|
Spar (mineralogy)
Spar is an old mining or mineralogy term used to refer to crystals that have readily discernible faces. A spar will easily break or cleave into rhomboidal, cubical, or laminated fragments with smooth shiny surfaces. The various spar minerals were a historical term among miners and alchemists for any nonmetallic mineral akin to gypsum, known in Old English as spærstān, ''spear stone'', referring to its crystalline projections. Thus, the word spar in mineralogy has the same root as "spear,". Amongst miners the term "spar" today is frequently used alone to express any bright crystalline substance. Most frequently, spar describes easily cleaved, lightly colored nonmetallic minerals such as feldspar, calcite or baryte. Baryte ( Ba S O4), the main source of barium, is also called "heavy spar" (Greek "barys" means "heavy"). Calcite often forms the dogtooth spar crystals found in vugs and caves. Formation underwater Generally, a spar will form underwater, either in a phreatic zone ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dogtooth Spar
Dogtooth spar is a speleothem that consists of very large calcite crystals resembling dogs' teeth. They form through mineral precipitation of water-borne calcite. Dogtooth spar crystals are found in caves, open spaces including veins and fractures, and geodes. The crystals are generally centimeters long, but anomalous samples decimeters long exist, notably in Sitting Bull Crystal Caverns. A layer of crystalline calcite can be found underneath the surface of crystal points. The crystals typically consist of acute scalenohedrons, twelve triangular crystal faces that ideally form scalene triangles. However, modification of these faces is common, and some may have many more than three edges. Calcite crystallizes in the rhombohedral system, and the most common scalenohedron form has the Miller index 11 Spar SPAR, originally DESPAR, styled as DE SPAR, is a Dutch multinational that provides branding, supplies and support services for independently owned and operated food retai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystals
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macroscopic single crystals are usually identifiable by their geometrical shape, consisting of flat faces with specific, characteristic orientations. The scientific study of crystals and crystal formation is known as crystallography. The process of crystal formation via mechanisms of crystal growth is called crystallization or solidification. The word ''crystal'' derives from the Ancient Greek word (), meaning both "ice" and "rock crystal", from (), "icy cold, frost". Examples of large crystals include snowflakes, diamonds, and table salt. Most inorganic solids are not crystals but polycrystals, i.e. many microscopic crystals fused together into a single solid. Polycrystals include most metals, rocks, ceramics, and ice. A third category of s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iceland Spar
Iceland spar, formerly called Iceland crystal ( is, silfurberg , ) and also called optical calcite, is a transparent variety of calcite, or crystallized calcium carbonate, originally brought from Iceland, and used in demonstrating the polarization of light (see polarimetry). It occurs in large readily cleavable crystals, is easily divisible into parallelepipeds, and is remarkable for its birefringence. This means that the refractive index of the crystal is different for light of different polarization. A ray of unpolarized light passing through the crystal is divided into two rays of perpendicular polarization directed at different angles. This double refraction causes objects seen through the crystal to appear doubled. Historically, the double-refraction property of this crystal was important to understanding the nature of light as a wave. This was studied at length by Christiaan Huygens and Isaac Newton. Sir George Stokes also studied the phenomenon. Its complete explanation i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selenite (mineral)
Selenite, satin spar, desert rose, gypsum flower are crystal habit varieties of the mineral gypsum. All varieties of gypsum, including selenite and alabaster, are composed of calcium sulfate dihydrate (meaning that it has two molecules of water), with the chemical formula CaSO4·2H2O. Selenite contains no significant selenium – The similar names both derive from Greek ( 'Moon') Some of the largest crystals ever found are of selenite, the largest specimen found in the Naica Mine's Cave of the Crystals being 12 metres long and weighing 55 tons. History and etymology "Selenite" is mostly synonymous with gypsum, but from the 15th century, it has named the transparent variety that occurs in crystals or crystalline masses. The name derives through Middle English from Latin , ultimately from Greek (, ). It got this name because people historically believed the mineral waxed and waned with the cycles of the Moon. [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solution (chemistry)
In chemistry, a solution is a special type of homogeneous mixture composed of two or more substances. In such a mixture, a solute is a substance dissolved in another substance, known as a solvent. If the attractive forces between the solvent and solute particles are greater than the attractive forces holding the solute particles together, the solvent particles pull the solute particles apart and surround them. These surrounded solute particles then move away from the solid solute and out into the solution. The mixing process of a solution happens at a scale where the effects of chemical polarity are involved, resulting in interactions that are specific to solvation. The solution usually has the state of the solvent when the solvent is the larger fraction of the mixture, as is commonly the case. One important parameter of a solution is the concentration, which is a measure of the amount of solute in a given amount of solution or solvent. The term "aqueous solution" is used when ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
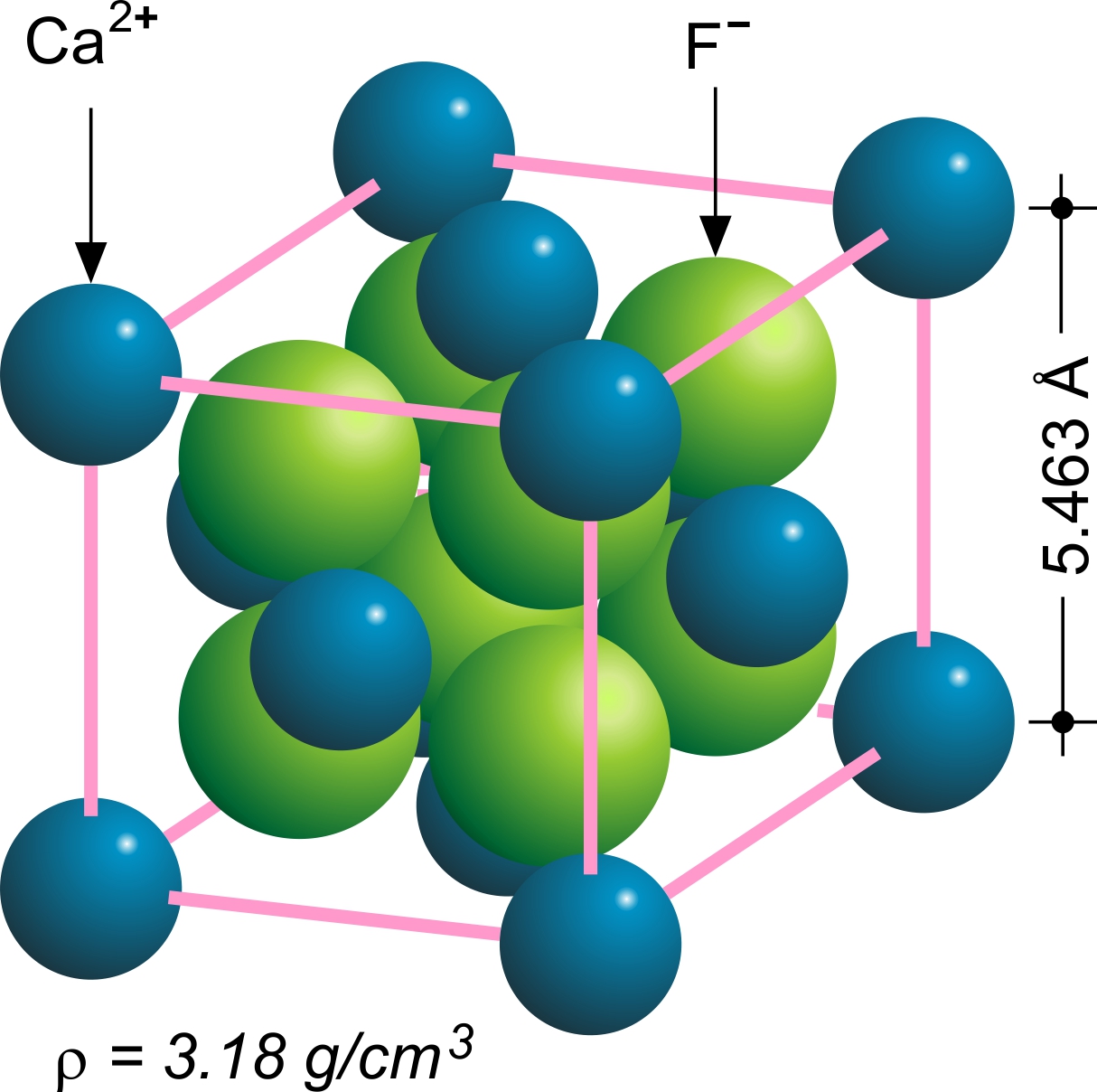
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon. The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite. Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite lenses have low dispersion, so lenses made from it exhibit less chromatic aberration, making them valuable in microscopes and telescopes. Fluorite optics are also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quartz
Quartz is a hard, crystalline mineral composed of silica (silicon dioxide). The atoms are linked in a continuous framework of SiO4 silicon-oxygen tetrahedra, with each oxygen being shared between two tetrahedra, giving an overall chemical formula of SiO2. Quartz is the second most abundant mineral in Earth's continental crust, behind feldspar. Quartz exists in two forms, the normal α-quartz and the high-temperature β-quartz, both of which are chiral. The transformation from α-quartz to β-quartz takes place abruptly at . Since the transformation is accompanied by a significant change in volume, it can easily induce microfracturing of ceramics or rocks passing through this temperature threshold. There are many different varieties of quartz, several of which are classified as gemstones. Since antiquity, varieties of quartz have been the most commonly used minerals in the making of jewelry and hardstone carvings, especially in Eurasia. Quartz is the mineral defining the val ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halite
Halite (), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride ( Na Cl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, pink, red, orange, yellow or gray depending on inclusion of other materials, impurities, and structural or isotopic abnormalities in the crystals. It commonly occurs with other evaporite deposit minerals such as several of the sulfates, halides, and borates. The name ''halite'' is derived from the Ancient Greek word for "salt", ἅλς (''háls''). Occurrence Halite dominantly occurs within sedimentary rocks where it has formed from the evaporation of seawater or salty lake water. Vast beds of sedimentary evaporite minerals, including halite, can result from the drying up of enclosed lakes and restricted seas. Such salt beds may be hundreds of meters thick and underlie broad areas. Halite occurs at the surface today in playas in regio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bottlebrushes (Cave Formation)
A bottlebrush is a cave formation which results when a stalactite is immersed in rising water which is supersaturated In physical chemistry, supersaturation occurs with a solution when the concentration of a solute exceeds the concentration specified by the value of solubility at equilibrium. Most commonly the term is applied to a solution of a solid in a liqu ... with calcium carbonate. The stalactite becomes coated with pool spar. References Speleothems {{geology-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Stalactite
A stalactite (, ; from the Greek 'stalaktos' ('dripping') via ''stalassein'' ('to drip') is a mineral formation that hangs from the ceiling of caves, hot springs, or man-made structures such as bridges and mines. Any material that is soluble and that can be deposited as a colloid, or is in suspension, or is capable of being melted, may form a stalactite. Stalactites may be composed of lava, minerals, mud, peat, pitch, sand, sinter, and amberat (crystallized urine of pack rats). A stalactite is not necessarily a speleothem, though speleothems are the most common form of stalactite because of the abundance of limestone caves. The corresponding formation on the floor of the cave is known as a stalagmite. Mnemonics have been developed for which word refers to which type of formation; one is that ''stalactite'' has a C for "ceiling", and ''stalagmite'' has a G for "ground". Another example is that ''stalactites'' "hang on ''T''ight" and ''stalagmites'' "''M''ight grow up" � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water Table
The water table is the upper surface of the zone of saturation. The zone of saturation is where the pores and fractures of the ground are saturated with water. It can also be simply explained as the depth below which the ground is saturated. The water table is the surface where the water pressure head is equal to the atmospheric pressure (where gauge pressure = 0). It may be visualized as the "surface" of the subsurface materials that are saturated with groundwater in a given vicinity. The groundwater may be from precipitation or from groundwater flowing into the aquifer. In areas with sufficient precipitation, water infiltrates through pore spaces in the soil, passing through the unsaturated zone. At increasing depths, water fills in more of the pore spaces in the soils, until a zone of saturation is reached. Below the water table, in the phreatic zone (zone of saturation), layers of permeable rock that yield groundwater are called aquifers. In less permeable soils, such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |