|
Photodegradation
Photodegradation is the alteration of materials by light. Commonly, the term is used loosely to refer to the combined action of sunlight and air, which cause oxidation and hydrolysis. Often photodegradation is intentionally avoided, since it destroys paintings and other artifacts. It is, however, partly responsible for remineralization of biomass and is used intentionally in some disinfection technologies. Photodegradation does not apply to how materials may be aged or degraded via infrared light or heat, but does include degradation in all of the ultraviolet light wavebands. Applications Foodstuffs The protection of food from photodegradation is very important. Some nutrients, for example, are affected by degradation when exposed to sunlight. In the case of beer, UV radiation causes a process that entails the degradation of hop bitter compounds to 3-methyl-2-buten-1-thiol and therefore changes the taste. As amber-colored glass has the ability to absorb UV radiation, beer bottles ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Singlet Oxygen
Singlet oxygen, systematically named dioxygen(singlet) and dioxidene, is a gaseous inorganic chemical with the formula O=O (also written as or ), which is in a quantum state where all electrons are spin paired. It is kinetically unstable at ambient temperature, but the rate of decay is slow. The lowest excited state of the diatomic oxygen molecule is a singlet state. It is a gas with physical properties differing only subtly from those of the more prevalent triplet ground state of O2. In terms of its chemical reactivity, however, singlet oxygen is far more reactive toward organic compounds. It is responsible for the photodegradation of many materials but can be put to constructive use in preparative organic chemistry and photodynamic therapy. Trace amounts of singlet oxygen are found in the upper atmosphere and also in polluted urban atmospheres where it contributes to the formation of lung-damaging nitrogen dioxide. It often appears and coexists confounded in environments ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sunlight
Sunlight is a portion of the electromagnetic radiation given off by the Sun, in particular infrared, visible, and ultraviolet light. On Earth, sunlight is scattered and filtered through Earth's atmosphere, and is obvious as daylight when the Sun is above the horizon. When direct solar radiation is not blocked by clouds, it is experienced as sunshine, a combination of bright light and radiant heat. When blocked by clouds or reflected off other objects, sunlight is diffused. Sources estimate a global average of between 164 watts to 340 watts per square meter over a 24-hour day; this figure is estimated by NASA to be about a quarter of Earth's average total solar irradiance. The ultraviolet radiation in sunlight has both positive and negative health effects, as it is both a requisite for vitamin D3 synthesis and a mutagen. Sunlight takes about 8.3 minutes to reach Earth from the surface of the Sun. A photon starting at the center of the Sun and changing direction eve ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Free Radicals
In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron. With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spontaneously dimerize. Most organic radicals have short lifetimes. A notable example of a radical is the hydroxyl radical (HO·), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (꞉) which have two unpaired electrons. Radicals may be generated in a number of ways, but typical methods involve redox reactions. Ionizing radiation, heat, electrical discharges, and electrolysis are known to produce radicals. Radicals are intermediates in many chemical reactions, more so than is apparent from the balanced equations. Radicals are important in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. A majority of n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxidize
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. There are two classes of redox reactions: * ''Electron-transfer'' – Only one (usually) electron flows from the reducing agent to the oxidant. This type of redox reaction is often discussed in terms of redox couples and electrode potentials. * ''Atom transfer'' – An atom transfers from one substrate to another. For example, in the rusting of iron, the oxidation state of iron atoms increases as the iron converts to an oxide, and simultaneously the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with the formation of oxides, other chemical species can serve the same function. In hydrogenation, C=C (and other) bonds ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Excited State
In quantum mechanics, an excited state of a system (such as an atom, molecule or nucleus) is any quantum state of the system that has a higher energy than the ground state (that is, more energy than the absolute minimum). Excitation refers to an increase in energy level above a chosen starting point, usually the ground state, but sometimes an already excited state. The temperature of a group of particles is indicative of the level of excitation (with the notable exception of systems that exhibit negative temperature). The lifetime of a system in an excited state is usually short: spontaneous or induced emission of a quantum of energy (such as a photon or a phonon) usually occurs shortly after the system is promoted to the excited state, returning the system to a state with lower energy (a less excited state or the ground state). This return to a lower energy level is often loosely described as decay and is the inverse of excitation. Long-lived excited states are often called ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ground State
The ground state of a quantum-mechanical system is its stationary state of lowest energy; the energy of the ground state is known as the zero-point energy of the system. An excited state is any state with energy greater than the ground state. In quantum field theory, the ground state is usually called the vacuum state or the vacuum. If more than one ground state exists, they are said to be degenerate. Many systems have degenerate ground states. Degeneracy occurs whenever there exists a unitary operator that acts non-trivially on a ground state and commutes with the Hamiltonian of the system. According to the third law of thermodynamics, a system at absolute zero temperature exists in its ground state; thus, its entropy is determined by the degeneracy of the ground state. Many systems, such as a perfect crystal lattice, have a unique ground state and therefore have zero entropy at absolute zero. It is also possible for the highest excited state to have absolute zero temper ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
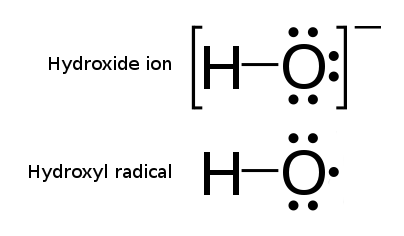
Hydroxyl Radical
The hydroxyl radical is the diatomic molecule . The hydroxyl radical is very stable as a dilute gas, but it decays very rapidly in the condensed phase. It is pervasive in some situations. Most notably the hydroxyl radicals are produced from the decomposition of hydroperoxides (ROOH) or, in atmospheric chemistry, by the reaction of excited atomic oxygen with water. It is also important in the field of radiation chemistry, since it leads to the formation of hydrogen peroxide and oxygen, which can enhance corrosion and SCC in coolant systems subjected to radioactive environments. In organic synthesis, hydroxyl radicals are most commonly generated by photolysis of 1-hydroxy-2(1''H'')-pyridinethione. Notation The unpaired electron of the hydroxyl radical is officially represented by a middle dot, •, beside the O. Biology Hydroxyl radicals can occasionally be produced as a byproduct of immune action. Macrophages and microglia most frequently generate this compound when exp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kevlar
Kevlar (para-aramid) is a strong, heat-resistant synthetic fiber, related to other aramids such as Nomex and Technora. Developed by Stephanie Kwolek at DuPont in 1965, the high-strength material was first used commercially in the early 1970s as a replacement for steel in racing tires. It is typically spun into ropes or fabric sheets that can be used as such, or as an ingredient in composite material components. Kevlar has many applications, ranging from bicycle tires and racing sails to bulletproof vests, all due to its high tensile strength-to-weight ratio; by this measure it is five times stronger than steel. It is also used to make modern marching drumheads that withstand high impact; and for mooring lines and other underwater applications. A similar fiber called Twaron with the same chemical structure was developed by Akzo in the 1970s; commercial production started in 1986, and Twaron is now manufactured by Teijin. History Poly-paraphenylene terephthalamide (K2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aramid
Aramid fibers, short for aromatic polyamide, are a class of heat-resistant and strong synthetic fibers. They are used in aerospace and military applications, for ballistic-rated body armor fabric and ballistic composites, in marine cordage, marine hull reinforcement, and as an asbestos substitute. The chain molecules in the fibers are highly oriented along the fiber axis. As a result, a higher proportion of the chemical bond contributes more to fiber strength than in many other synthetic fibers. Aramids have a very high melting point (>500 °C). Common aramid brand names include Kevlar, Nomex, and Twaron. Terminology and chemical structure ''Aramid'' is a shortened form of aromatic polyamide. The term was introduced in 1972, accepted in 1974 by the Federal Trade Commission of the USA as the name of a generic category of fiber distinct from nylon, and adopted by the International Standards Organisation in 1977. Aromatic in the name refers to the presence of aromatic rings ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aromatic Ring
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to saturated compounds having single bonds, and other geometric or connective non-cyclic arrangements with the same set of atoms. Aromatic rings are very stable and do not break apart easily. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have enhanced stability. The term ''aromaticity'' with this meaning is historically related to the concept of having an aroma, but is a distinct property from that meaning. Since the most common aromatic compounds are derivatives of benzene (an aromatic hydrocarbon common in petroleum and its distillates), the word ''aromatic'' occasionally refers informally to benzene derivatives, and so it was first defined. Nevertheless, many non- ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Abrasion (mechanical)
Abrasion is the process of scuffing, scratching, wearing down, marring, or rubbing away. It can be intentionally imposed in a controlled process using an abrasive. Abrasion can be an undesirable effect of exposure to normal use or exposure to the elements. In stone shaping Ancient artists, working in stone, used abrasion to create sculptures. The artist selected dense stones like carbonite and emery and rubbed them consistently against comparatively softer stones like limestone and granite. The artist used different sizes and shapes of abrasives, or turned them in various ways as they rubbed, to create effects on the softer stone's surface. Water was continuously poured over the surface to carry away particles. Abrasive technique in stone shaping was a long, tedious process that, with patience, resulted in eternal works of art in stone. Models The Archard equation is a simple model used to describe sliding wear and is based on the theory of asperity contact. Q = \frac H whe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |






.png)
