|
Pharmacological Activity
In pharmacology, biological activity or pharmacological activity describes the beneficial or adverse effects of a drug on living matter. When a drug is a complex chemical mixture, this activity is exerted by the substance's active ingredient or pharmacophore but can be modified by the other constituents. Among the various properties of chemical compounds, pharmacological/biological activity plays a crucial role since it suggests uses of the compounds in the medical applications. However, chemical compounds may show some adverse and toxic effects which may prevent their use in medical practice. Activity is generally dosage-dependent. Further, it is common to have effects ranging from beneficial to adverse for one substance when going from low to high doses. Activity depends critically on fulfillment of the ADME criteria. To be an effective drug, a compound not only must be active against a target, but also possess the appropriate ADME (Absorption, Distribution, Metabolism, and E ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pharmacology
Pharmacology is a branch of medicine, biology and pharmaceutical sciences concerned with drug or medication action, where a drug may be defined as any artificial, natural, or endogenous (from within the body) molecule which exerts a biochemical or physiological effect on the cell, tissue, organ, or organism (sometimes the word ''pharmacon'' is used as a term to encompass these endogenous and exogenous bioactive species). More specifically, it is the study of the interactions that occur between a living organism and chemicals that affect normal or abnormal biochemical function. If substances have medicinal properties, they are considered pharmaceuticals. The field encompasses drug composition and properties,functions,sources,synthesis and drug design, molecular and cellular mechanisms, organ/systems mechanisms, signal transduction/cellular communication, molecular diagnostics, interactions, chemical biology, therapy, and medical applications and antipathogenic capabili ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Toxicity
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a substructure of the organism, such as a cell ( cytotoxicity) or an organ such as the liver (hepatotoxicity). By extension, the word may be metaphorically used to describe toxic effects on larger and more complex groups, such as the family unit or society at large. Sometimes the word is more or less synonymous with poisoning in everyday usage. A central concept of toxicology is that the effects of a toxicant are dose-dependent; even water can lead to water intoxication when taken in too high a dose, whereas for even a very toxic substance such as snake venom there is a dose below which there is no detectable toxic effect. Toxicity is species-specific, making cross-species analysis problematic. Newer paradigms and metrics are evolving to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Physical Property
A physical property is any Property (philosophy), property that is Measurement, measurable, whose value describes a state of a physical system. The changes in the physical properties of a system can be used to describe its changes between momentary states. Physical properties are often referred to as observables. They are not modal properties. A Quantity, quantifiable physical property is called physical quantity. Physical properties are often characterized as intensive and extensive properties. An intensive property does not depend on the size or extent of the system, nor on the amount of matter in the object, while an extensive property shows an additive relationship. These classifications are in general only valid in cases when smaller subdivisions of the sample do not interact in some physical or chemical process when combined. Properties may also be classified with respect to the directionality of their nature. For example, isotropy, isotropic properties do not change with t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Property
Molecular properties include the chemical properties, physical properties, and structural properties of molecules, including drugs. Molecular properties typically do not include pharmacological or biological properties of a chemical compound. See also * Biological activity * Chemical property * Chemical structure * Lipinski's rule of five, describing molecular properties of drugs * Physical property A physical property is any Property (philosophy), property that is Measurement, measurable, whose value describes a state of a physical system. The changes in the physical properties of a system can be used to describe its changes between momenta ... * QSAR, quantitative structure-activity relationship References Physical quantities Molecules {{chem-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lipinski's Rule Of Five
Lipinski's rule of five, also known as Pfizer's rule of five or simply the rule of five (RO5), is a rule of thumb to evaluate druglikeness or determine if a chemical compound with a certain pharmacological or biological activity has chemical properties and physical properties that would make it a likely orally active drug in humans. The rule was formulated by Christopher A. Lipinski in 1997, based on the observation that most orally administered drugs are relatively small and moderately lipophilic molecules. The rule describes molecular properties important for a drug's pharmacokinetics in the human body, including their absorption, distribution, metabolism, and excretion ("ADME"). However, the rule does not predict if a compound is pharmacologically active. The rule is important to keep in mind during drug discovery when a pharmacologically active lead structure is optimized step-wise to increase the activity and selectivity of the compound as well as to ensure drug-like physi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Structure
A chemical structure determination includes a chemist's specifying the molecular geometry and, when feasible and necessary, the electronic structure of the target molecule or other solid. Molecular geometry refers to the spatial arrangement of atoms in a molecule and the chemical bonds that hold the atoms together, and can be represented using structural formulae and by molecular models; complete electronic structure descriptions include specifying the occupation of a molecule's molecular orbitals. Structure determination can be applied to a range of targets from very simple molecules (e.g., diatomic oxygen or nitrogen), to very complex ones (e.g., such as protein or DNA). Background Theories of chemical structure were first developed by August Kekulé, Archibald Scott Couper, and Aleksandr Butlerov, among others, from about 1858. These theories were first to state that chemical compounds are not a random cluster of atoms and functional groups, but rather had a definite o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Property
A chemical property is any of a material's properties that becomes evident during, or after, a chemical reaction; that is, any quality that can be established only by changing a substance's chemical identity.William L. Masterton, Cecile N. Hurley, "Chemistry: Principles and Reactions", 6th edition. Brooks/Cole Cengage Learning, 2009, p.1(Google books)/ref> Simply speaking, chemical properties cannot be determined just by viewing or touching the substance; the substance's internal structure must be affected greatly for its chemical properties to be investigated. When a substance goes under a chemical reaction, the properties will change drastically, resulting in chemical change. However, a catalytic property would also be a chemical property. Chemical properties can be contrasted with physical properties, which can be discerned without changing the substance's structure. However, for many properties within the scope of physical chemistry, and other disciplines at the boundary b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
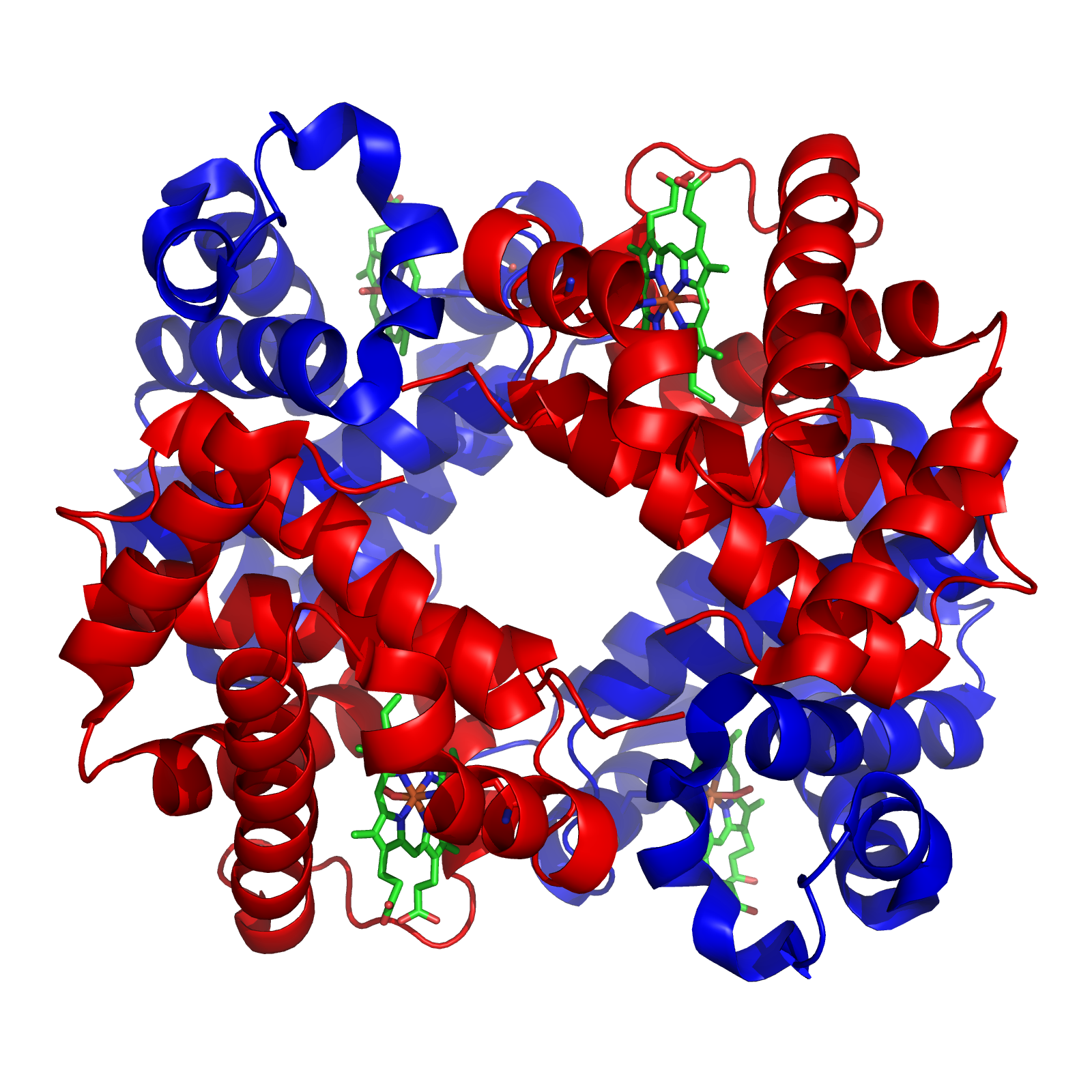
Blood
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells. Blood in the circulatory system is also known as ''peripheral blood'', and the blood cells it carries, ''peripheral blood cells''. Blood is composed of blood cells suspended in blood plasma. Plasma, which constitutes 55% of blood fluid, is mostly water (92% by volume), and contains proteins, glucose, mineral ions, hormones, carbon dioxide (plasma being the main medium for excretory product transportation), and blood cells themselves. Albumin is the main protein in plasma, and it functions to regulate the colloidal osmotic pressure of blood. The blood cells are mainly red blood cells (also called RBCs or erythrocytes), white blood cells (also called WBCs or leukocytes) and platelets (also called thrombocytes). The most abundant cells in verte ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Buffer Solution
A buffer solution (more precisely, pH buffer or hydrogen ion buffer) is an aqueous solution consisting of a mixture of a weak acid and its conjugate base, or vice versa. Its pH changes very little when a small amount of strong acid or base is added to it. Buffer solutions are used as a means of keeping pH at a nearly constant value in a wide variety of chemical applications. In nature, there are many living systems that use buffering for pH regulation. For example, the bicarbonate buffering system is used to regulate the pH of blood, and bicarbonate also acts as a buffer in the ocean. Principles of buffering Buffer solutions resist pH change because of a chemical equilibrium between the weak acid HA and its conjugate base A−: When some strong acid is added to an equilibrium mixture of the weak acid and its conjugate base, hydrogen ions (H+) are added, and the equilibrium is shifted to the left, in accordance with Le Chatelier's principle. Because of this, the hydroge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
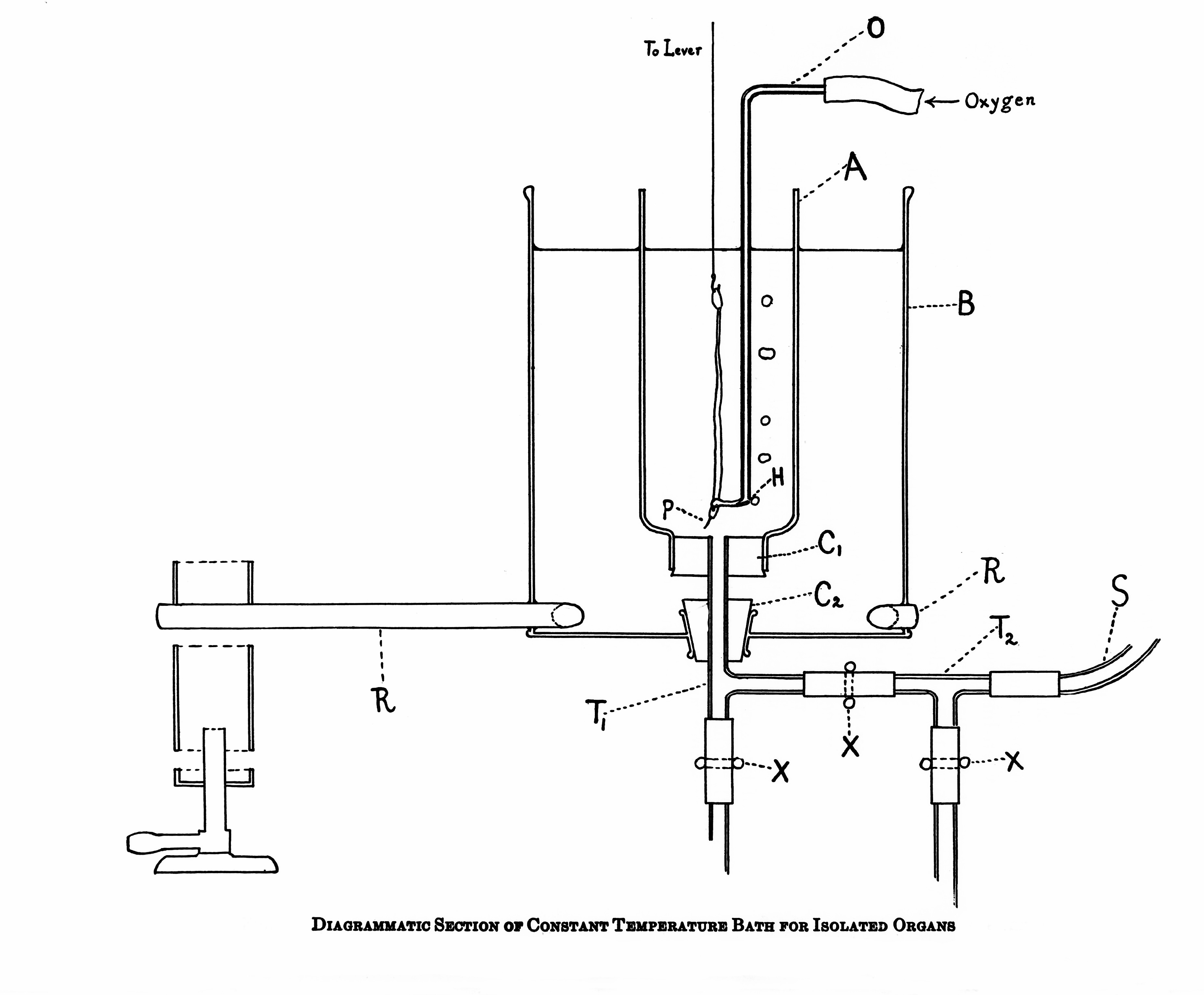
Simulated Body Fluid
A simulated body fluid (SBF) is a solution with an ion concentration close to that of human blood plasma, kept under mild conditions of pH and identical physiological temperature. SBF was first introduced by Kokubo et al. in order to evaluate the changes on a surface of a bioactive glass ceramic. Later, cell culture media (such as DMEM, MEM, α-MEM, etc.), in combination with some methodologies adopted in cell culture, were proposed as an alternative to conventional SBF in assessing the bioactivity of materials. Applications Surface modification of metallic implants For an artificial material to bond to living bone, the formation of bonelike apatite layer on the surface of an implant is of significant importance. The SBF can be used as an in vitro testing method to study the formation of apatite layer on the surface of implants so as to predict their in vivo bone bioactivity. The consumption of calcium and phosphate ions, present in the SBF solution, results in the spontaneous g ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
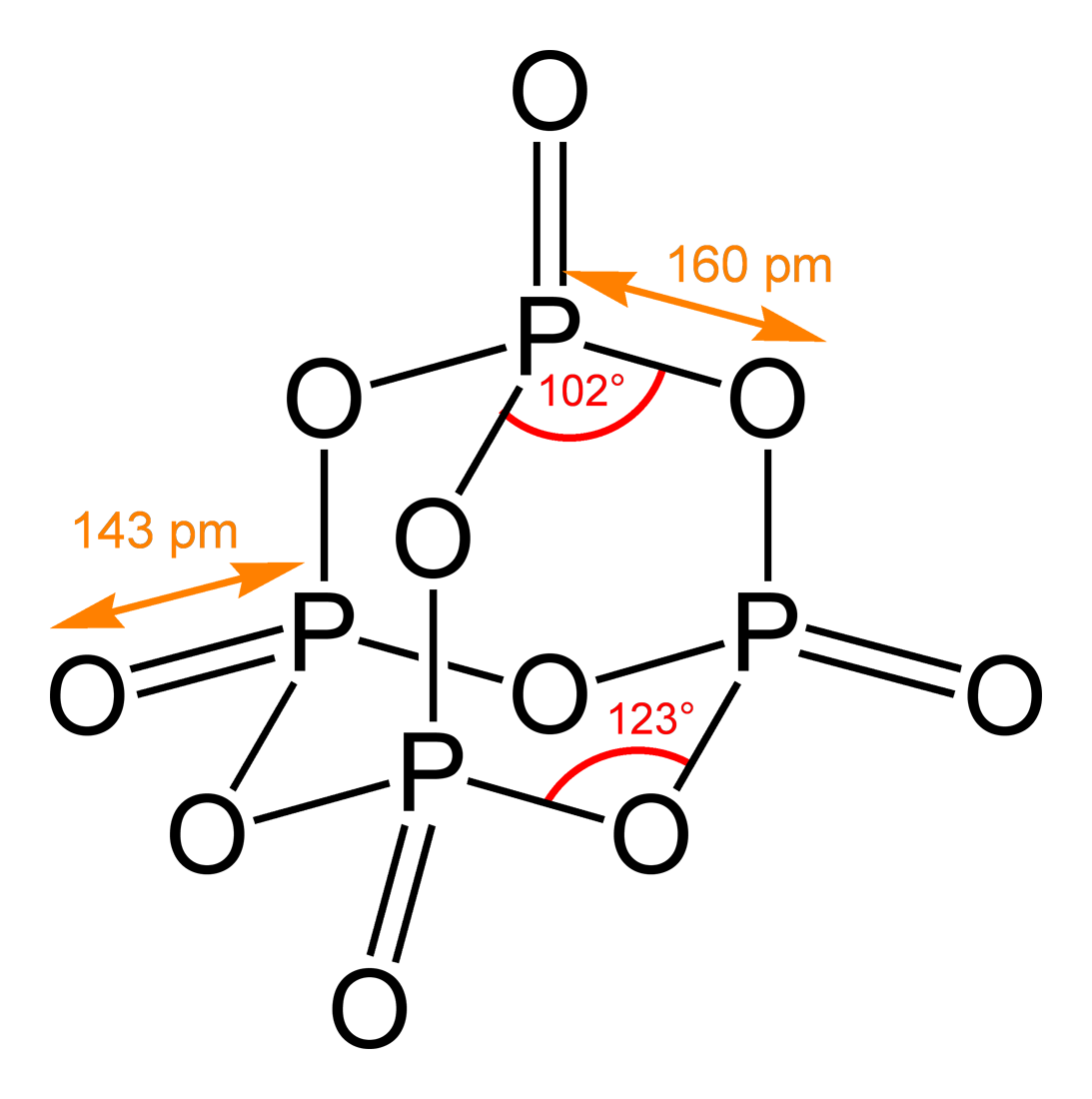
Calcium Phosphate
The term calcium phosphate refers to a family of materials and minerals containing calcium ions (Ca2+) together with inorganic phosphate anions. Some so-called calcium phosphates contain oxide and hydroxide as well. Calcium phosphates are white solids of nutritious value and are found in many living organisms, e.g., bone mineral and tooth enamel. In milk, it exists in a colloidal form in micelles bound to casein protein with magnesium, zinc, and citrate–collectively referred to as colloidal calcium phosphate (CCP). Various calcium phosphate minerals are used in the production of phosphoric acid and fertilizers. Overuse of certain forms of calcium phosphate can lead to nutrient-containing surface runoff and subsequent adverse effects upon receiving waters such as algal blooms and eutrophication (over-enrichment with nutrients and minerals). Orthophosphates, di- and monohydrogen phosphates These materials contain Ca2+ combined with , , or : * Monocalcium phosphate, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |