|
Polycarbonyl
Polycarbonyl, (also known as polymeric-CO, p-CO or poly-CO) is a solid, metastable, and explosive polymer of carbon monoxide. The polymer is produced by exposing carbon monoxide to high pressures. The structure of the solid appears amorphous, but may include a zig zag of equally-spaced CO groups. Formation Poly-CO can be produced at pressures of 5.2 GPa; it is amorphous and yellow to dark red in color. Polymerisation is catalysed by blue light at slightly lower pressures in the δ-phase of solid CO. Another white, crystalline phase can be made at higher temperatures at 6 or 7 GPa. R. J. Mills discovered this solid, which was first produced in a tungsten carbide anvil in 1947. Originally this was thought to be polymeric carbon suboxide, but the formation does not yield any gas byproduct such as carbon dioxide. The yield of the solid can be up to 95%. Properties The polymer is stable above about 80K. Below this temperature the ε form of solid molecular CO is formed instead. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxocarbons
In chemistry, an oxocarbon or oxide of carbon is a chemical compound consisting only of carbon and oxygen. The simplest and most common oxocarbons are carbon monoxide (CO) and carbon dioxide (). Many other stable (practically if not thermodynamically) or metastable oxides of carbon are known, but they are rarely encountered, such as carbon suboxide ( or ) and mellitic anhydride (). Many other oxides are known today, most of them synthesized since the 1960s. Some of these new oxides are stable at room temperature. Some are metastable or stable only at very low temperatures, but decompose to simpler oxocarbons when warmed. Many are inherently unstable and can be observed only momentarily as intermediates in chemical reactions or are so reactive that they exist only in gas phase or have only been detected by matrix isolation. Graphene oxide and other stable polymeric carbon oxides with unbounded molecular structures exist. Overview Carbon dioxide (CO2) occurs widely in nature, and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Metastable
In chemistry and physics, metastability denotes an intermediate Energy level, energetic state within a dynamical system other than the system's ground state, state of least energy. A ball resting in a hollow on a slope is a simple example of metastability. If the ball is only slightly pushed, it will settle back into its hollow, but a stronger push may start the ball rolling down the slope. Bowling pins show similar metastability by either merely wobbling for a moment or tipping over completely. A common example of metastability in science is isomerisation. Higher energy isomers are long lived because they are prevented from rearranging to their preferred ground state by (possibly large) barriers in the potential energy. During a metastable state of finite lifetime, all state-describing parameters reach and hold stationary values. In isolation: *the state of least energy is the only one the system will inhabit for an indefinite length of time, until more external energy is added ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymer
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part") is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals. The term "polymer" derives from the Greek word πολύς (''polus'', meaning "many, much") and μέρος (''meros'' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simplest molecule of the oxocarbon family. In coordination complexes the carbon monoxide ligand is called carbonyl. It is a key ingredient in many processes in industrial chemistry. The most common source of carbon monoxide is the partial combustion of carbon-containing compounds, when insufficient oxygen or heat is present to produce carbon dioxide. There are also numerous environmental and biological sources that generate and emit a significant amount of carbon monoxide. It is important in the production of many compounds, including drugs, fragrances, and fuels. Upon emission into the atmosphere, carbon monoxide affects several processes that contribute to climate change. Carbon monoxide has important biological roles across phylogenetic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amorphous Solid
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid, glassy solid) is a solid that lacks the long-range order that is characteristic of a crystal. Etymology The term comes from the Greek ''a'' ("without"), and ''morphé'' ("shape, form"). In some older articles and books, the term was used synonymously with glass. Today, "glassy solid" or "amorphous solid" is considered the overarching concept. Polymers are often amorphous. Structure Amorphous materials have an internal structure comprising interconnected structural blocks that can be similar to the basic structural units found in the corresponding crystalline phase of the same compound. Unlike crystalline materials, however, no long-range order exists. Localized order in amorphous materials can be categorized as short or medium range order. By convention, short range order extends only to the nearest neighbor shell, typically only 1-2 atomic spacings. Medium range order is then def ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' is methyl), with the formula . Many ketones are of great importance in biology and in industry. Examples include many sugars (ketoses), many steroids (e.g., testosterone), and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amorphous
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid, glassy solid) is a solid that lacks the long-range order that is characteristic of a crystal. Etymology The term comes from the Greek ''a'' ("without"), and ''morphé'' ("shape, form"). In some older articles and books, the term was used synonymously with glass. Today, "glassy solid" or "amorphous solid" is considered the overarching concept. Polymers are often amorphous. Structure Amorphous materials have an internal structure comprising interconnected structural blocks that can be similar to the basic structural units found in the corresponding crystalline phase of the same compound. Unlike crystalline materials, however, no long-range order exists. Localized order in amorphous materials can be categorized as short or medium range order. By convention, short range order extends only to the nearest neighbor shell, typically only 1-2 atomic spacings. Medium range order is then de ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymeric Carbon Suboxide
A polymer (; Greek '' poly-'', "many" + ''-mer'', "part") is a substance or material consisting of very large molecules called macromolecules, composed of many repeating subunits. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compounds, produces unique physical properties including toughness, high elasticity, viscoelasticity, and a tendency to form amorphous and semicrystalline structures rather than crystals. The term "polymer" derives from the Greek word πολύς (''polus'', meaning "many, much") and μέρος (''meros'', meaning "part ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Energy Density
In physics, energy density is the amount of energy stored in a given system or region of space per unit volume. It is sometimes confused with energy per unit mass which is properly called specific energy or . Often only the ''useful'' or extractable energy is measured, which is to say that inaccessible energy (such as rest mass energy) is ignored. In cosmological and other general relativistic contexts, however, the energy densities considered are those that correspond to the elements of the stress–energy tensor and therefore do include mass energy as well as energy densities associated with pressure. Energy per unit volume has the same physical units as pressure and in many situations is synonymous. For example, the energy density of a magnetic field may be expressed as and behaves like a physical pressure. Likewise, the energy required to compress a gas to a certain volume may be determined by multiplying the difference between the gas pressure and the external pressure by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Magnetic Resonance
Nuclear magnetic resonance (NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field) and respond by producing an electromagnetic signal with a frequency characteristic of the magnetic field at the nucleus. This process occurs near resonance, when the oscillation frequency matches the intrinsic frequency of the nuclei, which depends on the strength of the static magnetic field, the chemical environment, and the magnetic properties of the isotope involved; in practical applications with static magnetic fields up to ca. 20 tesla, the frequency is similar to VHF and UHF television broadcasts (60–1000 MHz). NMR results from specific magnetic properties of certain atomic nuclei. Nuclear magnetic resonance spectroscopy is widely used to determine the structure of organic molecules in solution and study molecular physics and crystals as well as non-crystalline materials. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
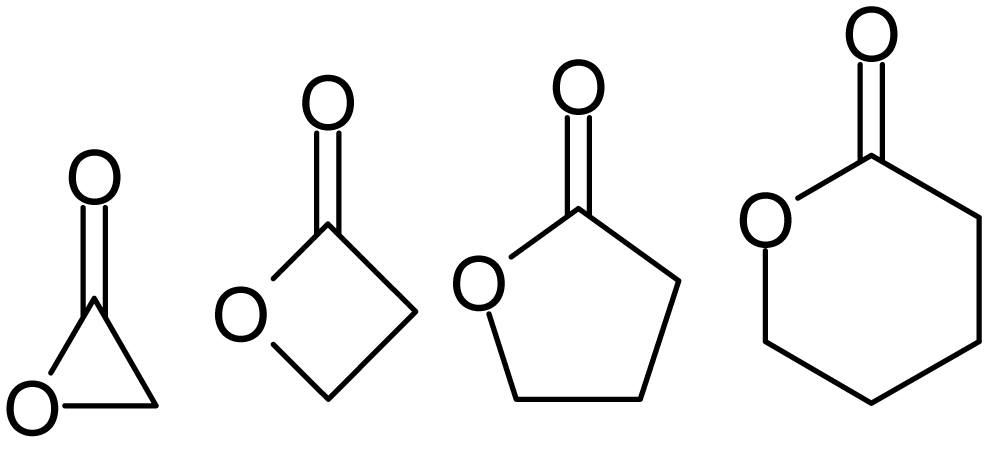
Lactone
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring. Lactones are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids, which takes place spontaneously when the ring that is formed is five- or six-membered. Lactones with three- or four-membered rings (α-lactones and β-lactones) are very reactive, making their isolation difficult. Special methods are normally required for the laboratory synthesis of small-ring lactones as well as those that contain rings larger than six-membered. Nomenclature Lactones are usually named according to the precursor acid molecule (''aceto'' = 2 carbon atoms, ''propio'' = 3, ''butyro'' = 4, ''valero'' = 5, ''capro'' = 6, etc.), with a ''-lactone'' suffix and a Greek letter prefix that specifies the number of carbon atoms in the heterocycle — that is, the distance between the relevant -OH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetronic Acid
Tetronic acid is a chemical compound, classified as a lactone, γ-lactone, with the molecular formula C4H4O3. It interconverts between keto and enol tautomers: : Many natural products such as ascorbic acid (vitamin C), penicillic acid, pulvinic acids, and abyssomicins possess the β-keto-γ-butyrolactone motif of tetronic acid. In organic synthesis, it is used as a precursor for other substituted and ring-fused furans and butenolides. It is also forms the structural core of a class of pesticides, known as tetronic acid insecticides, which includes spirodiclofen and spiromesifen. See also * 5-Hydroxy-2(5H)-furanone References {{reflist Furanones ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |