|
Pancreatic Elastase
Pancreatic elastase is a form of elastase that is produced in the acinar cells of the pancreas, initially produced as an inactive zymogen and later activated in the duodenum by trypsin. Elastases form a subfamily of serine proteases, characterized by a distinctive structure consisting of two beta barrel domains converging at the active site that hydrolyze amides and esters amongst many proteins in addition to elastin, a type of connective tissue that holds organs together. Pancreatic elastase 1 is a serine endopeptidase, a specific type of protease that has the amino acid serine at its active site. Although the recommended name is pancreatic elastase, it can also be referred to as elastase-1, pancreatopeptidase, PE, or serine elastase. The first isozyme, pancreatic elastase 1, was initially thought to be expressed in the pancreas. However it was later discovered that it was the only chymotrypsin-like elastase that was not expressed in the pancreas. In fact, pancreatic elastase is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elastase
In molecular biology, elastase is an enzyme from the class of ''proteases (peptidases)'' that break down proteins. In particular, it is a serine protease. Forms and classification Eight human genes exist for elastase: Some bacteria (including ''Pseudomonas aeruginosa'') also produce elastase. In bacteria, elastase is considered a virulence factor. Function Elastase breaks down elastin, an elastic fibre that, together with collagen, determines the mechanical properties of connective tissue. The neutrophil form breaks down the ''Outer membrane protein A'' (OmpA) of '' E. coli'' and other Gram-negative bacteria. Elastase also has the important immunological role of breaking down Shigella virulence factors. This is accomplished through the cleavage of peptide bonds in the target proteins. The specific peptide bonds cleaved are those on the carboxyl side of small, hydrophobic amino acids such as glycine, alanine, and valine. For more on how this is accomplished, see serine p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cofactor (biochemistry)
A cofactor is a non-protein chemical compound or metallic ion that is required for an enzyme's role as a catalyst (a catalyst is a substance that increases the rate of a chemical reaction). Cofactors can be considered "helper molecules" that assist in biochemical transformations. The rates at which these happen are characterized in an area of study called enzyme kinetics. Cofactors typically differ from ligands in that they often derive their function by remaining bound. Cofactors can be divided into two types: inorganic ions and complex organic molecules called coenzymes. Coenzymes are mostly derived from vitamins and other organic essential nutrients in small amounts. (Note that some scientists limit the use of the term "cofactor" for inorganic substances; both types are included here.) Coenzymes are further divided into two types. The first is called a "prosthetic group", which consists of a coenzyme that is tightly (or even covalently) and permanently bound to a protein. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme Inhibitor
An enzyme inhibitor is a molecule that binds to an enzyme and blocks its activity. Enzymes are proteins that speed up chemical reactions necessary for life, in which substrate molecules are converted into products. An enzyme facilitates a specific chemical reaction by binding the substrate to its active site, a specialized area on the enzyme that accelerates the most difficult step of the reaction. An enzyme inhibitor stops ("inhibits") this process, either by binding to the enzyme's active site (thus preventing the substrate itself from binding) or by binding to another site on the enzyme such that the enzyme's catalysis of the reaction is blocked. Enzyme inhibitors may bind reversibly or irreversibly. Irreversible inhibitors form a chemical bond with the enzyme such that the enzyme is inhibited until the chemical bond is broken. By contrast, reversible inhibitors bind non-covalently and may spontaneously leave the enzyme, allowing the enzyme to resume its function. Reve ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elafin
Elafin, also known as peptidase inhibitor 3 or skin-derived antileukoprotease (SKALP), is a protein that in humans is encoded by the ''PI3'' gene. Function This gene encodes an elastase-specific protease inhibitor, which contains a WAP-type four-disulfide core (WFDC) domain, and is thus a member of the WFDC domain family. Most WFDC gene members are localized to chromosome 20 Chromosome 20 is one of the 23 pairs of chromosomes in humans. Chromosome 20 spans around 66 million base pairs (the building material of DNA) and represents between 2 and 2.5 percent of the total DNA in cells. Chromosome 20 was fully sequenced i ...q12-q13 in two clusters: centromeric and telomeric. This gene belongs to the centromeric cluster. Clinical significance Elafin has been found to have utility in serving as a biomarker for graft versus host disease of the skin. Elafin plays some role in gut inflammation. References Further reading * * * * * * * * * * * * * * * * * * External link ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Substrate Specificity
Chemical specificity is the ability of binding site of a macromolecule (such as a protein) to bind specific ligands. The fewer ligands a protein can bind, the greater its specificity. Specificity describes the strength of binding between a given protein and ligand. This relationship can be described by a dissociation constant, which characterizes the balance between bound and unbound states for the protein-ligand system. In the context of a single enzyme and a pair of binding molecules, the two ligands can be compared as stronger or weaker ligands (for the enzyme) on the basis of their dissociation constants. (A lower value corresponds to a stronger binding.) Specificity for a set of ligands is unrelated to the ability of an enzyme to catalyze a given reaction, with the ligand as a substrate. If a given enzyme has a high chemical specificity, this means that the set of ligands to which it binds is limited, such that neither binding events nor catalysis can occur at an appreciab ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymorphism (biology)
In biology, polymorphism is the occurrence of two or more clearly different morphs or forms, also referred to as alternative ''phenotypes'', in the population of a species. To be classified as such, morphs must occupy the same habitat at the same time and belong to a panmictic population (one with random mating). Ford E.B. 1965. ''Genetic polymorphism''. Faber & Faber, London. Put simply, polymorphism is when there are two or more possibilities of a trait on a gene. For example, there is more than one possible trait in terms of a jaguar's skin colouring; they can be light morph or dark morph. Due to having more than one possible variation for this gene, it is termed 'polymorphism'. However, if the jaguar has only one possible trait for that gene, it would be termed "monomorphic". For example, if there was only one possible skin colour that a jaguar could have, it would be termed monomorphic. The term polyphenism can be used to clarify that the different forms arise from the s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
KRT6C
Keratin 6C (protein name K6C; gene name ''KRT6C''), is a type II cytokeratin, one of a number of isoforms of keratin 6 encoded by separate genes located within the type II keratin gene cluster on human chromosome 12q. This gene was uncovered recently by the Human Genome Project and its expression patterns in humans remains unknown. Keratins Keratins are the intermediate filament proteins that form a dense meshwork of filaments throughout the cytoplasm of epithelial cells. Keratins form heteropolymers consisting of a type I and a type II keratin. Keratins are generally expressed in particular pairs of type I and type II keratin proteins in a tissue-specific and cellular differentiation-specific manner. The keratin proteins of epithelial tissues are commonly known as "keratins" or are sometimes referred to as "epithelial keratins" or "cytokeratins". The specialized keratins of hair and nail are known as "hard keratins" or " trichocyte keratins". Trichocytes are the specialized ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diffuse Nonepidermolytic Palmoplantar Keratoderma
Palmoplantar keratodermas are a heterogeneous group of disorders characterized by abnormal thickening of the stratum corneum of the palms and soles. Autosomal recessive, dominant, X-linked, and acquired forms have all been described. Types Clinically, three distinct patterns of palmoplantar keratoderma may be identified: diffuse, focal, and punctate. Diffuse Diffuse palmoplantar keratoderma is a type of palmoplantar keratoderma that is characterized by an even, thick, symmetric hyperkeratosis over the whole of the palm and sole, usually evident at birth or in the first few months of life. Restated, diffuse palmoplantar keratoderma is an autosomal dominant disorder in which hyperkeratosis is confined to the palms and soles. The two major types can have a similar clinical appearance: *''Diffuse epidermolytic palmoplantar keratoderma'' (also known as "Palmoplantar keratoderma cum degeneratione granulosa Vörner," "Vörner's epidermolytic palmoplantar keratoderma", and "V� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha-2-macroglobulin
α2-Macroglobulin (α2M), or alpha-2-macroglobulin, is a large (720 KDa) plasma protein found in the blood. It is mainly produced by the liver, and also locally synthesized by macrophages, fibroblasts, and adrenocortical cells. In humans it is encoded by the ''A2M'' gene. α2-Macroglobulin acts as an antiprotease and is able to inactivate an enormous variety of proteinases. It functions as an inhibitor of fibrinolysis by inhibiting plasmin and kallikrein. It functions as an inhibitor of coagulation by inhibiting thrombin. α2-macroglobulin may act as a carrier protein because it also binds to numerous growth factors and cytokines, such as platelet-derived growth factor, basic fibroblast growth factor, TGF-β, insulin, and IL-1β. No specific deficiency with associated disease has been recognized, and no disease state is attributed to low concentrations of α2-macroglobulin. The concentration of α2-macroglobulin rises 10-fold or more in the nephrotic syndrome when other lower mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alpha 1-antitrypsin
Alpha-1 antitrypsin or α1-antitrypsin (A1AT, α1AT, A1A, or AAT) is a protein belonging to the serpin superfamily. It is encoded in humans by the ''SERPINA1'' gene. A protease inhibitor, it is also known as alpha1–proteinase inhibitor (A1PI) or alpha1-antiproteinase (A1AP) because it inhibits various proteases (not just trypsin). In older biomedical literature it was sometimes called serum trypsin inhibitor (STI, dated terminology), because its capability as a trypsin inhibitor was a salient feature of its early study. As a type of enzyme inhibitor, it protects tissues from enzymes of inflammatory cells, especially neutrophil elastase, and has a reference range in blood of 0.9–2.3 g/L (in the US the reference range is expressed as mg/dL or micromoles), but the concentration can rise manyfold upon acute inflammation. When the blood contains inadequate amounts of A1AT or functionally defective A1AT (such as in alpha-1 antitrypsin deficiency), neutrophil elastase is excessi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SLPI
Antileukoproteinase, also known as secretory leukocyte protease inhibitor (SLPI), is an enzyme that in humans is encoded by the ''SLPI'' gene. SLPI is a highly cationic single-chain protein with eight intramolecular disulfide bonds. It is found in large quantities in bronchial, cervical, and nasal mucosa, saliva, and seminal fluids. SLPI inhibits human leukocyte elastase, human cathepsin G, human trypsin, neutrophil elastase, and mast cell chymase. X-ray crystallography has shown that SLPI has two homologous domains of 53 and 54 amino acids, one of which exhibits anti-protease activity (C-terminal domain). The other domain (N-terminal domain) is not known to have any function. Function This gene encodes a secreted inhibitor which protects epithelial tissues from serine proteases. It is found in various secretions including seminal plasma, cervical mucus, and bronchial secretions, and has affinity for trypsin, leukocyte elastase, and cathepsin G. Its inhibitory effect contribut ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Denaturation (biochemistry)
In biochemistry, denaturation is a process in which proteins or nucleic acids lose the quaternary structure, tertiary structure, and secondary structure which is present in their native state, by application of some external stress or compound such as a strong acid or base, a concentrated inorganic salt, an organic solvent (e.g., alcohol or chloroform), agitation and radiation or heat. If proteins in a living cell are denatured, this results in disruption of cell activity and possibly cell death. Protein denaturation is also a consequence of cell death. Denatured proteins can exhibit a wide range of characteristics, from conformational change and loss of solubility to aggregation due to the exposure of hydrophobic groups. The loss of solubility as a result of denaturation is called ''coagulation''. Denatured proteins lose their 3D structure and therefore cannot function. Protein folding is key to whether a globular or membrane protein can do its job correctly; it must be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


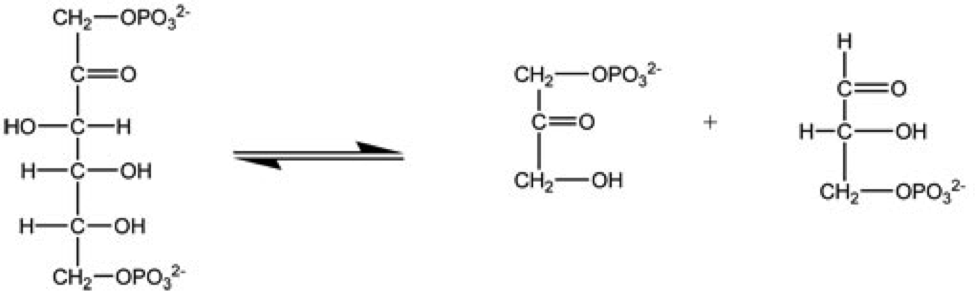

.png)