|
Trisiloxane
In organosilicon chemistry, a siloxane is an organic compound containing a functional group of two silicon atoms bound to an oxygen atom: . The Parent structure, parent siloxanes include the oligomeric and polymeric hydrides with the formulae and . Siloxanes also include Branching (polymer chemistry), branched compounds, the defining feature of which is that each pair of silicon centres is separated by one oxygen atom. The siloxane functional group forms the Polymer backbone, backbone of silicones , the premier example of which is polydimethylsiloxane (PDMS). The functional group (where the three Rs may be different) is called siloxy. Siloxanes are manmade and have many commercial and industrial applications because of the compounds’ Hydrophobe, hydrophobicity, low thermal conductivity, and high flexibility. Structure Siloxanes generally adopt structures expected for linked tetrahedral ("''sp''3-like") centers. The Silicon-oxygen bond, Si−O bond length is 1.64 Å ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hexamethylcyclotrisiloxane
Hexamethylcyclotrisiloxane, also known as D3 and D3, is the organosilicon compound with the formula . It is a colorless or white volatile solid. It finds limited use in organic chemistry. The larger tetrameric and pentameric siloxanes, respectively octamethylcyclotetrasiloxane and decamethylcyclopentasiloxane, are of significant industrial interest, whereas 1,000–10,000 tonnes per year of the trimer is manufactured and/or imported in the European Economic Area. Structure and reactions Hexamethylcyclotrisiloxane adopts a planar structure and is considered strained. It reacts with organolithium reagents to give, after hydrolysis, dimethylsilanols: : : Safety and environmental considerations The LD50 for the related pentamer (D5) is >50 g/kg in rats. See also * Octamethylcyclotetrasiloxane (D4) * Decamethylcyclopentasiloxane Decamethylcyclopentasiloxane, also known as D5 and D5, is an organosilicon compound with the formula . It is a colorless and odorless liquid that is sli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
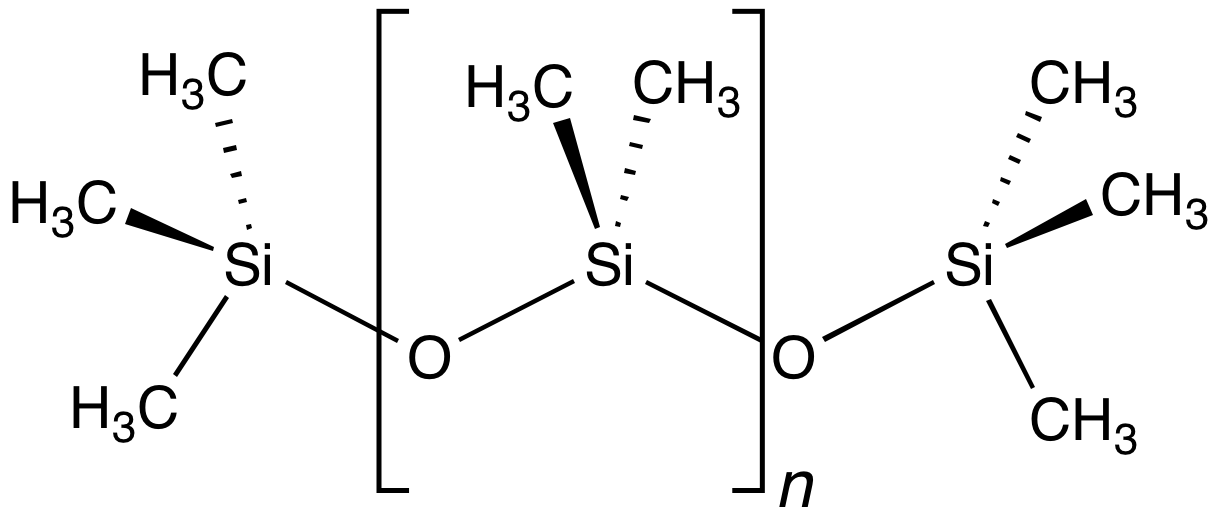
Polydimethylsiloxane
Polydimethylsiloxane (PDMS), also known as dimethylpolysiloxane or dimethicone, is a silicone polymer with a wide variety of uses, from cosmetics to industrial lubrication and passive daytime radiative cooling. PDMS is particularly known for its unusual rheological (or flow) properties. It is optically clear and, in general, inert, non-toxic, and non-flammable. It is one of several types of silicone oil (polymerized siloxane). The applications of PDMS range from contact lenses and medical devices to elastomers; it is also present in shampoos (as it makes hair shiny and slippery), food ( antifoaming agent), caulk, lubricants and heat-resistant tiles. Structure The chemical formula of PDMS is , where ''n'' is the number of repeating monomer units. Industrial synthesis can begin from dimethyldichlorosilane and water by the following net reaction: : + (''n''+1) The polymerization reaction evolves hydrochloric acid. For medical and domestic applications, a process wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicon Dioxide
Silicon dioxide, also known as silica, is an oxide of silicon with the chemical formula , commonly found in nature as quartz. In many parts of the world, silica is the major constituent of sand. Silica is one of the most complex and abundant families of materials, existing as a compound of several minerals and as a synthetic product. Examples include fused quartz, fumed silica, opal, and aerogels. It is used in structural materials, microelectronics, and as components in the food and pharmaceutical industries. All forms are white or colorless, although impure samples can be colored. Silicon dioxide is a common fundamental constituent of glass. Structure In the majority of silicon dioxides, the silicon atom shows tetrahedral coordination, with four oxygen atoms surrounding a central Si atomsee 3-D Unit Cell. Thus, SiO2 forms 3-dimensional network solids in which each silicon atom is covalently bonded in a tetrahedral manner to 4 oxygen atoms. In contrast, CO2 is a li ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cubane-type Cluster
A cubane-type cluster is an arrangement of atoms in a molecular structure that forms a cube. In the simplest case, the eight vertices are symmetry equivalent and the species has Oh symmetry group, symmetry. Such structure occurs in the hydrocarbon cubane (chemical formula ), which has carbon atoms at the corners of a cube and covalent bonds forming the edges. : Other compounds in the class have different elements in the corners, and various atoms or groups bonded to the corners. Most cubanes have more complicated structures, usually with nonequivalent vertices. They may be simple covalent compounds or macromolecular or supramolecular cluster compounds. Examples Heavier adamantogen cubanes with all vertices identical are all known, and exhibit only about half as much strain energy as cubane per molecule. The inert pair effect is believed to drive stability in cubanes with heavy main group elements: the bonding orbitals are almost entirely orbital hybridization, unhybridized ''p' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethylsilicon Dichloride
Dimethyldichlorosilane is a tetrahedral organosilicon compound with the formula . At room temperature it is a colorless liquid that readily reacts with water to form both linear and cyclic Si-O chains. Dimethyldichlorosilane is made on an industrial scale as the principal precursor to dimethylsilicone and polysilane compounds. History The first organosilicon compounds were reported in 1863 by Charles Friedel and James Crafts who synthesized tetraethylsilane from diethylzinc and silicon tetrachloride.Silicon: Organosilicon Chemistry. Encyclopedia of Inorganic Chemistry Online, 2nd ed.; Wiley: New Jersey, 2005. However, major progress in organosilicon chemistry did not occur until Frederick Kipping and his students began experimenting with diorganodichlorosilanes () that were prepared by reacting silicon tetrachloride with Grignard reagents. Unfortunately, this method suffered from many experimental problems. In the 1930s, the demand for silicones increased due to the need fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silanol
A silanol is a functional group in silicon chemistry with the connectivity Si–O–H. It is related to the hydroxy functional group (C–O–H) found in all alcohols. Silanols are often invoked as intermediates in organosilicon chemistry and silicate mineralogy. If a silanol contains one or more organic residues, it is an organosilanol. Preparation From alkoxysilanes The first isolated example of a silanol was , reported in 1871 by Albert Ladenburg. He prepared the “silicol” by hydrolysis of (Et = ). From silyl halides and related compounds Silanols are generally synthesized by hydrolysis of halosilanes, alkoxysilanes, or aminosilanes. Chlorosilanes are the most common reactants: :R3Si–Cl + H2O → R3Si–OH + HCl The hydrolysis of fluorosilanes requires more forcing reagents, i.e. alkali. The alkoxysilanes ( silyl ethers) of the type are slow to hydrolyze. Compared to the silyl ethers, silyl acetates are faster to hydrolyze, with th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicon Chloride
Silicon tetrachloride or tetrachlorosilane is the inorganic compound with the formula SiCl4. It is a colorless volatile liquid that fumes in air. It is used to produce high purity silicon and silica for commercial applications. It is a part of the chlorosilane family. Preparation Silicon tetrachloride is prepared by the chlorination of various silicon compounds such as ferrosilicon, silicon carbide, or mixtures of silicon dioxide and carbon. The ferrosilicon route is most common. In the laboratory, can be prepared by treating silicon with chlorine at : : It was first prepared by Jöns Jakob Berzelius in 1823. Brine can be contaminated with silica when the production of chlorine is a byproduct of a metal refining process from metal chloride ore. In rare occurrences, the silicon dioxide in silica is converted to silicon tetrachloride when the contaminated brine is electrolyzed. Reactions Hydrolysis and related reactions Like other chlorosilanes or silanes, silicon tetrachlorid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of Biomolecule, biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dichlorodimethylsilane
Dimethyldichlorosilane is a tetrahedral organosilicon compound with the formula . At room temperature it is a colorless liquid that readily reacts with water to form both linear and cyclic Si-O chains. Dimethyldichlorosilane is made on an industrial scale as the principal precursor to dimethylsilicone and polysilane compounds. History The first organosilicon compounds were reported in 1863 by Charles Friedel and James Crafts who synthesized tetraethylsilane from diethylzinc and silicon tetrachloride.Silicon: Organosilicon Chemistry. Encyclopedia of Inorganic Chemistry Online, 2nd ed.; Wiley: New Jersey, 2005. However, major progress in organosilicon chemistry did not occur until Frederick Kipping and his students began experimenting with diorganodichlorosilanes () that were prepared by reacting silicon tetrachloride with Grignard reagents. Unfortunately, this method suffered from many experimental problems. In the 1930s, the demand for silicones increased due to the need fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glass Transition Temperature
The glass–liquid transition, or glass transition, is the gradual and reversible transition in amorphous materials (or in amorphous regions within semicrystalline materials) from a hard and relatively brittle "glassy" state into a viscous or rubbery state as the temperature is increased. ISO 11357-2: Plastics – Differential scanning calorimetry – Part 2: Determination of glass transition temperature (1999). An amorphous solid that exhibits a glass transition is called a glass. The reverse transition, achieved by supercooling a viscous liquid into the glass state, is called vitrification. The glass-transition temperature ''T''g of a material characterizes the range of temperatures over which this glass transition occurs (as an experimental definition, typically marked as 100 s of relaxation time). It is always lower than the melting temperature, ''T''m, of the crystalline state of the material, if one exists, because the glass is a higher energy state (or enthalpy at const ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |