|
Serine Hydrolase
Serine hydrolases are one of the largest known enzyme classes comprising approximately ~200 enzymes or 1% of the genes in the human proteome. A defining characteristic of these enzymes is the presence of a particular serine at the active site, which is used for the hydrolysis of substrates. The hydrolysis of the ester or peptide bond proceeds in two steps. First, the acyl part of the substrate (the acid part of an ester or the part of a peptide ending in a carboxyl group) is transferred to the serine, making a new ester or amide bond and releasing the other part of the substrate (the alcohol of an ester or the part of the peptide ending in an amino group) is released. Later, in a slower step, the bond between the serine and the acyl group is hydrolyzed by water or hydroxide ion, regenerating free enzyme. Unlike other, non-catalytic, serines, the reactive serine of these hydrolases is typically activated by a proton relay involving a catalytic triad consisting of the serine, an acidi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products. Almost all metabolic processes in the cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are catalytic RNA molecules, called ribozymes. Enzymes' specificity comes from their unique three-dimensional structures. Like all catalysts, enzymes increase the react ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Serine Protease
Serine proteases (or serine endopeptidases) are enzymes that cleave peptide bonds in proteins. Serine serves as the nucleophilic amino acid at the (enzyme's) active site. They are found ubiquitously in both eukaryotes and prokaryotes. Serine proteases fall into two broad categories based on their structure: chymotrypsin-like (trypsin-like) or subtilisin-like. Classification The MEROPS protease classification system counts 16 superfamilies (as of 2013) each containing many families. Each superfamily uses the catalytic triad or dyad in a different protein fold and so represent convergent evolution of the catalytic mechanism. The majority belong to the S1 family of the PA clan (superfamily) of proteases. For superfamilies, P: superfamily, containing a mixture of nucleophile class families, S: purely serine proteases. superfamily. Within each superfamily, families are designated by their catalytic nucleophile, (S: serine proteases). Substrate specificity Serine p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diacylglycerol Lipase
Diacylglycerol lipase, also known as DAG lipase, DAGL or DGL, is a key enzyme in the biosynthesis of the endocannabinoid 2-arachidonoylglycerol. It catalyzes the hydrolysis of diacylglycerol, releasing a free fatty acid In chemistry, particularly in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated or unsaturated. Most naturally occurring fatty acids have an unbranched chain of an even number of carbon atoms, f ... and monoacylglycerol. Two separate genes encoding DGL enzymes have been cloned, termed DGLα () and DGLβ (), that share 33% sequence identity. Inhibitors The enzyme has been described to be inhibited selectively by two agents, RHC80267 and tetrahydrolipstatin. References External links * * {{Leukotrienergics EC 3.1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PNPLA2
Adipose triglyceride lipase, also known as patatin-like phospholipase domain-containing protein 2 and ATGL, is an enzyme that in humans is encoded by the ''PNPLA2'' gene. ATGL catalyses the first reaction of lipolysis, where triacylglycerols are hydrolysed to diacylglycerols. Properties ATGL has very high substrate specificity for triacylglycerols. It contains a catalytic dyad using serine-aspartic acid. Function ATGL catalyses the first reaction of lipolysis. It hydrolysis triacylglycerols to diacylglycerols by attacking the fatty acid attached to carbon-3 of glycerol. ATGL acts as a control mechanism of lipolysis, as variations in diacylglycerol concentration impact enzymes in later stages of lipolysis. Clinical significance Defects in ATGL can cause problems in lipolysis, leading to neutral lipid storage disease. As triacylglycerols are not hydrolysed to diacylglycerols, there is a build-up of triacylglycerol droplets in granulocyte Granulocytes are cells in th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monoacylglycerol Lipase
Monoacylglycerol lipase (EC 3.1.1.23; systematic name glycerol-ester acylhydrolase, also known as MAG lipase, acylglycerol lipase, MAGL, MGL or MGLL) is an enzyme that, in humans, is encoded by the ''MGLL'' gene. MAGL is a 33-kDa, membrane-associated member of the serine hydrolase superfamily and contains the classical GXSXG consensus sequence common to most serine hydrolases. The catalytic triad has been identified as Ser122, His269, and Asp239. Function Monoacylglycerol lipase catalyzes a reaction that uses water molecules to break the glycerol monoesters of long-chain fatty acids: : hydrolyses glycerol monoesters of long-chain fatty acids It functions together with hormone-sensitive lipase (LIPE) to hydrolyze intracellular triglyceride stores in adipocytes and other cells to fatty acids and glycerol. MGLL may also complement lipoprotein lipase (LPL) in completing hydrolysis of monoglycerides resulting from degradation of lipoprotein triglycerides. Monoacylglycerol li ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hormone Sensitive Lipase
Hormone-sensitive lipase (, HSL), also previously known as cholesteryl ester hydrolase (CEH), sometimes referred to as triacylglycerol lipase, is an enzyme that, in humans, is encoded by the ''LIPE'' gene, and catalyzes the following reaction: :(1) diacylglycerol + H2O = monoacylglycerol + a carboxylate :(2) triacylglycerol + H2O = diacylglycerol + a carboxylate :(3) monoacylglycerol + -H2O = glycerol + a carboxylate HSL is an intracellular neutral lipase capable of hydrolyzing a variety of esters. The enzyme has a long and a short form. The long form is expressed in steroidogenic tissues such as testis A testicle or testis (plural testes) is the male reproductive gland or gonad in all bilaterians, including humans. It is homologous to the female ovary. The functions of the testes are to produce both sperm and androgens, primarily testostero ..., where it converts cholesteryl esters to free cholesterol for steroid hormone production. The short form is expressed in adipoc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lipoprotein Lipase
Lipoprotein lipase (LPL) (EC 3.1.1.34, systematic name triacylglycerol acylhydrolase (lipoprotein-dependent)) is a member of the lipase gene family, which includes pancreatic lipase, hepatic lipase, and endothelial lipase. It is a water-soluble enzyme that hydrolyzes triglycerides in lipoproteins, such as those found in chylomicrons and very low-density lipoproteins (VLDL), into two free fatty acids and one monoacylglycerol molecule: : triacylglycerol + H2O = diacylglycerol + a carboxylate It is also involved in promoting the cellular uptake of chylomicron remnants, cholesterol-rich lipoproteins, and free fatty acids. LPL requires ApoC-II as a cofactor. LPL is attached to the luminal surface of endothelial cells in capillaries by the protein glycosylphosphatidylinositol HDL-binding protein 1 (GPIHBP1) and by heparan sulfated peptidoglycans. It is most widely distributed in adipose, heart, and skeletal muscle tissue, as well as in lactating mammary glands. Synthesis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Endothelial Lipase
Endothelial lipase (LIPG) is a form of lipase secreted by vascular endothelial cells in tissues with high metabolic rates and vascularization, such as the liver, lung, kidney, and thyroid gland. The LIPG enzyme is a vital component to many biological processes. These processes include lipoprotein metabolism, cytokine expression, and lipid composition in cells. Unlike the lipases that hydrolyze Triglycerides, endothelial lipase primarily hydrolyzes phospholipids. Due to the hydrolysis specificity, endothelial lipase contributes to multiple vital systems within the body. On the contrary to the beneficial roles that LIPG plays within the body, endothelial lipase is thought to play a potential role in cancer and inflammation. Knowledge obtained in vitro and in vivo suggest the relations to these conditions, but human interaction knowledge lacks due to the recent discovery of endothelial lipase. Endothelial lipase was first characterized in 1999. The two independent research groups whic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
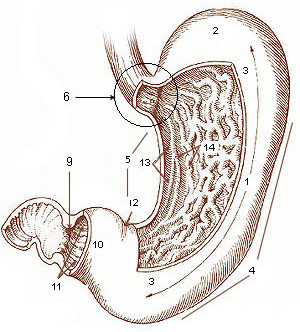
Gastric Lipase
The stomach is a muscular, hollow organ in the gastrointestinal tract of humans and many other animals, including several invertebrates. The stomach has a dilated structure and functions as a vital organ in the digestive system. The stomach is involved in the gastric phase of digestion, following chewing. It performs a chemical breakdown by means of enzymes and hydrochloric acid. In humans and many other animals, the stomach is located between the oesophagus and the small intestine. The stomach secretes digestive enzymes and gastric acid to aid in food digestion. The pyloric sphincter controls the passage of partially digested food (chyme) from the stomach into the duodenum, where peristalsis takes over to move this through the rest of intestines. Structure In the human digestive system, the stomach lies between the oesophagus and the duodenum (the first part of the small intestine). It is in the left upper quadrant of the abdominal cavity. The top of the stomach lies ag ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hepatic Lipase
Hepatic lipase (HL), also called hepatic triglyceride lipase (HTGL) or LIPC (for "lipase, hepatic"), is a form of lipase, catalyzing the hydrolysis of triacylglyceride. Hepatic lipase is coded by chromosome 15 and its gene is also often referred to as HTGL or LIPC. Hepatic lipase is expressed mainly in liver cells, known as hepatocytes, and endothelial cells of the liver. The hepatic lipase can either remain attached to the liver or can unbind from the liver endothelial cells and is free to enter the body's circulation system. When bound on the endothelial cells of the liver, it is often found bound to HSPG, heparan sulfate proteoglycans (HSPG), keeping HL inactive and unable to bind to HDL (high density lipoprotein) or IDL (intermediate density lipoprotein). When it is free in the bloodstream, however, it is found associated with HDL to maintain it inactive. This is because the triacylglycerides in HDL serve as a substrate, but the lipoprotein contains proteins around the triac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pancreatic Lipase
Triglyceride lipases () are a family of lipolytic enzymes that hydrolyse ester linkages of triglycerides. Lipases are widely distributed in animals, plants and prokaryotes. At least three tissue-specific isozymes exist in higher vertebrates, pancreatic, hepatic and gastric/lingual. These lipases are closely related to each other and to lipoprotein lipase (), which hydrolyses triglycerides of chylomicrons and very low density lipoproteins (VLDL). The most conserved region in all these proteins is centred on a serine residue which has been shown to participate, with a histidine and an aspartic acid residue, in a charge relay system. Such a region is also present in lipases of prokaryotic origin and in lecithin-cholesterol acyltransferase () (LCAT), which catalyzes fatty acid transfer between phosphatidylcholine and cholesterol. Human pancreatic lipase ''Pancreatic lipase'', also known as ''pancreatic triacylglycerol lipase'' or ''steapsin'', is an enzyme secreted from the pancre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lipase
Lipase ( ) is a family of enzymes that catalyzes the hydrolysis of fats. Some lipases display broad substrate scope including esters of cholesterol, phospholipids, and of lipid-soluble vitamins and sphingomyelinases; however, these are usually treated separately from "conventional" lipases. Unlike esterases, which function in water, lipases "are activated only when adsorbed to an oil–water interface". Lipases perform essential roles in digestion, transport and processing of dietary lipids in most, if not all, organisms. Structure and catalytic mechanism Classically, lipases catalyse the hydrolysis of triglycerides: :triglyceride + H2O → fatty acid + diacylglycerol :diacylglycerol + H2O → fatty acid + monacylglycerol :monacylglycerol + H2O → fatty acid + glycerol Lipases are serine hydrolases, i.e. they function by transesterification generating an acyl serine intermediate. Most lipases act at a specific position on the glycerol backbone of a lipi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |