|
Phenolate
Phenolates (also called phenoxides) are anions, salts, and esters of phenols. They may be formed by reaction of phenols with strong base. Properties Alkali metal phenolates, such as sodium phenolate hydrolyze in aqueous solution to form basic solutions. At pH = 10, phenol and phenolate are in approximately 1:1 proportions. Phenolate anions are enolates. As such, they react as nucleophiles at both oxygen and carbon positions. In general, reaction at oxygen occurs under kinetic control, whereas reaction at carbon occurs under thermodynamic control. Uses Alkyl aryl ethers can be synthesized through the Williamson ether synthesis by treating sodium phenolate with an alkyl halide: :C6H5ONa + CH3I → C6H5OCH3 + NaI :C6H5ONa + (CH3O)2SO2 → C6H5OCH3 + (CH3O)SO3Na Production of salicylic acid Salicylic acid is produced in the Kolbe–Schmitt reaction between carbon dioxide and sodium phenolate. : See also * Sodium phenolate Sodium phenoxide (sodium phenolate) is an organ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenolate
Phenolates (also called phenoxides) are anions, salts, and esters of phenols. They may be formed by reaction of phenols with strong base. Properties Alkali metal phenolates, such as sodium phenolate hydrolyze in aqueous solution to form basic solutions. At pH = 10, phenol and phenolate are in approximately 1:1 proportions. Phenolate anions are enolates. As such, they react as nucleophiles at both oxygen and carbon positions. In general, reaction at oxygen occurs under kinetic control, whereas reaction at carbon occurs under thermodynamic control. Uses Alkyl aryl ethers can be synthesized through the Williamson ether synthesis by treating sodium phenolate with an alkyl halide: :C6H5ONa + CH3I → C6H5OCH3 + NaI :C6H5ONa + (CH3O)2SO2 → C6H5OCH3 + (CH3O)SO3Na Production of salicylic acid Salicylic acid is produced in the Kolbe–Schmitt reaction between carbon dioxide and sodium phenolate. : See also * Sodium phenolate Sodium phenoxide (sodium phenolate) is an organ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Phenoxide
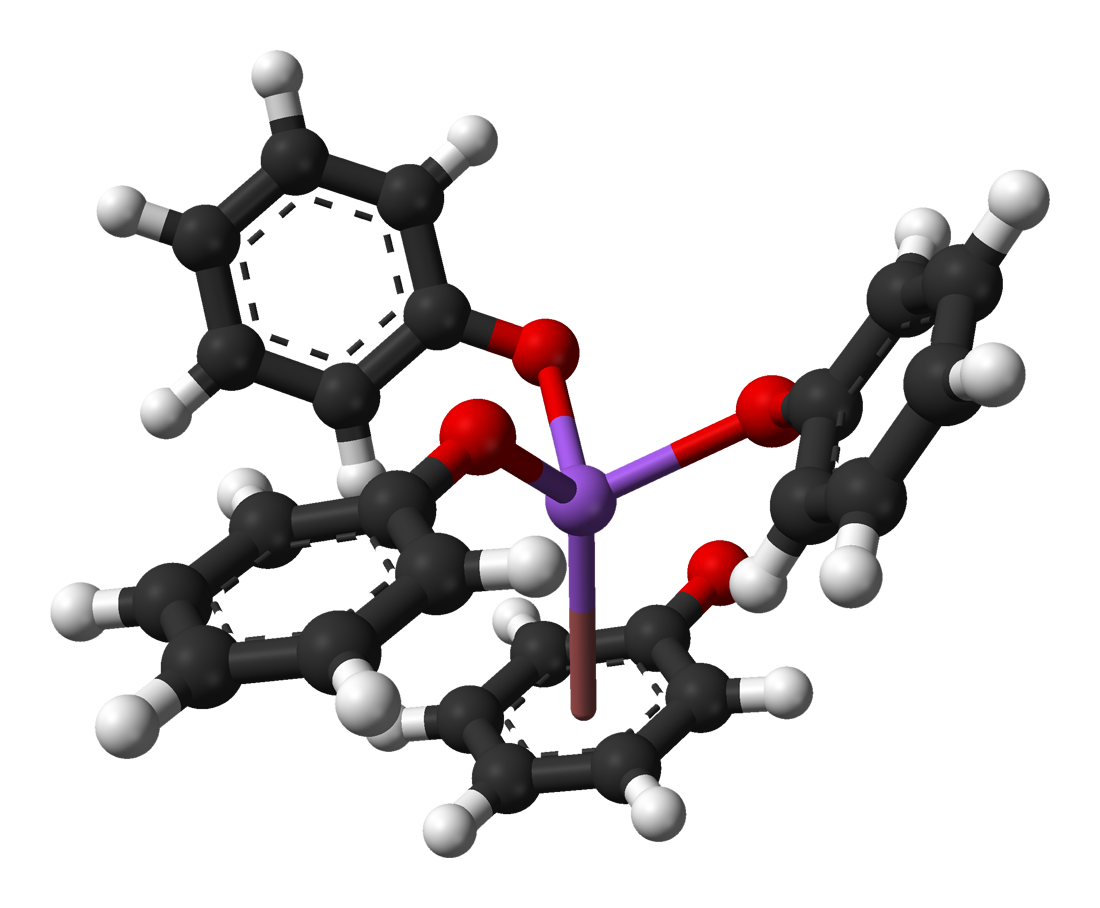
Sodium phenoxide (sodium phenolate) is an organic compound with the formula NaOC6H5. It is a white crystalline solid. Its anion, phenoxide, also known as phenolate, is the conjugate base of phenol. It is used as a precursor to many other organic compounds, such as aryl ethers. Synthesis and structure Most commonly, solutions of sodium phenoxide are produced by treating phenol with sodium hydroxide. Anhydrous derivatives can be prepared by combining phenol and sodium. A related, updated procedure uses sodium methoxide instead of sodium hydroxide: :NaOCH3 + HOC6H5 → NaOC6H5 + HOCH3 Sodium phenoxide can also be produced by the "alkaline fusion" of benzenesulfonic acid, whereby the sulfonate groups are displaced by hydroxide: :C6H5SO3Na + 2 NaOH → C6H5ONa + Na2SO3 This route once was the principal industrial route to phenol. Structure Like other sodium alkoxides, solid sodium phenoxide adopts a complex structure involving multiple Na-O bonds. Solvent-free material i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Phenolate
Sodium phenoxide (sodium phenolate) is an organic compound with the formula NaOC6H5. It is a white crystalline solid. Its anion, phenoxide, also known as phenolate, is the conjugate base of phenol. It is used as a precursor to many other organic compounds, such as aryl ethers. Synthesis and structure Most commonly, solutions of sodium phenoxide are produced by treating phenol with sodium hydroxide. Anhydrous derivatives can be prepared by combining phenol and sodium. A related, updated procedure uses sodium methoxide instead of sodium hydroxide: :NaOCH3 + HOC6H5 → NaOC6H5 + HOCH3 Sodium phenoxide can also be produced by the "alkaline fusion" of benzenesulfonic acid, whereby the sulfonate groups are displaced by hydroxide: :C6H5SO3Na + 2 NaOH → C6H5ONa + Na2SO3 This route once was the principal industrial route to phenol. Structure Like other sodium alkoxides, solid sodium phenoxide adopts a complex structure involving multiple Na-O bonds. Solvent-free material i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons. A cation is a positively charged ion with fewer electrons than protons while an anion is a negatively charged ion with more electrons than protons. Opposite electric charges are pulled towards one another by electrostatic force, so cations and anions attract each other and readily form ionic compounds. Ions consisting of only a single atom are termed atomic or monatomic ions, while two or more atoms form molecular ions or polyatomic ions. In the case of physical ionization in a fluid (gas or liquid), "ion pairs" are created by spontaneous molecule collisions, where each generated pair consists of a free electron and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Salt (chemistry)
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively charged sodium ions and negatively charged chloride ions. The component ions in a salt compound can be either inorganic, such as chloride (Cl−), or organic, such as acetate (). Each ion can be either monatomic, such as fluoride (F−), or polyatomic, such as sulfate (). Types of salt Salts can be classified in a variety of ways. Salts that produce hydroxide ions when dissolved in water are called ''alkali salts'' and salts that produce hydrogen ions when dissolved in water are called ''acid salts''. ''Neutral salts'' are those salts that are neither acidic nor basic. Zwitterions contain an anionic and a cationic centre in the same molecule, but are not considered salts. Examples of zwitterions are amino acids, many metabolites, peptid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phenols
In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of one or more hydroxyl groups (— O H) bonded directly to an aromatic hydrocarbon group. The simplest is phenol, . Phenolic compounds are classified as simple phenols or polyphenols based on the number of phenol units in the molecule. Phenols are both synthesized industrially and produced by plants and microorganisms. Properties Acidity Phenols are more acidic than typical alcohols. The acidity of the hydroxyl group in phenols is commonly intermediate between that of aliphatic alcohols and carboxylic acids (their pKa is usually between 10 and 12). Deprotonation of a phenol forms a corresponding negative phenolate ion or phenoxide ion, and the corresponding salts are called phenolates or phenoxides (aryloxides according to the IUPAC Gold Book). Condensation with aldehydes and ketones Phenols are susceptible to Electrophilic aromatic substitutions. Condensation with formald ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enolate
In organic chemistry, enolates are organic anions derived from the deprotonation of carbonyl () compounds. Rarely isolated, they are widely used as reagents in the synthesis of organic compounds. Bonding and structure Enolate anions are electronically related to allyl anions. The anionic charge is delocalized over the oxygen and the two carbon sites. Thus they have the character of both an alkoxide and a carbanion. Although they are often drawn as being simple salts, in fact they adopt complicated structures often featuring aggregates. Preparation Deprotonation of enolizable ketones, aromatic alcohols, aldehydes, and esters gives enolates. With strong bases, the deprotonation is quantitative. Typically enolates are generated from using lithium diisopropylamide (LDA). Often, as in conventional Claisen condensations, Mannich reactions, and aldol condensations, enolates are generated in low concentrations with alkoxide bases. Under such conditions, they exist in low concent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleophile
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are Lewis bases. ''Nucleophilic'' describes the affinity of a nucleophile to bond with positively charged atomic nuclei. Nucleophilicity, sometimes referred to as nucleophile strength, refers to a substance's nucleophilic character and is often used to compare the affinity of atoms. Neutral nucleophilic reactions with solvents such as alcohols and water are named solvolysis. Nucleophiles may take part in nucleophilic substitution, whereby a nucleophile becomes attracted to a full or partial positive charge, and nucleophilic addition. Nucleophilicity is closely related to basicity. History The terms ''nucleophile'' and ''electrophile'' were introduced by Christopher Kelk Ingold in 1933, replacing the terms ''anionoid'' and ''cationoid' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kinetic Control
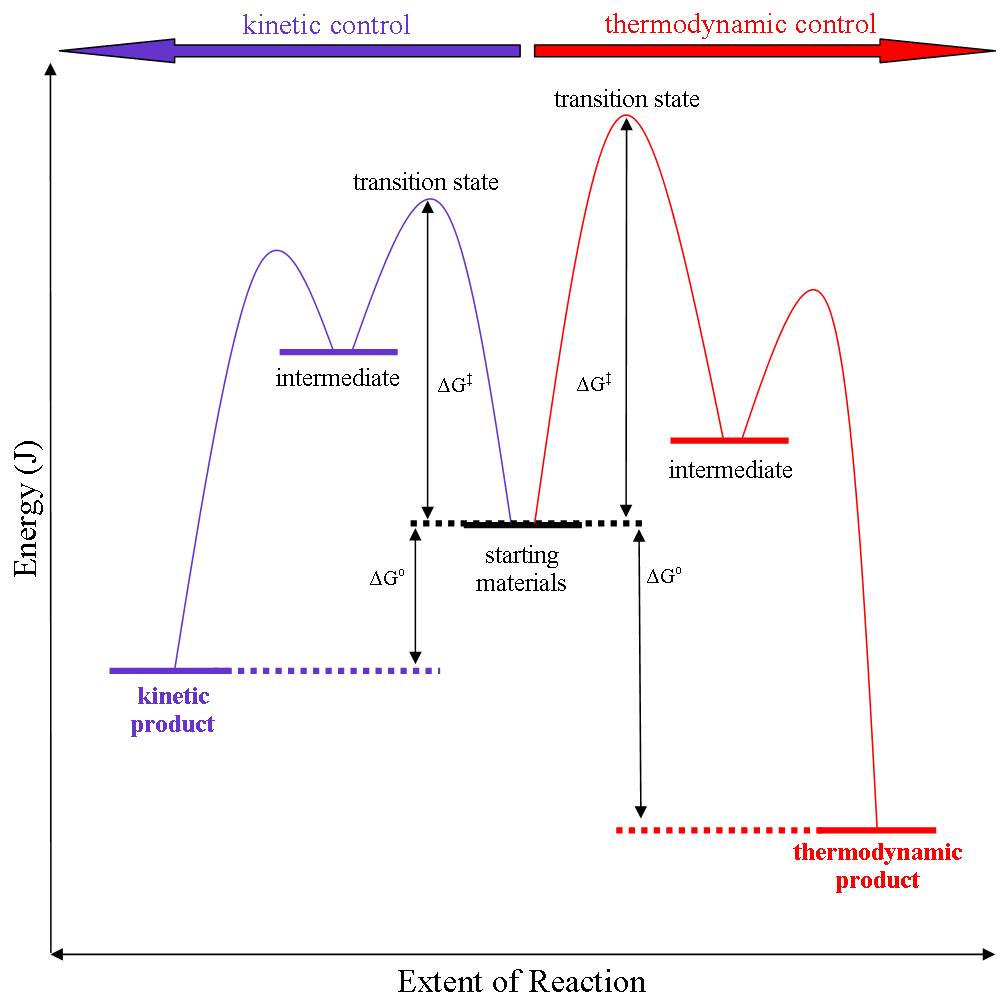
Thermodynamic reaction control or kinetic reaction control in a chemical reaction can decide the composition in a reaction product mixture when competing pathways lead to different products and the reaction conditions influence the selectivity or stereoselectivity. The distinction is relevant when product A forms faster than product B because the activation energy for product A is lower than that for product B, yet product B is more stable. In such a case A is the kinetic product and is favoured under kinetic control and B is the thermodynamic product and is favoured under thermodynamic control.Introduction to Organic Chemistry I, Seth Robert Elsheimer, Blackwell Publishing, 2000 The conditions of the reaction, such as temperature, pressure, or solvent, affect which reaction pathway may be favored: either the kinetically controlled or the thermodynamically controlled one. Note this is only true if the activation energy of the two pathways differ, with one pathway having a lower '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Williamson Ether Synthesis
The Williamson ether synthesis is an organic reaction, forming an ether from an organohalide and a deprotonated alcohol ( alkoxide). This reaction was developed by Alexander Williamson in 1850. Typically it involves the reaction of an alkoxide ion with a primary alkyl halide via an SN2 reaction. This reaction is important in the history of organic chemistry because it helped prove the structure of ethers. The general reaction mechanism is as follows: An example is the reaction of sodium ethoxide with chloroethane to form diethyl ether and sodium chloride: :C2H5Cl + C2H5ONa -> C2H5OC2H5 + NaCl Mechanism The Williamson ether reaction follows an SN2 bimolecular nucleophilic substitution mechanism. In an SN2 reaction mechanism there is a backside attack of an electrophile by a nucleophile and it occurs in a concerted mechanism (happens all at once). In order for the SN2 reaction to take place there must be a good leaving group which is strongly electronegative, commonly a hali ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Haloalkane
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes that contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kolbe–Schmitt Reaction
The Kolbe–Schmitt reaction or Kolbe process (named after Hermann Kolbe and Rudolf Schmitt) is a carboxylation chemical reaction that proceeds by heating sodium phenoxide (the sodium salt of phenol) with carbon dioxide under pressure (100 atm, 125 °C), then treating the product with sulfuric acid. The final product is an aromatic hydroxy acid which is also known as salicylic acid (the precursor to aspirin). 500px, center, The Kolbe–Schmitt reaction By using potassium hydroxide, 4-hydroxybenzoic acid is accessible, an important precursor for the versatile paraben class of biocides used e.g. in personal care products. The methodology is also used in the industrial synthesis of 3-hydroxy-2-naphthoic acid; the regiochemistry of the carboxylation in this case is sensitive to temperature.. Reaction mechanism The Kolbe–Schmitt reaction proceeds via the nucleophilic addition of a phenoxide, classically sodium phenoxide (NaOC6H5), to carbon dioxide to give the salicylate. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |