|
Trifluoride
Trifluorides are compounds in which one atom or ion has three fluorine atoms or ions associated. Many metals form trifluorides, such as iron, the rare-earth elements, and the metals in the aluminium and scandium columns of the periodic table. Most metal trifluorides are poorly soluble in water except Ferric fluoride and Indium(III) fluoride, but several are soluble in other solvents. List of trifluorides * Actinium trifluoride, AcF3 *Aluminium trifluoride * Americium trifluoride, AmF3 *Antimony trifluoride, SbF3, sometimes called Swart's reagent * Arsenic trifluoride, AsF3 *Bismuth trifluoride, BiF3 * Boron trifluoride, BF3, a pungent colourless toxic gas * Bromotrifluoromethane, (carbon monobromide trifluoride) *Bromine trifluoride, BrF3 * Carbon trifluoride, C2F6, Hexafluoroethane * Cerium trifluoride, CeF3 *Chlorine trifluoride, ClF3 * Chromium trifluoride, CrF3 *Cobalt trifluoride, CoF3 * Curium trifluoride, CmF3 * Diethylaminosulfur trifluoride (DAST) is the organosulfur comp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
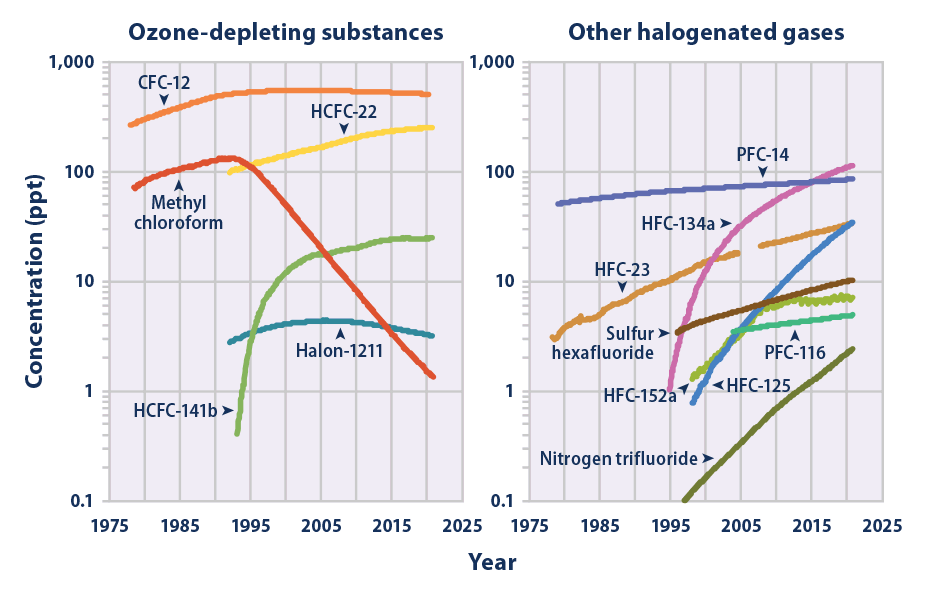
Nitrogen Trifluoride
Nitrogen trifluoride () is an inorganic, colorless, non-flammable, toxic gas with a slightly musty odor. It finds increasing use within the manufacturing of flat-panel displays, photovoltaics, LEDs and other microelectronics. Nitrogen trifluoride is also an extremely strong and long-lived greenhouse gas. Its atmospheric burden exceeded 2 parts per trillion during 2019 and has doubled every five years since the late 20th century. Synthesis and reactivity Nitrogen trifluoride did not exist in significant quantities on Earth prior to its synthesis by humans. It is a rare example of a binary fluoride that can be prepared directly from the elements only at very uncommon conditions, such as an electric discharge. After first attempting the synthesis in 1903, Otto Ruff prepared nitrogen trifluoride by the electrolysis of a molten mixture of ammonium fluoride and hydrogen fluoride. It proved to be far less reactive than the other nitrogen trihalides nitrogen trichloride, nitrogen trib ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron Trifluoride
Boron trifluoride is the inorganic compound with the formula BF3. This pungent, colourless, and toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds. Structure and bonding The geometry of a molecule of BF3 is trigonal planar. Its D3h symmetry conforms with the prediction of VSEPR theory. The molecule has no dipole moment by virtue of its high symmetry. The molecule is isoelectronic with the carbonate anion, . BF3 is commonly referred to as " electron deficient," a description that is reinforced by its exothermic reactivity toward Lewis bases. In the boron trihalides, BX3, the length of the B–X bonds (1.30 Å) is shorter than would be expected for single bonds, and this shortness may indicate stronger B–X π-bonding in the fluoride. A facile explanation invokes the symmetry-allowed overlap of a p orbital on the boron atom with the in-phase combination of the three similarly oriented p orbitals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phosphorus Trifluoride
Phosphorus trifluoride (formula P F3), is a colorless and odorless gas. It is highly toxic and reacts slowly with water. Its main use is as a ligand in metal complexes. As a ligand, it parallels carbon monoxide in metal carbonyls, and indeed its toxicity is due to its binding with the iron in blood hemoglobin in a similar way to carbon monoxide. Physical properties Phosphorus trifluoride has an F−P−F bond angle of approximately 96.3°. Gaseous PF3 has a standard enthalpy of formation of −945 kJ/mol (−226 kcal/ mol). The phosphorus atom has a nuclear magnetic resonance chemical shift of 97 ppm (downfield of H3PO4). Properties Phosphorus trifluoride hydrolyzes especially at high pH, but it is less hydrolytically sensitive than phosphorus trichloride. It does not attack glass except at high temperatures, and anhydrous potassium hydroxide may be used to dry it with little loss. With hot metals, phosphides and fluorides are formed. With Lewis bases such a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine Trifluoride
Chlorine trifluoride is an interhalogen compound with the formula ClF3. This colorless, poisonous, corrosive, and extremely reactive gas condenses to a pale-greenish yellow liquid, the form in which it is most often sold (pressurized at room temperature). The compound is primarily of interest in plasmaless cleaning and etching operations in the semiconductor industry, in nuclear reactor fuel processing, as a component in rocket fuels, and other industrial operations. Preparation, structure, and properties It was first reported in 1930 by Ruff and Krug who prepared it by fluorination of chlorine; this also produced ClF (chlorine monofluoride) and the mixture was separated by distillation. :3 F2 + Cl2 → 2 ClF3 The molecular geometry of ClF3 is approximately T-shaped, with one short bond (1.598 Å) and two long bonds (1.698 Å). This structure agrees with the prediction of VSEPR theory, which predicts lone pairs of electrons as occupying two equatorial positions o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
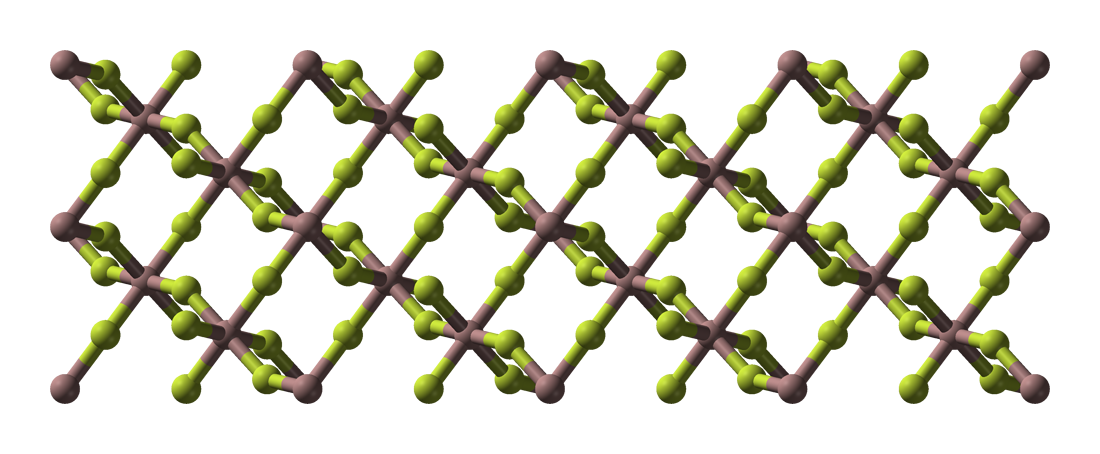
Bismuth Trifluoride
Bismuth(III) fluoride or bismuth trifluoride is a chemical compound of bismuth and fluorine. The chemical formula is BiF3. It is a grey-white powder melting at 649 °C. It occurs in nature as the rare mineral gananite. Synthesis Bismuth fluoride can be prepared by reacting bismuth(III) oxide with hydrofluoric acid: :Bi2O3 + 6 HF → 2 BiF3 + 3 H2O Structure α-BiF3 has a cubic crystalline structure (Pearson symbol cF16, space group Fm-3m, No. 225). BiF3 is the prototype for the D03 structure, which is adopted by several intermetallics, including Mg3Pr, Cu3Sb, Fe3Si, and AlFe3, as well as by the hydride LaH3.0. The unit cell is face-centered cubic with Bi at the face centers and vertices, and F at the octahedral site (mid-edges, center), and tetrahedral sites (centers of the 8 sub cubes) - thus the primitive cell contains 4 Bi and 12 F. Alternatively, with the unit cell shifted (1/4,1/4,1/4) the description can be of a fcc cell with face, edge, corner, and centers filled ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromine Trifluoride
Bromine trifluoride is an interhalogen compound with the formula BrF3. At room temperature, it is a straw-coloured liquid with a pungent odor which decomposes violently on contact with water and organic compounds. It is a powerful fluorinating agent and an ionizing inorganic solvent. It is used to produce uranium hexafluoride (UF6) in the processing and reprocessing of nuclear fuel. Synthesis Bromine trifluoride was first described by Paul Lebeau in 1906, who obtained the material by the reaction of bromine with fluorine at 20 °C: : The disproportionation of bromine monofluoride also gives bromine trifluoride: : Structure Like ClF3 and IF3, the BrF3 molecule is T-shaped and planar. In the VSEPR formalism, the bromine center is assigned two electron pairs. The distance from the bromine each axial fluorine is 1.81 Å and to the equatorial fluorine is 1.72 Å. The angle between an axial fluorine and the equatorial fluorine is slightly smaller than 90° — the 86.2° ang ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodine Trifluoride
Iodine trifluoride is an interhalogen compound with the chemical formula IF3. It is a yellow solid which decomposes above −28 °C. It can be synthesised from the elements, but care must be taken to avoid the formation of IF5. Reactions F2 reacts with I2 to yield IF3 at −45 °C in CCl3F. Alternatively, at low temperatures, the fluorination reaction I2 + 3 XeF2 → 2IF3 + 3 Xe can be used. Not much is known about iodine trifluoride as it is so unstable. Structure The iodine atom of iodine trifluoride has five electron pairs, of which two are lone-pairs, and the molecule is T-shaped as predicted by VSEPR Theory Valence shell electron pair repulsion (VSEPR) theory ( , ), is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. It is also named the Gillespie-Nyholm the .... References {{Fluorine compounds Fluorides Interhalogen compounds Iodine compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gold Trifluoride
Gold(III) fluoride, , is an orange solid that sublimes at 300 °C. It is a powerful fluorinating agent. Preparation AuF3 can be prepared by reacting AuCl3 with F2 or BrF3. Structure The crystal structure of AuF3 consists of spirals of square-planar The square planar molecular geometry in chemistry describes the stereochemistry (spatial arrangement of atoms) that is adopted by certain chemical compounds. As the name suggests, molecules of this geometry have their atoms positioned at the corn ... AuF4 units. References External links * Fluorides Metal halides Gold(III) compounds Fluorinating agents Gold–halogen compounds {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cobalt Trifluoride
Cobalt(III) fluoride is the inorganic compound with the formula . Hydrates are also known. The anhydrous compound is a hygroscopic brown solid. It is used to synthesize organofluorine compounds. The related cobalt(III) chloride is also known but is extremely unstable.Arthur W. Chester, El-Ahmadi Heiba, Ralph M. Dessau, and William J. Koehl Jr. (1969): "The interaction of cobalt(III) with chloride ion in acetic acid". ''Inorganic and Nuclear Chemistry Letters'', volume 5, issue 4, pages 277-283. Cobalt(III) bromide and cobalt(III) iodide have not been synthesized. Structure Anhydrous Anhydrous cobalt trifluoride crystallizes in the rhombohedral group, specifically according to the aluminium trifluoride motif, with ''a'' = 527.9 pm, ''α'' = 56.97°. Each cobalt atom is bound to six fluorine atoms in octahedral geometry, with Co–F distances of 189 pm. Each fluoride is a doubly bridging ligand. Hydrates A hydrate is known. It is conjectured to be better described as . There ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antimony Trifluoride
Antimony trifluoride is the inorganic compound with the formula SbF3. Sometimes called Swarts' reagent, is one of two principal fluorides of antimony, the other being SbF5. It appears as a white solid. As well as some industrial applications, it is used as a reagent in inorganic and organofluorine chemistry. Preparation and structure In solid SbF3, the Sb centres have octahedral molecular geometry and are linked by bridging fluoride ligands. Three Sb–F bonds are short (192 pm) and three are long (261 pm). Because it is a polymer, SbF3 is far less volatile than related compounds AsF3 and SbCl3. SbF3 is prepared by treating antimony trioxide with hydrogen fluoride: :Sb2O3 + 6 HF → 2 SbF3 + 3 H2O The compound is a mild Lewis acid, hydrolyzing slowly in water. With fluorine, it is oxidized to give antimony pentafluoride. :SbF3 + F2 → SbF5 Applications It is used as a fluorination reagent in organic chemistry. This application was reported by the B ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gallium Trifluoride
Gallium(III) fluoride ( Ga F3) is a chemical compound. It is a white solid that melts under pressure above 1000 °C but sublimes around 950 °C. It has the FeF3 structure where the gallium atoms are 6-coordinate. GaF3 can be prepared by reacting F2 or HF with Ga2O3 or by thermal decomposition of (NH4)3GaF6.Anthony John Downs, (1993), ''Chemistry of Aluminium, Gallium, Indium, and Thallium'', Springer, GaF3 is virtually insoluble in water. Solutions of GaF3 in HF can be evaporated to form the trihydrate, GaF3·3H2O, which on heating gives a hydrated form of GaF2(OH). Gallium(III) fluoride reacts with mineral acids to form hydrofluoric acid Hydrofluoric acid is a Solution (chemistry), solution of hydrogen fluoride (HF) in water. Solutions of HF are colourless, acidic and highly Corrosive substance, corrosive. It is used to make most fluorine-containing compounds; examples include th .... References Further reading * Fluorides Gallium compounds Metal halides [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arsenic Trifluoride
Arsenic trifluoride is a chemical compound of arsenic and fluorine with the chemical formula AsF3. It is a colorless liquid which reacts readily with water. Preparation and properties It can be prepared by reacting hydrogen fluoride, HF, with arsenic trioxide Arsenic trioxide, sold under the brand name Trisenox among others, is an inorganic compound and medication. As an industrial chemical, whose major uses include in the manufacture of wood preservatives, pesticides, and glass. As a medication, it ...: :6HF + As2O3 → 2AsF3 + 3H2O It has a pyramidal molecular structure in the gas phase which is also present in the solid. In the gas phase the As-F bond length is 170.6 pm and the F-As-F bond angle 96.2°. Arsenic trifluoride is used as a fluorinating agent for the conversion of non-metal chlorides to fluorides, in this respect it is less reactive than SbF3. Salts containing AsF4− anion can be prepared for example CsAsF4. the potassium salt KAs2F7 prepared from KF an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

platinum(0)-from-xtal-2008-3D-balls.png)