl group is often referred to as a carbonyl compound. The term carbonyl can also refer to carbon monoxide as a ligand in an inorganic or organometallic complex (a metal carbonyl, e.g. nickel carbonyl). The remainder of this article concerns itself with the organic chemistry definition of carbonyl, where carbon and oxygen share a double bond. Carbonyl compounds In organic chemistry, a carbonyl group characterizes the following types of compounds: Other organic carbonyls are urea and the carbamates, the derivatives of acyl chlorides chloroformates and phosgene, carbonate esters, thioesters, lactones, lactams, hydroxamates, and isocyanates. Examples of inorganic carbonyl compounds are carbon dioxide and carbonyl sulfide. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehydration Reaction
In chemistry, a dehydration reaction is a chemical reaction that involves the loss of water from the reacting molecule or ion. Dehydration reactions are common processes, the reverse of a hydration reaction. Dehydration reactions in organic chemistry Esterification The classic example of a dehydration reaction is the Fischer esterification, which involves treating a carboxylic acid with an alcohol to give an ester :RCO2H + R′OH RCO2R′ + H2O Often such reactions require the presence of a dehydrating agent, i.e. a substance that reacts with water. Etherification Two monosaccharides, such as glucose and fructose, can be joined together (to form saccharose) using dehydration synthesis. The new molecule, consisting of two monosaccharides, is called a disaccharide. Nitrile formation Nitriles are often prepared by dehydration of primary amides. :RC(O)NH2 → RCN + H2O Ketene formation Ketene is produced by heating acetic acid and trapping the product: :CH3CO2H → CH2=C= ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the substance and w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SN2 Reaction
The SN2 reaction is a type of reaction mechanism that is common in organic chemistry. In this mechanism, one bond is broken and one bond is formed in a concerted way, i.e., in one step. The name SN2 refers to the Hughes-Ingold symbol of the mechanism: "SN" indicates that the reaction is a nucleophilic substitution, and "2" that it proceeds via a bi-molecular mechanism, which means both the reacting species are involved in the rate-determining step. The other major type of nucleophilic substitution is the SN1, but many other more specialized mechanisms describe substitution reactions. The SN2 reaction can be considered as an analogue of the associative substitution in the field of inorganic chemistry. Reaction mechanism The reaction most often occurs at an aliphatic sp3 carbon center with an electronegative, stable leaving group attached to it (often denoted X), which is frequently a halide atom. The breaking of the C–X bond and the formation of the new bond (often deno ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
The Journal Of Organic Chemistry
''The Journal of Organic Chemistry'', colloquially known as ''JOC'', is a peer-reviewed scientific journal for original contributions of fundamental research in all branches of theory and practice in organic and bioorganic chemistry. It is published by the publishing arm of the American Chemical Society, with 24 issues per year. According to the ''Journal Citation Reports'', the journal had a 2017 impact factor of 4.805 and it is the journal that received the most cites (100,091 in 2017) in the field of organic chemistry. According to Web of Knowledge (and as December 2012), eleven papers from the journal have received more than 1,000 citations, with the most cited paper having received 7,967 citations. The current editor-in-chief is Scott J. Miller from Yale University. Indexing ''J. Org. Chem.'' is currently indexed in: See also *Organic Letters *Organometallics ''Organometallics'' is a biweekly journal published by the American Chemical Society. Its area of focus is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterocyclic Compounds
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of these heterocycles. Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles. 59% of US FDA-approved drugs contain nitrogen heterocycles. Classification The study of heterocyclic chemistry focuses especially on unsaturated derivatives, and the preponderance of work and applications involves unstrained 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridine, thiophene, pyrrole, and furan are quinol ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, J.; Greeves, N. and Warren, S. (2012) ''Organic Chemistry''. Oxford University Press. pp. 1–15. . Study of structure determines their structural formula. Study of properties includes physical and chemical properties, and evaluation of chemical reactivity to understand their behavior. The study of organic reactions includes the chemical synthesis of natural products, drugs, and polymers, and study of individual organic molecules in the laboratory and via theoretical ( in silico) study. The range of chemicals studied in organic chemistry includes hydrocarbons (compounds containing only carbon and hydrogen) as well as compounds based on carbon, but also containing other elements, especially oxygen, nitrogen, sulfur, phosphorus (included in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |