|
Triangulene
Triangulene (also known as Clar's hydrocarbon) is the smallest triplet-ground-state polybenzenoid. It exists as a biradical with the chemical formula . It was first hypothesized by Czech chemist Erich Clar in 1953. Its first confirmed synthesis was published in a February 2017 issue of '' Nature Nanotechnology'', in a project led by researchers David Fox and Anish Mistry at the University of Warwick in collaboration with IBM. Other attempts by Japanese researchers have been successful only in making substituted triangulene derivatives. A six-step synthesis yielded two isomers of dihydrotriangulene which were then deposited on xenon or copper base. The researchers used a combined scanning tunneling and atomic force microscope (STM/AFM) to remove individual hydrogen atoms. The synthesized molecule of triangulene remained stable at high-vacuum low-temperature conditions for four days, giving the scientists plenty of time to characterize it (also using STM/AFM). 'n''riangule ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Erich Clar
Erich Clar (23 August 1902 in Hřensko – 27 March 1987 in Estepona) was an Austrian organic chemist who studied polycyclic aromatic hydrocarbon chemistry. He is considered as the father of that field. In 1941, he authored "Aromatische Kohlenwasserstoffe" (Springer-Verlag) and in 1964 the greatly expanded two-volume ''Polycyclic Hydrocarbons'', which described the syntheses, properties, and UV-visible absorption spectra of hundreds of PAHs. He discovered the Clar reaction of the cyclic ketone perinaphthenone to form dibenzo d,lmerylene in a 400 C melt of zinc dust, zinc (II) chloride, and sodium chloride. He created the Sextet Theory, now eponymously called ''Clar's rule'', to describe the behavior of polycyclic aromatic hydrocarbon isomers. This was described in his book ''The Aromatic Sextet''. He was awarded the August Kekulé Medal by the Chemical Society of the GDR in 1965, the highest award given by that society to foreign scientists, and the first Polycyclic Aromatic Hydroca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triplet State
In quantum mechanics, a triplet is a quantum state of a system with a spin of quantum number =1, such that there are three allowed values of the spin component, = −1, 0, and +1. Spin, in the context of quantum mechanics, is not a mechanical rotation but a more abstract concept that characterizes a particle's intrinsic angular momentum. It is particularly important for systems at atomic length scales, such as individual atoms, protons, or electrons. Almost all molecules encountered in daily life exist in a singlet state, but molecular oxygen is an exception. At room temperature, O2 exists in a triplet state, which can only undergo a chemical reaction by making the forbidden transition into a singlet state. This makes it kinetically nonreactive despite being thermodynamically one of the strongest oxidants. Photochemical or thermal activation can bring it into the singlet state, which makes it kinetically as well as thermodynamically a very strong oxidant. __TOC__ Two spin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is the chemical element with the symbol H and atomic number 1. Hydrogen is the lightest element. At standard conditions hydrogen is a gas of diatomic molecules having the formula . It is colorless, odorless, tasteless, non-toxic, and highly combustible. Hydrogen is the most abundant chemical substance in the universe, constituting roughly 75% of all normal matter.However, most of the universe's mass is not in the form of baryons or chemical elements. See dark matter and dark energy. Stars such as the Sun are mainly composed of hydrogen in the plasma state. Most of the hydrogen on Earth exists in molecular forms such as water and organic compounds. For the most common isotope of hydrogen (symbol 1H) each atom has one proton, one electron, and no neutrons. In the early universe, the formation of protons, the nuclei of hydrogen, occurred during the first second after the Big Bang. The emergence of neutral hydrogen atoms throughout the universe occurre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inelastic Electron Tunneling Spectroscopy
Inelastic electron tunneling spectroscopy (IETS) is an experimental tool for studying the vibrations of molecular adsorbates on metal oxides. It yields vibrational spectra of the adsorbates with high resolution (< 0.5 meV) and high sensitivity (< 1013 molecules are required to provide a spectrum). An additional advantage is the fact that optically forbidden transitions may be observed as well. Within IETS, an oxide layer with molecules adsorbed on it is put between two metal plates. A is applied between the two contacts. An energy diagram of the metal-oxide-metal device under bias is shown in the top figure. The metal contacts are characterized by a constant , filled up t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
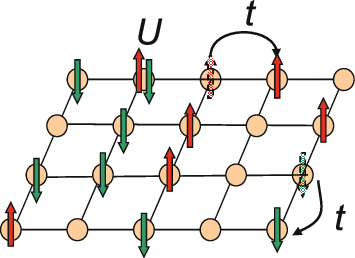
Hubbard Model
The Hubbard model is an approximate model used to describe the transition between conducting and insulating systems. It is particularly useful in solid-state physics. The model is named for John Hubbard. The Hubbard model states that each electron experiences competing forces: one pushes it to tunnel to neighboring atoms, while the other pushes it away from its neighbors. Its Hamiltonian thus has two terms: a kinetic term allowing for tunneling ("hopping") of particles between lattice sites and a potential term reflecting on-site interaction. The particles can either be fermions, as in Hubbard's original work, or bosons, in which case the model is referred to as the " Bose–Hubbard model". The Hubbard model is a useful approximation for particles in a periodic potential at sufficiently low temperatures, where all the particles may be assumed to be in the lowest Bloch band, and long-range interactions between the particles can be ignored. If interactions between partic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polycyclic Aromatic Hydrocarbon
A polycyclic aromatic hydrocarbon (PAH) is a class of organic compounds that is composed of multiple aromatic rings. The simplest representative is naphthalene, having two aromatic rings and the three-ring compounds anthracene and phenanthrene. PAHs are uncharged, non-polar and planar. Many are colorless. Many of them are found in coal and in oil deposits, and are also produced by the combustion of organic matter—for example, in engines and incinerators or when biomass burns in forest fires. Polycyclic aromatic hydrocarbons are discussed as possible starting materials for abiotic syntheses of materials required by the earliest forms of life. Nomenclature and structure The terms polyaromatic hydrocarbon or polynuclear aromatic hydrocarbon are also used for this concept. By definition, polycyclic aromatic hydrocarbons have multiple rings, precluding benzene from being considered a PAH. Some sources, such as the US EPA and CDC, consider naphthalene to be the simplest PA ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
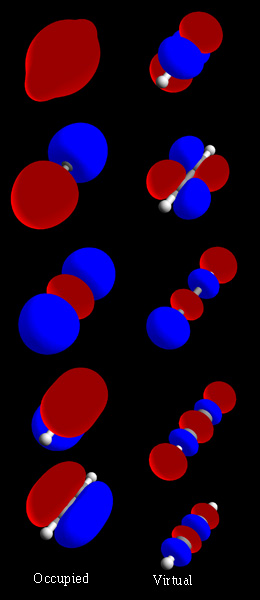
Molecular Orbitals
In chemistry, a molecular orbital is a mathematical function describing the location and wave-like behavior of an electron in a molecule. This function can be used to calculate chemical and physical properties such as the probability of finding an electron in any specific region. The terms ''atomic orbital'' and ''molecular orbital'' were introduced by Robert S. Mulliken in 1932 to mean ''one-electron orbital wave functions''. At an elementary level, they are used to describe the ''region'' of space in which a function has a significant amplitude. In an isolated atom, the orbital electrons' location is determined by functions called atomic orbitals. When multiple atoms combine chemically into a molecule, the electrons' locations are determined by the molecule as a whole, so the atomic orbitals combine to form molecular orbitals. The electrons from the constituent atoms occupy the molecular orbitals. Mathematically, molecular orbitals are an approximate solution to the Sch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tight Binding
In solid-state physics, the tight-binding model (or TB model) is an approach to the calculation of electronic band structure using an approximate set of wave functions based upon superposition of wave functions for isolated atoms located at each atomic site. The method is closely related to the LCAO method (linear combination of atomic orbitals method) used in chemistry. Tight-binding models are applied to a wide variety of solids. The model gives good qualitative results in many cases and can be combined with other models that give better results where the tight-binding model fails. Though the tight-binding model is a one-electron model, the model also provides a basis for more advanced calculations like the calculation of surface states and application to various kinds of many-body problem and quasiparticle calculations. Introduction The name "tight binding" of this electronic band structure model suggests that this quantum mechanical model describes the properties of tight ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cryogenics
In physics, cryogenics is the production and behaviour of materials at very low temperatures. The 13th IIR International Congress of Refrigeration (held in Washington DC in 1971) endorsed a universal definition of “cryogenics” and “cryogenic” by accepting a threshold of 120 K (or –153 °C) to distinguish these terms from the conventional refrigeration. This is a logical dividing line, since the normal boiling points of the so-called permanent gases (such as helium, hydrogen, neon, nitrogen, oxygen, and normal air) lie below 120K while the Freon refrigerants, hydrocarbons, and other common refrigerants have boiling points above 120K. The U.S. National Institute of Standards and Technology considers the field of cryogenics as that involving temperatures below -153 Celsius (120K; -243.4 Fahrenheit) Discovery of superconducting materials with critical temperatures significantly above the boiling point of nitrogen has provided new interest in reliable, low cost me ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vacuum
A vacuum is a space devoid of matter. The word is derived from the Latin adjective ''vacuus'' for "vacant" or " void". An approximation to such vacuum is a region with a gaseous pressure much less than atmospheric pressure. Physicists often discuss ideal test results that would occur in a ''perfect'' vacuum, which they sometimes simply call "vacuum" or free space, and use the term partial vacuum to refer to an actual imperfect vacuum as one might have in a laboratory or in space. In engineering and applied physics on the other hand, vacuum refers to any space in which the pressure is considerably lower than atmospheric pressure. The Latin term ''in vacuo'' is used to describe an object that is surrounded by a vacuum. The ''quality'' of a partial vacuum refers to how closely it approaches a perfect vacuum. Other things equal, lower gas pressure means higher-quality vacuum. For example, a typical vacuum cleaner produces enough suction to reduce air pressure by around 20%. But ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atomic-force Microscopy
Atomic force microscopy (AFM) or scanning force microscopy (SFM) is a very-high-resolution type of scanning probe microscopy (SPM), with demonstrated resolution on the order of fractions of a nanometer, more than 1000 times better than the optical diffraction limit. Overview Atomic force microscopy (AFM) is a type of scanning probe microscopy (SPM), with demonstrated resolution on the order of fractions of a nanometer, more than 1000 times better than the optical diffraction limit. The information is gathered by "feeling" or "touching" the surface with a mechanical probe. Piezoelectric elements that facilitate tiny but accurate and precise movements on (electronic) command enable precise scanning. Despite the name, the Atomic Force Microscope does not use the Nuclear force. Abilities The AFM has three major abilities: force measurement, topographic imaging, and manipulation. In force measurement, AFMs can be used to measure the forces between the probe and the sample as a f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |