|
Potassium Sulfide
Potassium sulfide is an inorganic compound with the formula K2 S. The colourless solid is rarely encountered, because it reacts readily with water, a reaction that affords potassium hydrosulfide (KSH) and potassium hydroxide (KOH). Most commonly, the term potassium sulfide refers loosely to this mixture, not the anhydrous solid. Structure It adopts "antifluorite structure," which means that the small K+ ions occupy the tetrahedral (F−) sites in fluorite, and the larger S2− centers occupy the eight-coordinate sites. Li2S, Na2S, and Rb2S crystallize similarly.Holleman, A. F.; Wiberg, E. "Inorganic Chemistry" Academic Press: San Diego, 2001. . Synthesis and reactions It can be produced by heating K2SO4 with carbon ( coke): :K2SO4 + 4 C → K2S + 4 CO In the laboratory, pure K2S may be prepared by the reaction of potassium and sulfur in anhydrous ammonia. Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 360. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Sulfide
Hydrogen sulfide is a chemical compound with the formula . It is a colorless chalcogen-hydride gas, and is poisonous, corrosive, and flammable, with trace amounts in ambient atmosphere having a characteristic foul odor of rotten eggs. The underground mine gas term for foul-smelling hydrogen sulfide-rich gas mixtures is ''stinkdamp''. Swedish chemist Carl Wilhelm Scheele is credited with having discovered the chemical composition of purified hydrogen sulfide in 1777. The British English spelling of this compound is hydrogen sulphide, a spelling no longer recommended by the Royal Society of Chemistry or the International Union of Pure and Applied Chemistry. Hydrogen sulfide is toxic to humans and most other animals by inhibiting cellular respiration in a manner similar to hydrogen cyanide. When it is inhaled or it or its salts are ingested in high amounts, damage to organs occurs rapidly with symptoms ranging from breathing difficulties to convulsions and death. Despite this, the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glitter
Glitter is an assortment of small, reflective particles that come in a variety of shapes, sizes, and colors. Glitter particles reflect light at different angles, causing the surface to sparkle or shimmer. Glitter is similar to confetti, sparkles and sequins, but somewhat smaller. Since prehistoric times, glitter has been made from many different materials including stones such as malachite, and mica, as well as insects and glass. Modern glitter is usually manufactured from the combination of aluminum and plastic which is rarely recycled leading some scientists to call for bans on plastic glitter. Antiquity Glittering surfaces have been found to be used since prehistoric times in the arts and in cosmetics. The modern English word "glitter" comes from the Middle English word ''gliteren'', possibly by way of the Old Norse word '' glitra.'' However, as early as 30,000 years ago, mica flakes were used to give cave paintings a glittering appearance. Prehistoric humans are believed ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Senko Hanabi
Senko hanabi ( 線香花火 ''senkō hanabi'' ) (sparkler - literally: incense-stick fireworks) is a traditional Japanese firework. Essays about them date back to at least 1927. They are a thin shaft of twisted tissue paper about 20 centimeters long with one end containing a few grains of a black gunpowder. The black powder composition consists of three basic chemicals: potassium nitrate, sulfur, and charcoal. The pointed end is lit and held straight down, so that the flame is at the bottom. After a few seconds a glowing, molten slag will form. This is reportedly potassium sulfide, which contains carbon from the charcoal. The molten ball will ignite the second phase of the senko hanabi, silently spraying an array of delicate branching sparks with a range of up to . They are ignited away from the wind and held with a steady hand, so that the delicate molten head does not drop and that the two phases of ignition are completed. Senko hanabi are included in packets of fireworks and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Black Powder
Gunpowder, also commonly known as black powder to distinguish it from modern smokeless powder, is the earliest known chemical explosive. It consists of a mixture of sulfur, carbon (in the form of charcoal) and potassium nitrate (saltpeter). The sulfur and carbon act as fuels while the saltpeter is an oxidizer. Gunpowder has been widely used as a propellant in firearms, artillery, rocketry, and pyrotechnics, including use as a blasting agent for explosives in quarrying, mining, building pipelines and road building. Gunpowder is classified as a low explosive because of its relatively slow decomposition rate and consequently low brisance. Low explosives deflagrate (i.e., burn at subsonic speeds), whereas high explosives detonate, producing a supersonic shockwave. Ignition of gunpowder packed behind a projectile generates enough pressure to force the shot from the muzzle at high speed, but usually not enough force to rupture the gun barrel. It thus makes a good propellant but is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the substance and w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coke (fuel)
Coke is a grey, hard, and porous coal-based fuel with a high carbon content and few impurities, made by heating coal or oil in the absence of air—a destructive distillation process. It is an important industrial product, used mainly in iron ore smelting, but also as a fuel in stoves and forges when air pollution is a concern. The unqualified term "coke" usually refers to the product derived from low-ash and low-sulphur bituminous coal by a process called coking. A similar product called petroleum coke, or pet coke, is obtained from crude oil in oil refineries. Coke may also be formed naturally by geologic processes.B. Kwiecińska and H. I. Petersen (2004): "Graphite, semi-graphite, natural coke, and natural char classification — ICCP system". ''International Journal of Coal Geology'', volume 57, issue 2, pages 99-116. History China Historical sources dating to the 4th century describe the production of coke in ancient China. The Chinese first used coke for heating ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbothermic Reaction
Carbothermic reactions involve the reduction of substances, often metal oxides (O^2-), using carbon as the reducing agent. These chemical reactions are usually conducted at temperatures of several hundred degrees Celsius. Such processes are applied for production of the elemental forms of many elements. The ability of metals to participate in carbothermic reactions can be predicted from Ellingham diagrams. Carbothermal reactions produce carbon monoxide and sometimes carbon dioxide. The facility of these conversions is attributable to the entropy of reaction: two solids, the metal oxide and carbon, are converted to a new solid (metal) and a gas (CO), the latter having high entropy. Applications A prominent example is that of iron ore smelting. Many reactions are involved, but the simplified equation is usually shown as: : 2 + 3C → 4Fe + 3 On a more modest scale, about 1 million tons of elemental phosphorus is produced annually by carbothermic reactions. Calcium phosphate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Sulfide
Sodium sulfide is a chemical compound with the formula Na2 S, or more commonly its hydrate Na2S·9 H2O. Both the anhydrous and the hydrated salts in pure crystalline form are colorless solids, although technical grades of sodium sulfide are generally yellow to brick red owing to the presence of polysulfides and commonly supplied as a crystalline mass, in flake form, or as a fused solid. They are water-soluble, giving strongly alkaline solutions. When exposed to moist air, Na2S and its hydrates emit hydrogen sulfide, an extremely toxic, flammable and corrosive gas which smells like rotten eggs. Some commercial samples are specified as Na2S·''x''H2O, where a weight percentage of Na2S is specified. Commonly available grades have around 60% Na2S by weight, which means that ''x'' is around 3. These grades of sodium sulfide are often marketed as 'sodium sulfide flakes'. Structure Na2S adopts the antifluorite structure, which means that the Na+ centers occupy sites of the fluoride in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium Sulfide
Lithium sulfide is the inorganic compound with the formula Li2 S. It crystallizes in the antifluorite motif, described as the salt (Li+)2S2−. It forms a solid yellow-white deliquescent powder. In air, it easily hydrolyses to release hydrogen sulfide (rotten egg odor). Preparation Lithium sulfide is prepared by treating lithium with sulfur. This reaction is conveniently conducted in anhydrous ammonia. :2 Li + S → Li2S The THF-soluble triethylborane adduct of lithium sulfide can be generated using superhydride Lithium triethylborohydride is the organoboron compound with the formula Li Et3 BH. Commonly referred to as LiTEBH or Superhydride, it is a powerful reducing agent used in organometallic and organic chemistry. It is a colorless or white liquid bu .... Reactions and applications Lithium sulfide has been considered for use in lithium–sulfur batteries. References External links Lithium Sulfide {{Sulfides Lithium salts Sulfides Fluorite crystal structure [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
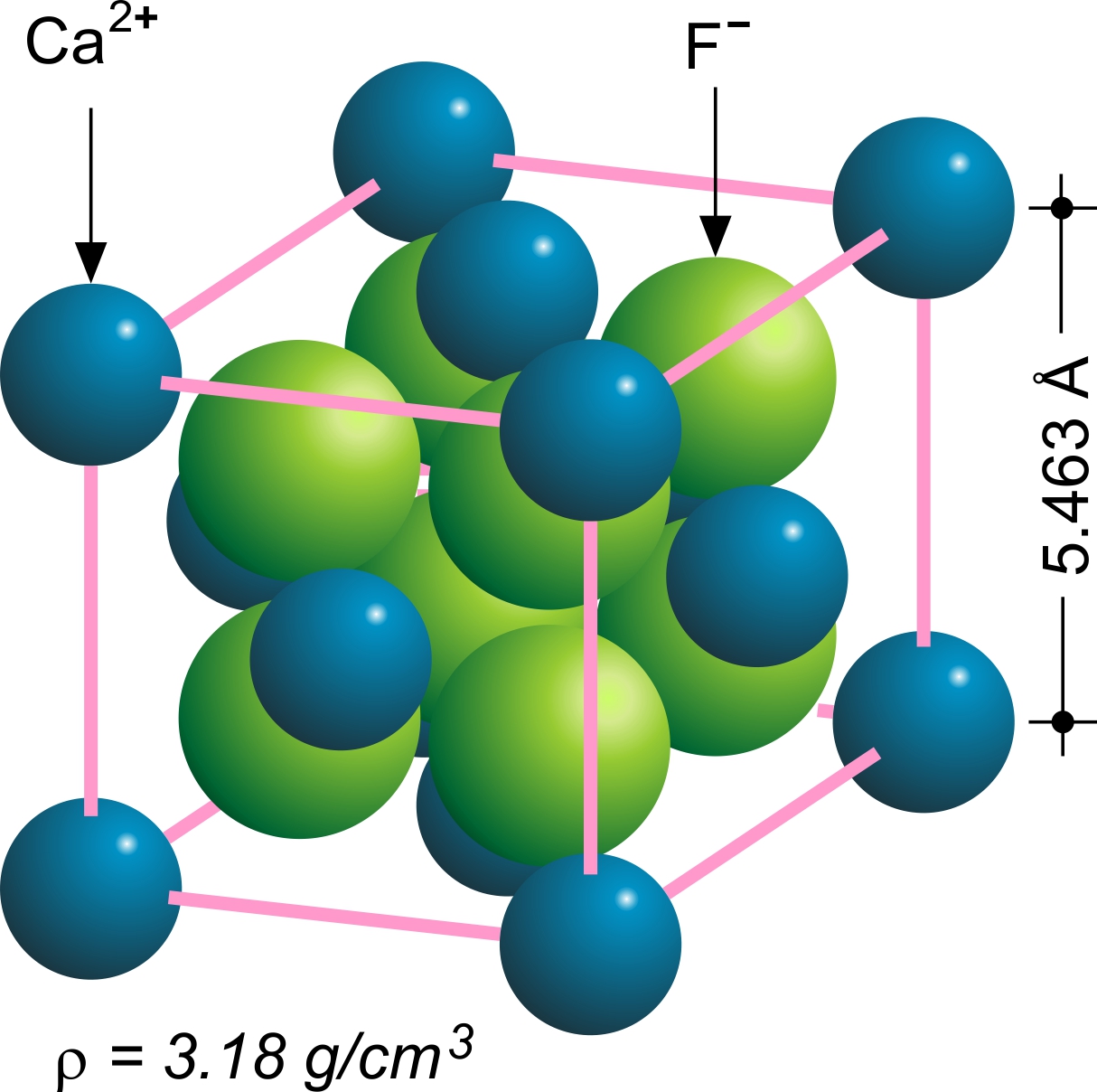
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon. The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite. Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite lenses have low dispersion, so lenses made from it exhibit less chromatic aberration, making them valuable in microscopes and telescopes. Fluorite optics are also ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Potassium Hydrosulfide
Potassium hydrosulfide is the inorganic compound with the formula KSH. This colourless salt consists of the cation and the bisulfide anion . It is the product of the half-neutralization of hydrogen sulfide with potassium hydroxide. The compound is used in the synthesis of some organosulfur compounds. It is prepared by neutralizing aqueous KOH with . Aqueous solutions of potassium sulfide consist of a mixture of potassium hydrosulfide and potassium hydroxide. The structure of the potassium hydrosulfide resembles that for potassium chloride. Their structure is however complicated by the non-spherical symmetry of the anions, but these tumble rapidly in the solid at high temperatures. Addition of sulfur gives dipotassium pentasulfide. Synthesis Potassium hydrosulfide is synthesized by the reaction between a solution of potassium sulfide with excess hydrogen sulfide Hydrogen sulfide is a chemical compound with the formula . It is a colorless chalcogen-hydride gas, and is poiso ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |