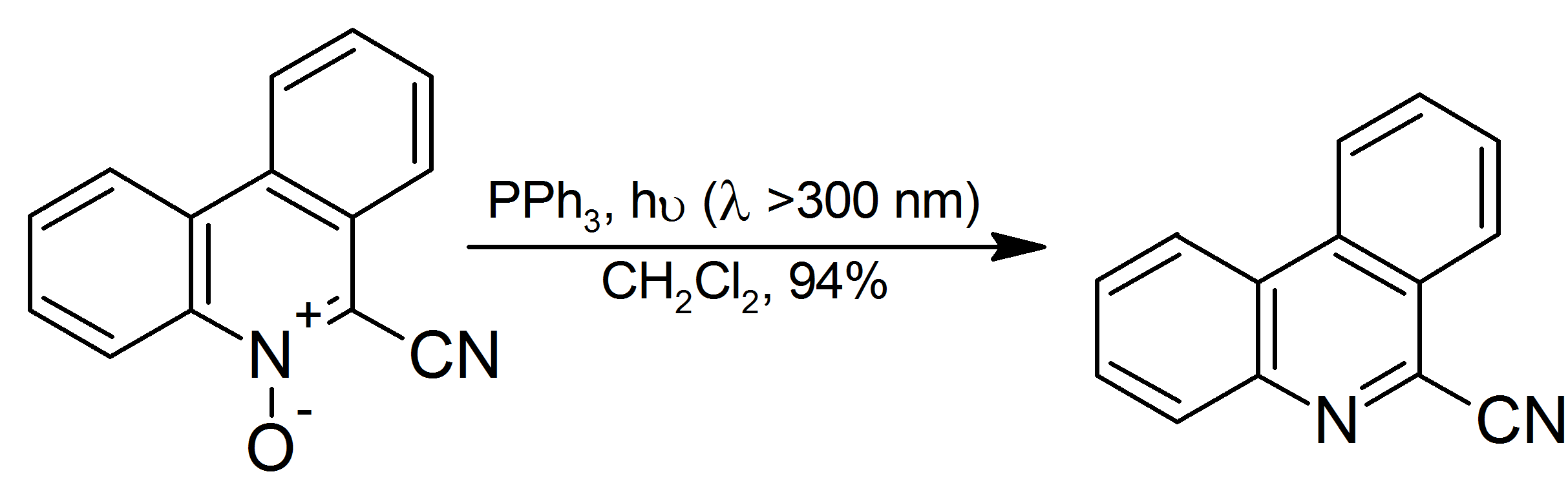
|
Trioctylphosphine
Trioctylphosphine is an organophosphorus compound with the formula P(C8H17)3 sometimes abbreviated TOP. It is usually encountered as a syrup. The compound is colorless. Reactions Trioctylphosphine reacts with oxygen to form trioctylphosphine oxide. For this reason it is usually handled with air-free techniques. TOP reacts with elemental selenium to give trioctylphosphine selenide (TOPSe), which is a reagent for the preparation of cadmium selenide and related semiconductors. See also * Triphenylphosphine * Trioctylphosphine oxide Trioctylphosphine oxide (TOPO) is an organophosphorus compound with the formula OP(C8H17)3. Frequently referred to as TOPO, this compound is used as an extraction or stabilizing agent. It is an air-stable white solid at room temperature. Prepar ... References {{organic-compound-stub Phosphines ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trioctylphosphine Oxide
Trioctylphosphine oxide (TOPO) is an organophosphorus compound with the formula OP(C8H17)3. Frequently referred to as TOPO, this compound is used as an extraction or stabilizing agent. It is an air-stable white solid at room temperature. Preparation and use TOPO is usually prepared by oxidation of trioctylphosphine, which in turn is produced by alkylation of phosphorus trichloride. The main use of TOPO is in solvent extraction of metals, especially uranium. The high lipophilicity and high polarity are properties key to this application. Its high polarity, which results from the dipolar phosphorus-oxygen bond, allows this compound to bind to metal ions. The octyl groups confer solubility in low polarity solvents such as kerosene. In the research laboratory, both trioctylphosphine and TOPO are frequently useful as a capping ligand for the production of quantum dots such as those consisting of CdSe. In these cases, TOPO serves as solvent for the synthesis and solubilizes the gro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trioctylphosphine Selenide
Trioctylphosphine selenide (TOPSe) is an organophosphorus compound with the formula SeP(C8H17)3. It is used as a source of selenium in the preparation of cadmium selenide. TOPSe is a white, air-stable solid that is soluble in organic solvents. The molecule features a tetrahedral phosphorus center. Preparation and use TOPSe is usually prepared by oxidation of trioctylphosphine with elemental selenium: : Often the reaction is conducted without isolation of the TOPSe. As a solution with trioctylphosphine oxide, TOPSe reacts with dimethylcadmium to give cadmium selenide. The mechanism is proposed to proceed in two steps, beginning with the formation of cadmium metal followed by its oxidation with the TOPSe. Similarly it has been used to produce lead selenide Lead selenide (PbSe), or lead(II) selenide, a selenide of lead, is a semiconductor material. It forms cubic crystals of the NaCl structure; it has a direct bandgap of 0.27 eV at room temperature. (Note that incorrec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trioctylphosphine Oxide
Trioctylphosphine oxide (TOPO) is an organophosphorus compound with the formula OP(C8H17)3. Frequently referred to as TOPO, this compound is used as an extraction or stabilizing agent. It is an air-stable white solid at room temperature. Preparation and use TOPO is usually prepared by oxidation of trioctylphosphine, which in turn is produced by alkylation of phosphorus trichloride. The main use of TOPO is in solvent extraction of metals, especially uranium. The high lipophilicity and high polarity are properties key to this application. Its high polarity, which results from the dipolar phosphorus-oxygen bond, allows this compound to bind to metal ions. The octyl groups confer solubility in low polarity solvents such as kerosene. In the research laboratory, both trioctylphosphine and TOPO are frequently useful as a capping ligand for the production of quantum dots such as those consisting of CdSe. In these cases, TOPO serves as solvent for the synthesis and solubilizes the gro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organophosphorus Compound
Organophosphorus compounds are organic compounds containing phosphorus. They are used primarily in pest control as an alternative to chlorinated hydrocarbons that persist in the environment. Some organophosphorus compounds are highly effective insecticides, although some are extremely toxic to humans, including sarin and VX nerve agents. Organophosphorus chemistry is the corresponding science of the properties and reactivity of organophosphorus compounds. Phosphorus, like nitrogen, is in group 15 of the periodic table, and thus phosphorus compounds and nitrogen compounds have many similar properties. The definition of organophosphorus compounds is variable, which can lead to confusion. In industrial and environmental chemistry, an organophosphorus compound need contain only an organic substituent, but need not have a direct phosphorus-carbon (P-C) bond. Thus a large proportion of pesticides (e.g., malathion), are often included in this class of compounds. Phosphorus can adopt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Formula
In chemistry, a chemical formula is a way of presenting information about the chemical proportions of atoms that constitute a particular chemical compound or molecule, using chemical element symbols, numbers, and sometimes also other symbols, such as parentheses, dashes, brackets, commas and ''plus'' (+) and ''minus'' (−) signs. These are limited to a single typographic line of symbols, which may include subscripts and superscripts. A chemical formula is not a chemical name, and it contains no words. Although a chemical formula may imply certain simple chemical structures, it is not the same as a full chemical structural formula. Chemical formulae can fully specify the structure of only the simplest of molecules and chemical substances, and are generally more limited in power than chemical names and structural formulae. The simplest types of chemical formulae are called '' empirical formulae'', which use letters and numbers indicating the numerical ''proportions'' of ato ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Air-free Technique
Air-free techniques refer to a range of manipulations in the chemistry laboratory for the handling of compounds that are air-sensitive. These techniques prevent the compounds from reacting with components of air, usually water and oxygen; less commonly carbon dioxide and nitrogen. A common theme among these techniques is the use of a fine (100–10−3 Torr) or high (10−3–10−6 Torr) vacuum to remove air, and the use of an inert gas: preferably argon, but often nitrogen. The two most common types of air-free technique involve the use of a glovebox and a Schlenk line, although some rigorous applications use a high-vacuum line. In both methods, glassware (often Schlenk tubes) are pre-dried in ovens prior to use. They may be flame-dried to remove adsorbed water. Prior to coming into an inert atmosphere, vessels are further dried by ''purge-and-refill'' — the vessel is subjected to a vacuum to remove gases and water, and then refilled with inert gas. This cycle is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cadmium Selenide
Cadmium selenide is an inorganic compound with the formula Cd Se. It is a black to red-black solid that is classified as a II-VI semiconductor of the n-type. Much of the current research on this compound is focused on its nanoparticles. Structure Three crystalline forms of CdSe are known which follow the structures of: wurtzite (hexagonal), sphalerite (cubic) and rock-salt (cubic). The sphalerite CdSe structure is unstable and converts to the wurtzite form upon moderate heating. The transition starts at about 130 °C, and at 700 °C it completes within a day. The rock-salt structure is only observed under high pressure. Production The production of cadmium selenide has been carried out in two different ways. The preparation of bulk crystalline CdSe is done by the High-Pressure Vertical Bridgman method or High-Pressure Vertical Zone Melting. Cadmium selenide may also be produced in the form of nanoparticles. (see applications for explanation) Several methods for t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triphenylphosphine
Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is widely used in the synthesis of organic and organometallic compounds. PPh3 exists as relatively air stable, colorless crystals at room temperature. It dissolves in non-polar organic solvents such as benzene and diethyl ether. Preparation and structure Triphenylphosphine can be prepared in the laboratory by treatment of phosphorus trichloride with phenylmagnesium bromide or phenyllithium. The industrial synthesis involves the reaction between phosphorus trichloride, chlorobenzene, and sodium: :PCl3 + 3 PhCl + 6 Na → PPh3 + 6 NaCl Triphenylphosphine crystallizes in triclinic and monoclinic modification. In both cases, the molecule adopts a pyramidal structure with propeller-like arrangement of the three phenyl groups. Principal reactions with chalcogens, halogens, and acids Oxidation Triphenylphosphine undergoes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |