|
Thermonuclear Detonation
Thermonuclear fusion is the process of atomic nuclei combining or “fusing” using high temperatures to drive them close enough together for this to become possible. There are two forms of thermonuclear fusion: ''uncontrolled'', in which the resulting energy is released in an uncontrolled manner, as it is in thermonuclear weapons ("hydrogen bombs") and in most stars; and ''controlled'', where the fusion reactions take place in an environment allowing some or all of the energy released to be harnessed for constructive purposes. Temperature requirements Temperature is a measure of the average kinetic energy of particles, so by heating the material it will gain energy. After reaching sufficient temperature, given by the Lawson criterion, the energy of accidental collisions within the plasma is high enough to overcome the Coulomb barrier and the particles may fuse together. In a deuterium–tritium fusion reaction, for example, the energy necessary to overcome the Coulomb bar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermonuclear Weapon
A thermonuclear weapon, fusion weapon or hydrogen bomb (H bomb) is a second-generation nuclear weapon design. Its greater sophistication affords it vastly greater destructive power than first-generation nuclear bombs, a more compact size, a lower mass, or a combination of these benefits. Characteristics of nuclear fusion reactions make possible the use of non-fissile depleted uranium as the weapon's main fuel, thus allowing more efficient use of scarce fissile material such as uranium-235 () or plutonium-239 (). The first full-scale thermonuclear test was carried out by the United States in 1952; the concept has since been employed by most of the world's nuclear powers in the design of their weapons. Modern fusion weapons consist essentially of two main components: a nuclear fission primary stage (fueled by or ) and a separate nuclear fusion secondary stage containing thermonuclear fuel: the heavy hydrogen isotopes deuterium and tritium, or in modern weapons lithium deuteride ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gravity
In physics, gravity () is a fundamental interaction which causes mutual attraction between all things with mass or energy. Gravity is, by far, the weakest of the four fundamental interactions, approximately 1038 times weaker than the strong interaction, 1036 times weaker than the electromagnetic force and 1029 times weaker than the weak interaction. As a result, it has no significant influence at the level of subatomic particles. However, gravity is the most significant interaction between objects at the macroscopic scale, and it determines the motion of planets, stars, galaxies, and even light. On Earth, gravity gives weight to physical objects, and the Moon's gravity is responsible for sublunar tides in the oceans (the corresponding antipodal tide is caused by the inertia of the Earth and Moon orbiting one another). Gravity also has many important biological functions, helping to guide the growth of plants through the process of gravitropism and influencing the circ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Exothermic
In thermodynamics, an exothermic process () is a thermodynamic process or reaction that releases energy from the system to its surroundings, usually in the form of heat, but also in a form of light (e.g. a spark, flame, or flash), electricity (e.g. a battery), or sound (e.g. explosion heard when burning hydrogen). The term ''exothermic'' was first coined by 19th-century French chemist Marcellin Berthelot. The opposite of an exothermic process is an endothermic process, one that absorbs energy usually in the form of heat. The concept is frequently applied in the physical sciences to chemical reactions where chemical bond energy is converted to thermal energy (heat). Two types of chemical reactions Exothermic and endothermic describe two types of chemical reactions or systems found in nature, as follows: Exothermic After an exothermic reaction, more energy has been released to the surroundings than was absorbed to initiate and maintain the reaction. An example would be the burn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
S-process
The slow neutron-capture process, or ''s''-process, is a series of reactions in nuclear astrophysics that occur in stars, particularly asymptotic giant branch stars. The ''s''-process is responsible for the creation (nucleosynthesis) of approximately half the atomic nuclei heavier than iron. In the ''s''-process, a seed nucleus undergoes neutron capture to form an isotope with one higher atomic mass. If the new isotope is stable, a series of increases in mass can occur, but if it is unstable, then beta decay will occur, producing an element of the next higher atomic number. The process is ''slow'' (hence the name) in the sense that there is sufficient time for this radioactive decay to occur before another neutron is captured. A series of these reactions produces stable isotopes by moving along the valley of beta-decay stable isobars in the table of nuclides. A range of elements and isotopes can be produced by the ''s''-process, because of the intervention of alpha decay steps ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
R-process
In nuclear astrophysics, the rapid neutron-capture process, also known as the ''r''-process, is a set of nuclear reactions that is responsible for the creation of approximately half of the atomic nuclei heavier than iron, the "heavy elements", with the other half produced by the p-process and ''s''-process. The ''r''-process usually synthesizes the most neutron-rich stable isotopes of each heavy element. The ''r''-process can typically synthesize the heaviest four isotopes of every heavy element, and the two heaviest isotopes, which are referred to as ''r-only nuclei'', can be created via the ''r''-process only. Abundance peaks for the ''r''-process occur near mass numbers (elements Se, Br, and Kr), (elements Te, I, and Xe) and (elements Os, Ir, and Pt). The ''r''-process entails a succession of ''rapid'' neutron captures (hence the name) by one or more heavy seed nuclei, typically beginning with nuclei in the abundance peak centered on 56Fe. The captures must be rapid in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Endothermic
In thermochemistry, an endothermic process () is any thermodynamic process with an increase in the enthalpy (or internal energy ) of the system.Oxtoby, D. W; Gillis, H.P., Butler, L. J. (2015).''Principle of Modern Chemistry'', Brooks Cole. p. 617. In such a process, a closed system usually absorbs thermal energy from its surroundings, which is heat transfer into the system. Thus, an endothermic reaction generally leads to an increase in the temperature of the system and a decrease in that of the surroundings. It may be a chemical process, such as dissolving ammonium nitrate () in water (), or a physical process, such as the melting of ice cubes. The term was coined by 19th-century French chemist Marcellin Berthelot. The opposite of an endothermic process is an exothermic process, one that releases or "gives out" energy, usually in the form of heat and sometimes as electrical energy. Thus in each term (endothermic and exothermic) the prefix refers to where heat (or electrical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Binding Energy
In physics and chemistry, binding energy is the smallest amount of energy required to remove a particle from a system of particles or to disassemble a system of particles into individual parts. In the former meaning the term is predominantly used in condensed matter physics, atomic physics, and chemistry, whereas in nuclear physics the term ''separation energy'' is used. A bound system is typically at a lower energy level than its unbound constituents. According to relativity theory, a decrease in the total energy of a system is accompanied by a decrease in the total mass, where . Types of binding energy There are several types of binding energy, each operating over a different distance and energy scale. The smaller the size of a bound system, the higher its associated binding energy. Mass–energy relation A bound system is typically at a lower energy level than its unbound constituents because its mass must be less than the total mass of its unbound constituents. For sys ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicon Burning Process
In astrophysics, silicon burning is a very brief sequence of nuclear fusion reactions that occur in massive stars with a minimum of about 8–11 solar masses. Silicon burning is the final stage of fusion for massive stars that have run out of the fuels that power them for their long lives in the ''main sequence'' on the Hertzsprung–Russell diagram. It follows the previous stages of hydrogen, helium, carbon, neon and oxygen burning processes. Silicon burning begins when gravitational contraction raises the star's core temperature to 2.7–3.5 billion kelvin ( GK). The exact temperature depends on mass. When a star has completed the silicon-burning phase, no further fusion is possible. The star catastrophically collapses and may explode in what is known as a Type II supernova. Nuclear fusion sequence and silicon photodisintegration After a star completes the oxygen-burning process, its core is composed primarily of silicon and sulfur. If it has sufficiently high mass, it further ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solar Mass
The solar mass () is a standard unit of mass in astronomy, equal to approximately . It is often used to indicate the masses of other stars, as well as stellar clusters, nebulae, galaxies and black holes. It is approximately equal to the mass of the Sun. This equates to about two nonillion (short scale), two quintillion (long scale) kilograms or 2000 quettagrams: The solar mass is about times the mass of Earth (), or times the mass of Jupiter (). History of measurement The value of the gravitational constant was first derived from measurements that were made by Henry Cavendish in 1798 with a torsion balance. The value he obtained differs by only 1% from the modern value, but was not as precise. The diurnal parallax of the Sun was accurately measured during the transits of Venus in 1761 and 1769, yielding a value of (9 arcseconds, compared to the present value of ). From the value of the diurnal parallax, one can determine the distance to the Sun from the geometry o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triple-alpha Process
The triple-alpha process is a set of nuclear fusion reactions by which three helium-4 nuclei (alpha particles) are transformed into carbon. Triple-alpha process in stars Helium accumulates in the cores of stars as a result of the proton–proton chain reaction and the carbon–nitrogen–oxygen cycle. Nuclear fusion reaction of two helium-4 nuclei produces beryllium-8, which is highly unstable, and decays back into smaller nuclei with a half-life of , unless within that time a third alpha particle fuses with the beryllium-8 nucleus to produce an excited resonance state of carbon-12, called the Hoyle state, which nearly always decays back into three alpha particles, but once in about 2421.3 times releases energy and changes into the stable base form of carbon-12. When a star runs out of hydrogen to fuse in its core, it begins to contract and heat up. If the central temperature rises to 108 K, six times hotter than the Sun's core, alpha particles can fuse fast enough to get pa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
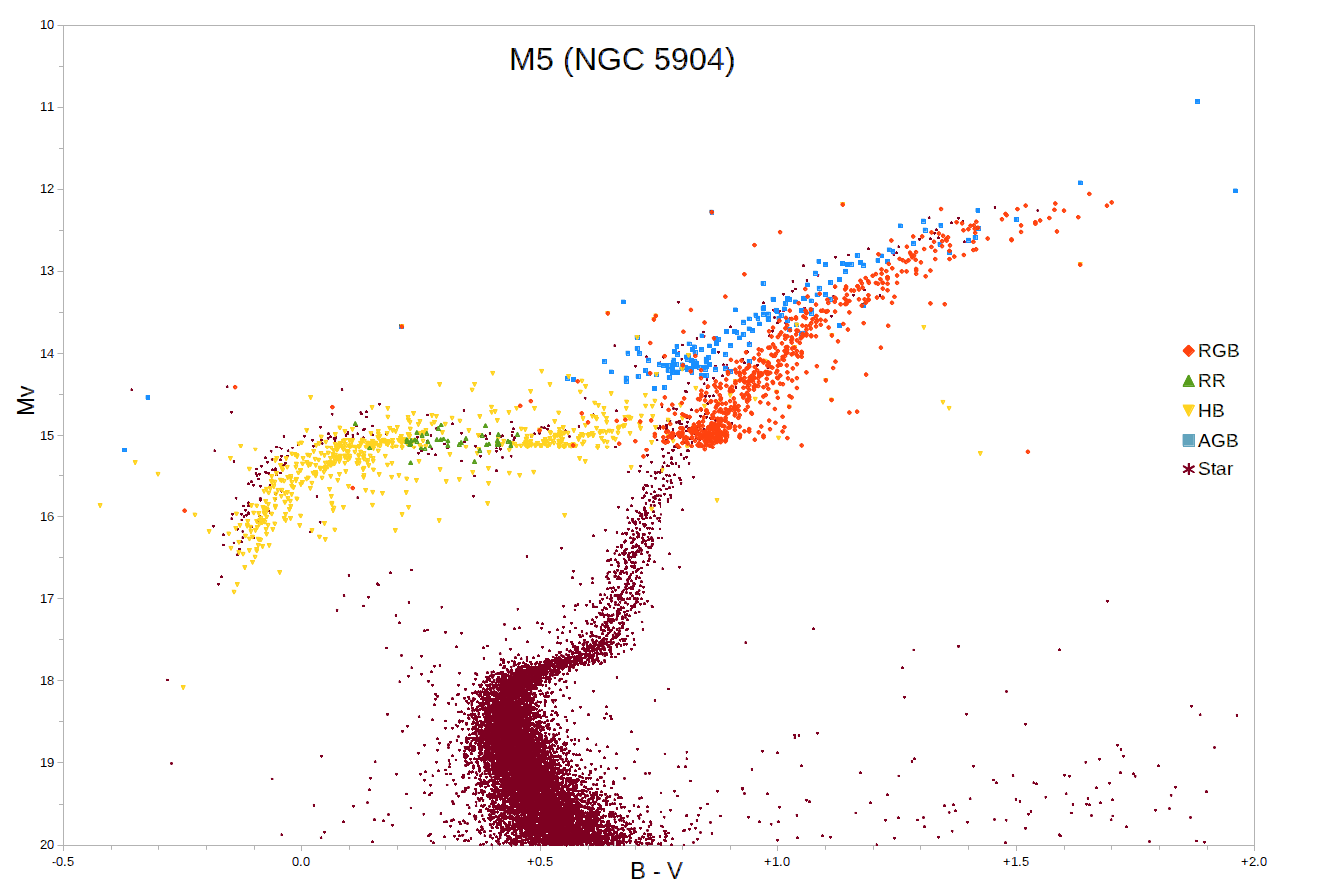
Asymptotic Giant Branch
The asymptotic giant branch (AGB) is a region of the Hertzsprung–Russell diagram populated by evolved cool luminous stars. This is a period of stellar evolution undertaken by all low- to intermediate-mass stars (about 0.5 to 8 solar masses) late in their lives. Observationally, an asymptotic-giant-branch star will appear as a bright red giant with a luminosity ranging up to thousands of times greater than the Sun. Its interior structure is characterized by a central and largely inert core of carbon and oxygen, a shell where helium is undergoing fusion to form carbon (known as helium burning), another shell where hydrogen is undergoing fusion forming helium (known as hydrogen burning), and a very large envelope of material of composition similar to main-sequence stars (except in the case of carbon stars). Stellar evolution When a star exhausts the supply of hydrogen by nuclear fusion processes in its core, the core contracts and its temperature increases, causing the outer l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium
Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense solid element. Like all alkali metals, lithium is highly reactive and flammable, and must be stored in vacuum, inert atmosphere, or inert liquid such as purified kerosene or mineral oil. When cut, it exhibits a metallic luster, but moist air corrodes it quickly to a dull silvery gray, then black tarnish. It never occurs freely in nature, but only in (usually ionic) compounds, such as pegmatitic minerals, which were once the main source of lithium. Due to its solubility as an ion, it is present in ocean water and is commonly obtained from brines. Lithium metal is isolated electrolytically from a mixture of lithium chloride and potassium chloride. The nucleus of the lithium atom verges on instability, since the two stable lithium isotopes foun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |