|
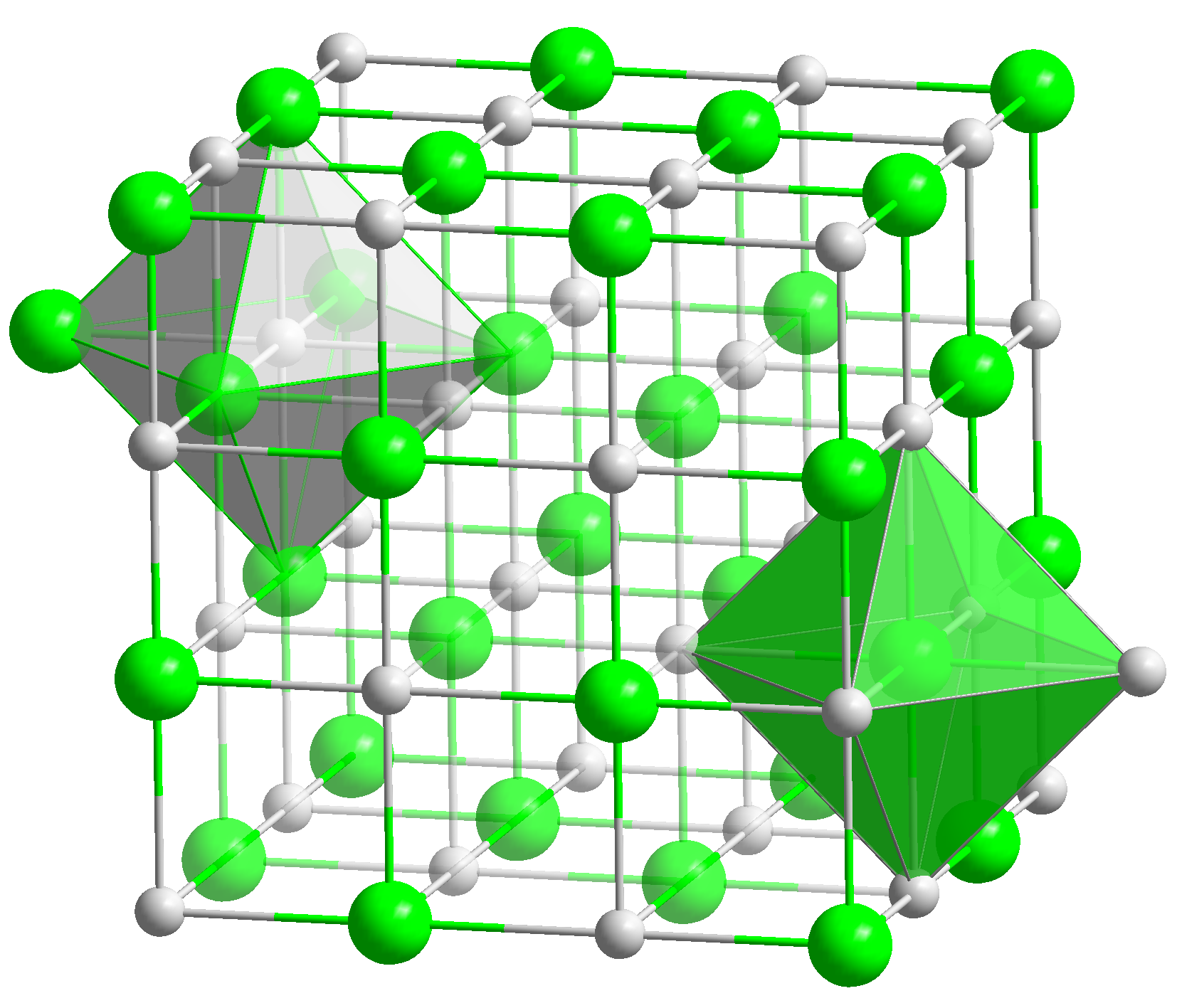
Sodium Polonide
Sodium polonide is a radioactive chemical compound with the formula Na2 Po. This salt is a polonide, a set of very chemically stable compounds of polonium.. Due to the difference in electronegativity (ΔEN) between sodium and polonium (≈ 1.1 under the Pauling system) and the slight non-metallic character of polonium, it is intermediate between intermetallic phases and ionic compounds. Production This salt may be produced from the reaction between aqueous hydrogen polonide and sodium metal: :H2Po + 2 Na → Na2Po + H2 This method of synthesis is hampered by the chemical instability of hydrogen polonide. Sodium polonide may also be produced by heating sodium and polonium together at 300–400 °C. Crystal structure Like lithium polonide and potassium polonide, sodium polonide has the antifluorite In solid state chemistry, the fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Academic Press
Academic Press (AP) is an academic book publisher founded in 1941. It was acquired by Harcourt, Brace & World in 1969. Reed Elsevier bought Harcourt in 2000, and Academic Press is now an imprint of Elsevier. Academic Press publishes reference books, serials and online products in the subject areas of: * Communications engineering * Economics * Environmental science * Finance * Food science and nutrition * Geophysics * Life sciences * Mathematics and statistics * Neuroscience * Physical sciences * Psychology Well-known products include the ''Methods in Enzymology'' series and encyclopedias such as ''The International Encyclopedia of Public Health'' and the ''Encyclopedia of Neuroscience''. See also * Akademische Verlagsgesellschaft (AVG) — the German predecessor, founded in 1906 by Leo Jolowicz (1868–1940), the father of Walter Jolowicz Walter may refer to: People * Walter (name), both a surname and a given name * Little Walter, American blues harmonica player Marion Wa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polonium
Polonium is a chemical element with the symbol Po and atomic number 84. Polonium is a chalcogen. A rare and highly radioactive metal with no stable isotopes, polonium is chemically similar to selenium and tellurium, though its metallic character resembles that of its horizontal neighbors in the periodic table: thallium, lead, and bismuth. Due to the short half-life of all its isotopes, its natural occurrence is limited to tiny traces of the fleeting polonium-210 (with a half-life of 138 days) in uranium ores, as it is the penultimate daughter of natural uranium-238. Though slightly longer-lived isotopes exist, they are much more difficult to produce. Today, polonium is usually produced in milligram quantities by the neutron irradiation of bismuth. Due to its intense radioactivity, which results in the radiolysis of chemical bonds and radioactive self-heating, its chemistry has mostly been investigated on the trace scale only. Polonium was discovered in July 1898 by Marie Sk� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Compounds
Sodium atoms have 11 electrons, one more than the stable configuration of the noble gas neon. As a result, sodium usually forms ionic compounds involving the Na+ cation. Sodium is a reactive alkali metal and is much more stable in ionic compounds. It can also form intermetallic compounds and organosodium compounds. Sodium compounds are often soluble in water. Metallic sodium Metallic sodium is generally less reactive than potassium and more reactive than lithium. Sodium metal is highly reducing, with the standard reduction potential for the Na+/Na couple being −2.71 volts, though potassium and lithium have even more negative potentials. The thermal, fluidic, chemical, and nuclear properties of molten sodium metal have caused it to be one of the main coolants of choice for the fast breeder reactor. Such nuclear reactors are seen as a crucial step for the production of clean energy. Salts and oxides Sodium compounds are of immense commercial importance, being particularly centra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antifluorite
In solid state chemistry, the fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ions occupy the regular sites of a face-centered cubic (FCC) structure. Many compounds, notably the common mineral fluorite (CaF2), adopt this structure. Many compounds with formula M2X have an antifluorite structure. In these the locations of the anions and cations are reversed relative to fluorite (an anti-structure); the anions occupy the FCC regular sites whereas the cations occupy the tetrahedral interstitial sites. For example, Magnesium silicide, Mg2Si, has a lattice parameter of 6.338 Å with magnesium cations occupying the tetrahedral interstitial sites, in which each silicide anion is surrounded by eight magnesium cations and each magnesium cation is surrounded by four silicide anions in a tetrahedral fashion. File:Fluorite Structure.jpg, The fluorite structure of calcium fluoride CaF2. File:Anti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Polonide
Potassium polonide is a chemical compound with the formula K2 Po. It is a polonide, a set of very chemically stable compounds of polonium.. Characteristics Potassium polonide is thermally more unstable and has stronger electron affinity than potassium telluride (K2Te). Production Potassium polonide may be produced from a redox reaction between hydrogen polonide and potassium metal: :H2Po + 2 K → K2Po + H2 It may also be produced by heating potassium and polonium together at 300–400 °C. At higher temperature, this reaction may reverse. Crystal structure Like sodium polonide, potassium polonide has the antifluorite In solid state chemistry, the fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ions occupy the regular sites of a face-centered cubic (FCC) structure ... structure. References {{inorganic-chemistry-stub Polonides Potassium compounds Fluorite crystal structur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium Polonide
Lithium polonide is a chemical compound with the formula Li2 Po. It is a polonide, a set of very chemically stable compounds of polonium.. Production Lithium polonide may be produced from a redox reaction between aqueous hydrogen polonide and lithium metal or from an acid-base reaction of H2Po with strong lithium-containing bases: :H2Po + 2 Li → Li2Po + H2 It may also be produced by heating lithium and polonium together at 300–400 °C. Crystal structure Like sodium polonide, lithium polonide has the antifluorite In solid state chemistry, the fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ions occupy the regular sites of a face-centered cubic (FCC) structure ... structure. References {{inorganic-chemistry-stub Lithium compounds Polonides Fluorite crystal structure ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Polonide
Polonium hydride (also known as polonium dihydride, hydrogen polonide, or polane) is a chemical compound with the formula Po H2. It is a liquid at room temperature, the second hydrogen chalcogenide with this property after water. It is very unstable chemically and tends to decompose into elemental polonium and hydrogen. It is a volatile and very labile compound, from which many polonides can be derived. Additionally, like all polonium compounds, it is highly radioactive. Preparation Polonium hydride cannot be produced by direct reaction from the elements upon heating. Other unsuccessful routes to synthesis include the reaction of polonium tetrachloride (PoCl4) with lithium aluminium hydride (LiAlH4), which only produces elemental polonium, and the reaction of hydrochloric acid with magnesium polonide (MgPo). The fact that these synthesis routes do not work may be caused by the radiolysis of polonium hydride upon formation. Trace quantities of polonium hydride may be prepared by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Intermetallic
An intermetallic (also called an intermetallic compound, intermetallic alloy, ordered intermetallic alloy, and a long-range-ordered alloy) is a type of metallic alloy that forms an ordered solid-state compound between two or more metallic elements. Intermetallics are generally hard and brittle, with good high-temperature mechanical properties. They can be classified as stoichiometric or nonstoichiometic intermetallic compounds. Although the term "intermetallic compounds", as it applies to solid phases, has been in use for many years, its introduction was regretted, for example by Hume-Rothery in 1955. Definitions Research definition Schulze in 1967 defined intermetallic compounds as ''solid phases containing two or more metallic elements, with optionally one or more non-metallic elements, whose crystal structure differs from that of the other constituents''. Under this definition, the following are included: #Electron (or Hume-Rothery) compounds #Size packing phases. e.g. Lav ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding. The loosely defined term electropositivity is the opposite of electronegativity: it characterizes an element's tendency to donate valence electrons. On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more "pull" it will have on electrons) and the number and location ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polonide
A polonide is a chemical compound of the radioactive element polonium with any element less electronegative than polonium. Polonides are usually prepared by a direct reaction between the elements at temperatures of around 300–400 °C... They are amongst the most chemically stable compounds of polonium,. and can be divided into two broad groups: *ionic polonides, which appear to contain the Po2− anion; *intermetallic polonides, in which the bonding is more complex. Some polonides are intermediate between these two cases and others are non-stoichiometric compounds. Alloys containing polonium are also classed as polonides. As polonium is immediately below tellurium in the periodic table, there are many chemical and structural similarities between polonides and tellurides. Naturally occurring polonides Lead polonide (PbPo) occurs naturally, as lead is produced in the alpha decay of polonium. Ionic polonides The polonides of the most electropositive metals show classic ionic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable isotope is 23Na. The free metal does not occur in nature, and must be prepared from compounds. Sodium is the sixth most abundant element in the Earth's crust and exists in numerous minerals such as feldspars, sodalite, and halite (NaCl). Many salts of sodium are highly water-soluble: sodium ions have been leached by the action of water from the Earth's minerals over eons, and thus sodium and chlorine are the most common dissolved elements by weight in the oceans. Sodium was first isolated by Humphry Davy in 1807 by the electrolysis of sodium hydroxide. Among many other useful sodium compounds, sodium hydroxide (lye) is used in soap manufacture, and sodium chloride (edible salt) is a de-icing agent and a nutrient for animals including h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Oxide
Sodium oxide is a chemical compound with the formula Na2 O. It is used in ceramics and glasses. It is a white solid but the compound is rarely encountered. Instead "sodium oxide" is used to describe components of various materials such as glasses and fertilizers which contain oxides that include sodium and other elements. Structure The structure of sodium oxide has been determined by X-ray crystallography. Most alkali metal oxides M2O (M = Li, Na, K, Rb) crystallise in the antifluorite structure. In this motif the positions of the anions and cations are reversed relative to their positions in CaF2, with sodium ions tetrahedrally coordinated to 4 oxide ions and oxide cubically coordinated to 8 sodium ions. Preparation Sodium oxide is produced by the reaction of sodium with sodium hydroxide, sodium peroxide, or sodium nitrite: : 2 NaOH + 2 Na → 2 Na2O + H2 To the extent that NaOH is contaminated with water, correspondingly greater amounts of sodium are employed. Excess s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |