|
Silver-108
Naturally occurring silver (47Ag) is composed of the two stable isotopes 107Ag and 109Ag in almost equal proportions, with 107Ag being slightly more abundant (51.839% natural abundance). 40 radioisotopes have been characterized with the most stable being 105Ag with a half-life of 41.29 days, 111Ag with a half-life of 7.43 days, and 112Ag with a half-life of 3.13 hours. All of the remaining radioactive isotopes have half-lives that are less than an hour, and the majority of these have half-lives that are less than 3 minutes. This element has numerous meta states, with the most stable being 108mAg (half-life 439 years), 110mAg (half-life 249.86 days) and 106mAg (half-life 8.28 days). Isotopes of silver range in atomic weight from 91.960 u (92Ag) to 132.969 u (133Ag). The primary decay mode before the most abundant stable isotope, 107Ag, is electron capture and the primary mode after is beta decay. The primary decay products before 107Ag are palladium (element 46) isot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Meta State
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy higher energy levels than in the ground state of the same nucleus. "Metastable" describes nuclei whose excited states have half-lives 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half life (ordinarily on the order of 10−12 seconds). The term "metastable" is usually restricted to isomers with half-lives of 10−9 seconds or longer. Some references recommend 5 × 10−9 seconds to distinguish the metastable half life from the normal "prompt" gamma-emission half-life. Occasionally the half-lives are far longer than this and can last minutes, hours, or years. For example, the nuclear isomer survives so long (at least 1015 years) that it has never been observed to decay spontaneously. The half-life of a nuclear isomer can even exceed that of the ground state of the same nuclide, as shown by as well as , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isomeric Transition
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy higher energy levels than in the ground state of the same nucleus. "Metastable" describes nuclei whose excited states have half-lives 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half life (ordinarily on the order of 10−12 seconds). The term "metastable" is usually restricted to isomers with half-lives of 10−9 seconds or longer. Some references recommend 5 × 10−9 seconds to distinguish the metastable half life from the normal "prompt" gamma-emission half-life. Occasionally the half-lives are far longer than this and can last minutes, hours, or years. For example, the nuclear isomer survives so long (at least 1015 years) that it has never been observed to decay spontaneously. The half-life of a nuclear isomer can even exceed that of the ground state of the same nuclide, as shown by as well as , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silver
Silver is a chemical element with the Symbol (chemistry), symbol Ag (from the Latin ', derived from the Proto-Indo-European wikt:Reconstruction:Proto-Indo-European/h₂erǵ-, ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical conductivity, thermal conductivity, and reflectivity of any metal. The metal is found in the Earth's crust in the pure, free elemental form ("native silver"), as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite. Most silver is produced as a byproduct of copper, gold, lead, and zinc Refining (metallurgy), refining. Silver has long been valued as a precious metal. Silver metal is used in many bullion coins, sometimes bimetallism, alongside gold: while it is more abundant than gold, it is much less abundant as a native metal. Its purity is typically measured on a per-mille basis; a 94%-pure alloy is described as "0.940 fine". As one of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Meteorite
A meteorite is a solid piece of debris from an object, such as a comet, asteroid, or meteoroid, that originates in outer space and survives its passage through the atmosphere to reach the surface of a planet or Natural satellite, moon. When the original object enters the atmosphere, various factors such as friction, pressure, and chemical interactions with the atmospheric gases cause it to heat up and radiate energy. It then becomes a meteor and forms a Meteoroid#Fireball, fireball, also known as a shooting star; astronomers call the brightest examples "Bolide#Astronomy, bolides". Once it settles on the larger body's surface, the meteor becomes a meteorite. Meteorites vary greatly in size. For geologists, a bolide is a meteorite large enough to create an impact crater. Meteorites that are recovered after being observed as they transit the atmosphere and Impact event, impact the Earth are called meteorite falls. All others are known as meteorite finds. Meteorites have traditiona ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Silver
Naturally occurring silver (47Ag) is composed of the two stable isotopes 107Ag and 109Ag in almost equal proportions, with 107Ag being slightly more abundant (51.839% natural abundance). 40 radioisotopes have been characterized with the most stable being 105Ag with a half-life of 41.29 days, 111Ag with a half-life of 7.43 days, and 112Ag with a half-life of 3.13 hours. All of the remaining radioactive isotopes have half-lives that are less than an hour, and the majority of these have half-lives that are less than 3 minutes. This element has numerous meta states, with the most stable being 108mAg (half-life 439 years), 110mAg (half-life 249.86 days) and 106mAg (half-life 8.28 days). Isotopes of silver range in atomic weight from 91.960 u (92Ag) to 132.969 u (133Ag). The primary decay mode before the most abundant stable isotope, 107Ag, is electron capture and the primary mode after is beta decay. The primary decay products before 107Ag are palladium (element 46) isot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
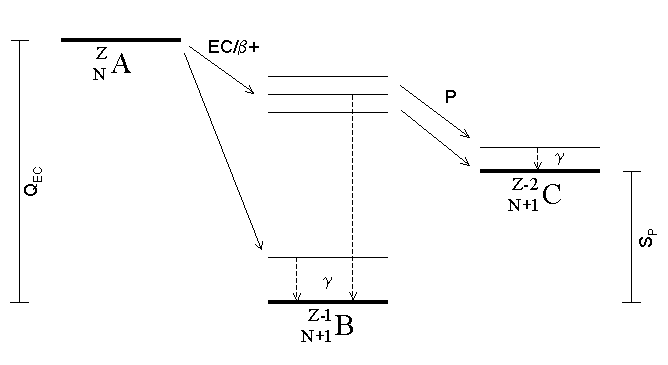
Neutron Emission
Neutron emission is a mode of radioactive decay in which one or more neutrons are ejected from a nucleus. It occurs in the most neutron-rich/proton-deficient nuclides, and also from excited states of other nuclides as in photoneutron emission and beta-delayed neutron emission. As only a neutron is lost by this process the number of protons remains unchanged, and an atom does not become an atom of a different element, but a different isotope of the same element. Neutrons are also produced in the spontaneous and induced fission of certain heavy nuclides. Spontaneous neutron emission As a consequence of the Pauli exclusion principle, nuclei with an excess of protons or neutrons have a higher average energy per nucleon. Nuclei with a sufficient excess of neutrons have a greater energy than the combination of a free neutron and a nucleus with one less neutron, and therefore can decay by neutron emission. Nuclei which can decay by this process are described as lying beyond the neutron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fission Product
Nuclear fission products are the atomic fragments left after a large atomic nucleus undergoes nuclear fission. Typically, a large nucleus like that of uranium fissions by splitting into two smaller nuclei, along with a few neutrons, the release of heat energy (kinetic energy of the nuclei), and gamma rays. The two smaller nuclei are the ''fission products''. (See also Fission products (by element)). About 0.2% to 0.4% of fissions are ternary fissions, producing a third light nucleus such as helium-4 (90%) or tritium (7%). The fission products themselves are usually unstable and therefore radioactive. Due to being relatively neutron-rich for their atomic number, many of them quickly undergo beta decay. This releases additional energy in the form of beta particles, antineutrinos, and gamma rays. Thus, fission events normally result in beta and gamma radiation, even though this radiation is not produced directly by the fission event itself. The produced radionuclides have varyi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Spontaneous Fission
Spontaneous fission (SF) is a form of radioactive decay that is found only in very heavy chemical elements. The nuclear binding energy of the elements reaches its maximum at an atomic mass number of about 56 (e.g., iron-56); spontaneous breakdown into smaller nuclei and a few isolated nuclear particles becomes possible at greater atomic mass numbers. History By 1908, physicists understood that alpha decay involved ejection of helium nuclei from a decaying atom. Like cluster decay, alpha decay is not typically categorized as a process of fission. The first nuclear fission process discovered was fission induced by neutrons. Because cosmic rays produce some neutrons, it was difficult to distinguish between induced and spontaneous events. Cosmic rays can be reliably shielded by a thick layer of rock or water. Spontaneous fission was identified in 1940 by Soviet physicists Georgy Flyorov and Konstantin Petrzhak by their observations of uranium in the Moscow Metro Dinamo station ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proton Emission
Proton emission (also known as proton radioactivity) is a rare type of radioactive decay in which a proton is ejected from a nucleus. Proton emission can occur from high-lying excited states in a nucleus following a beta decay, in which case the process is known as beta-delayed proton emission, or can occur from the ground state (or a low-lying isomer) of very proton-rich nuclei, in which case the process is very similar to alpha decay. For a proton to escape a nucleus, the proton separation energy must be negative—the proton is therefore unbound, and tunnels out of the nucleus in a finite time. Proton emission is not seen in naturally occurring isotopes; proton emitters can be produced via nuclear reactions, usually using linear particle accelerators. Although prompt (i.e. not beta-delayed) proton emission was observed from an isomer in cobalt-53 as early as 1969, no other proton-emitting states were found until 1981, when the proton radioactive ground states of lutetium-15 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solar System
The Solar SystemCapitalization of the name varies. The International Astronomical Union, the authoritative body regarding astronomical nomenclature, specifies capitalizing the names of all individual astronomical objects but uses mixed "Solar System" and "solar system" structures in theinaming guidelines document. The name is commonly rendered in lower case ('solar system'), as, for example, in the ''Oxford English Dictionary'' an''Merriam-Webster's 11th Collegiate Dictionary''. is the gravity, gravitationally bound system of the Sun and the objects that orbit it. It Formation and evolution of the Solar System, formed 4.6 billion years ago from the gravitational collapse of a giant interstellar molecular cloud. The solar mass, vast majority (99.86%) of the system's mass is in the Sun, with most of the Jupiter mass, remaining mass contained in the planet Jupiter. The four inner Solar System, inner system planets—Mercury (planet), Mercury, Venus, Earth and Mars—are terrest ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Accretion (astrophysics)
In astrophysics, accretion is the accumulation of particles into a massive object by gravitationally attracting more matter, typically gaseous matter, in an accretion disk. Most astronomical objects, such as galaxies, stars, and planets, are formed by accretion processes. Overview The accretion model that Earth and the other terrestrial planets formed from meteoric material was proposed in 1944 by Otto Schmidt, followed by the ''protoplanet theory'' of William McCrea (1960) and finally the ''capture theory'' of Michael Woolfson. For details of Kant's position, see In 1978, Andrew Prentice resurrected the initial Laplacian ideas about planet formation and developed the ''modern Laplacian theory''. None of these models proved completely successful, and many of the proposed theories were descriptive. The 1944 accretion model by Otto Schmidt was further developed in a quantitative way in 1969 by Viktor Safronov. He calculated, in detail, the different stages of terrestrial plane ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleosynthetic
Nucleosynthesis is the process that creates new atomic nuclei from pre-existing nucleons (protons and neutrons) and nuclei. According to current theories, the first nuclei were formed a few minutes after the Big Bang, through nuclear reactions in a process called Big Bang nucleosynthesis. After about 20 minutes, the universe had expanded and cooled to a point at which these high-energy collisions among nucleons ended, so only the fastest and simplest reactions occurred, leaving our universe containing hydrogen and helium. The rest is traces of other elements such as lithium and the hydrogen isotope deuterium. Nucleosynthesis in stars and their explosions later produced the variety of elements and isotopes that we have today, in a process called cosmic chemical evolution. The amounts of total mass in elements heavier than hydrogen and helium (called 'metals' by astrophysicists) remains small (few percent), so that the universe still has approximately the same composition. Stars stel ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |

_Halflife_of_Radionulides_depending_on_Z²_to_A_ratio.png)