|
Parietal Cells, Gastric
Parietal cells (also known as oxyntic cells) are epithelial cells in the stomach that secrete hydrochloric acid (HCl) and intrinsic factor. These cells are located in the gastric glands found in the lining of the fundus and body regions of the stomach. They contain an extensive secretory network of canaliculi from which the HCl is secreted by active transport into the stomach. The enzyme hydrogen potassium ATPase (H+/K+ ATPase) is unique to the parietal cells and transports the H+ against a concentration gradient of about 3 million to 1, which is the steepest ion gradient formed in the human body. Parietal cells are primarily regulated via histamine, acetylcholine and gastrin signalling from both central and local modulators. Structure Canaliculus A canaliculus is an adaptation found on gastric parietal cells. It is a deep infolding, or little channel, which serves to increase the surface area, e.g. for secretion. The parietal cell membrane is dynamic; the numbers ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
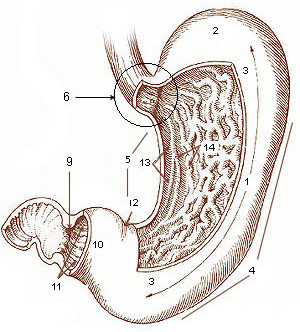
Stomach
The stomach is a muscular, hollow organ in the gastrointestinal tract of humans and many other animals, including several invertebrates. The stomach has a dilated structure and functions as a vital organ in the digestive system. The stomach is involved in the gastric phase of digestion, following chewing. It performs a chemical breakdown by means of enzymes and hydrochloric acid. In humans and many other animals, the stomach is located between the oesophagus and the small intestine. The stomach secretes digestive enzymes and gastric acid to aid in food digestion. The pyloric sphincter controls the passage of partially digested food ( chyme) from the stomach into the duodenum, where peristalsis takes over to move this through the rest of intestines. Structure In the human digestive system, the stomach lies between the oesophagus and the duodenum (the first part of the small intestine). It is in the left upper quadrant of the abdominal cavity. The top of the stomach lies ag ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon Dioxide
Carbon dioxide (chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is transparent to visible light but absorbs infrared radiation, acting as a greenhouse gas. It is a trace gas in Earth's atmosphere at 421 parts per million (ppm), or about 0.04% by volume (as of May 2022), having risen from pre-industrial levels of 280 ppm. Burning fossil fuels is the primary cause of these increased CO2 concentrations and also the primary cause of climate change.IPCC (2022Summary for policy makersiClimate Change 2022: Mitigation of Climate Change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA Carbon dioxide is soluble in water and is found in groundwater, lakes, ice caps, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proteolysis
Proteolysis is the breakdown of proteins into smaller polypeptides or amino acids. Uncatalysed, the hydrolysis of peptide bonds is extremely slow, taking hundreds of years. Proteolysis is typically catalysed by cellular enzymes called proteases, but may also occur by intra-molecular digestion. Proteolysis in organisms serves many purposes; for example, digestive enzymes break down proteins in food to provide amino acids for the organism, while proteolytic processing of a polypeptide chain after its synthesis may be necessary for the production of an active protein. It is also important in the regulation of some physiological and cellular processes including apoptosis, as well as preventing the accumulation of unwanted or misfolded proteins in cells. Consequently, abnormality in the regulation of proteolysis can cause disease. Proteolysis can also be used as an analytical tool for studying proteins in the laboratory, and it may also be used in industry, for example in food proc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pepsin
Pepsin is an endopeptidase that breaks down proteins into smaller peptides. It is produced in the gastric chief cells of the stomach lining and is one of the main digestive enzymes in the digestive systems of humans and many other animals, where it helps digest the proteins in food. Pepsin is an aspartic protease, using a catalytic aspartate in its active site. It is one of three principal endopeptidases (enzymes cutting proteins in the middle) in the human digestive system, the other two being chymotrypsin and trypsin. There are also exopeptidases which remove individual amino acids at both ends of proteins (carboxypeptidases produced by the pancreas and aminopeptidases secreted by the small intestine). During the process of digestion, these enzymes, each of which is specialized in severing links between particular types of amino acids, collaborate to break down dietary proteins into their components, i.e., peptides and amino acids, which can be readily absorbed by the sma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zymogen
In biochemistry, a zymogen (), also called a proenzyme (), is an inactive precursor of an enzyme. A zymogen requires a biochemical change (such as a hydrolysis reaction revealing the active site, or changing the configuration to reveal the active site) for it to become an active enzyme. The biochemical change usually occurs in Golgi bodies, where a specific part of the precursor enzyme is cleaved in order to activate it. The inactivating piece which is cleaved off can be a peptide unit, or can be independently-folding domains comprising more than 100 residues. Although they limit the enzyme's ability, these N-terminal extensions of the enzyme or a “prosegment” often aid in the stabilization and folding of the enzyme they inhibit. The pancreas secretes zymogens partly to prevent the enzymes from digesting proteins in the cells in which they are synthesised. Enzymes like pepsin are created in the form of pepsinogen, an inactive zymogen. Pepsinogen is activated when chief ce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pepsinogen
Pepsin is an endopeptidase that breaks down proteins into smaller peptides. It is produced in the gastric chief cells of the stomach lining and is one of the main digestive enzymes in the digestive systems of humans and many other animals, where it helps digest the proteins in food. Pepsin is an aspartic protease, using a catalytic aspartate in its active site. It is one of three principal endopeptidases (enzymes cutting proteins in the middle) in the human digestive system, the other two being chymotrypsin and trypsin. There are also exopeptidases which remove individual amino acids at both ends of proteins (carboxypeptidases produced by the pancreas and aminopeptidases secreted by the small intestine). During the process of digestion, these enzymes, each of which is specialized in severing links between particular types of amino acids, collaborate to break down dietary proteins into their components, i.e., peptides and amino acids, which can be readily absorbed by the small ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amino Acids
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha amino acids appear in the genetic code. Amino acids can be classified according to the locations of the core structural functional groups, as Alpha and beta carbon, alpha- , beta- , gamma- or delta- amino acids; other categories relate to Chemical polarity, polarity, ionization, and side chain group type (aliphatic, Open-chain compound, acyclic, aromatic, containing hydroxyl or sulfur, etc.). In the form of proteins, amino acid '' residues'' form the second-largest component (water being the largest) of human muscles and other tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as neurotransmitter transport and biosynthesis. It is thought that they played a key role in enabling lif ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
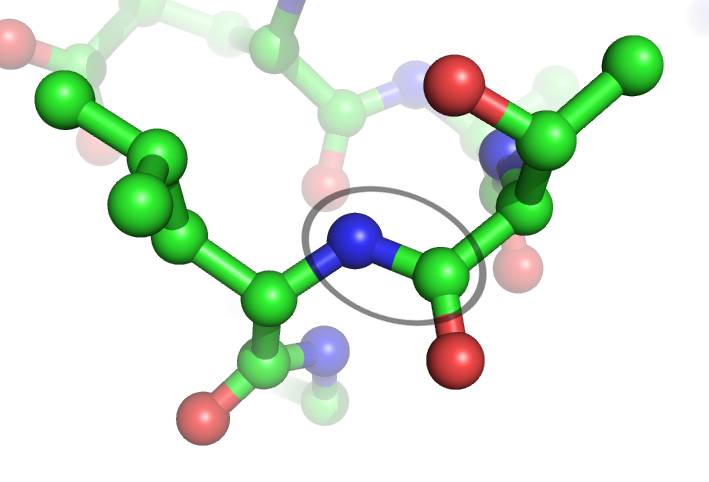
Peptide Bonds
In organic chemistry, a peptide bond is an amide type of covalent chemical bond linking two consecutive alpha-amino acids from C1 (carbon number one) of one alpha-amino acid and N2 (nitrogen number two) of another, along a peptide or protein chain. It can also be called a eupeptide bond to distinguish it from an isopeptide bond, which is another type of amide bond between two amino acids. Synthesis When two amino acids form a ''dipeptide'' through a ''peptide bond'', it is a type of condensation reaction. In this kind of condensation, two amino acids approach each other, with the non-side chain (C1) carboxylic acid moiety of one coming near the non-side chain (N2) amino moiety of the other. One loses a hydrogen and oxygen from its carboxyl group (COOH) and the other loses a hydrogen from its amino group (NH2). This reaction produces a molecule of water (H2O) and two amino acids joined by a peptide bond (−CO−NH−). The two joined amino acids are called a dipeptide. The am ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid residue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Denaturation (biochemistry)
In biochemistry, denaturation is a process in which proteins or nucleic acids lose the quaternary structure, tertiary structure, and secondary structure which is present in their native state, by application of some external stress or compound such as a strong acid or base, a concentrated inorganic salt, an organic solvent (e.g., alcohol or chloroform), agitation and radiation or heat. If proteins in a living cell are denatured, this results in disruption of cell activity and possibly cell death. Protein denaturation is also a consequence of cell death. Denatured proteins can exhibit a wide range of characteristics, from conformational change and loss of solubility to aggregation due to the exposure of hydrophobic groups. The loss of solubility as a result of denaturation is called ''coagulation''. Denatured proteins lose their 3D structure and therefore cannot function. Protein folding is key to whether a globular or membrane protein can do its job correctly; it must be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Parietal Cells
Parietal cells (also known as oxyntic cells) are epithelial cells in the stomach that secrete hydrochloric acid (HCl) and intrinsic factor. These cells are located in the gastric glands found in the lining of the fundus and body regions of the stomach. They contain an extensive secretory network of canaliculi from which the HCl is secreted by active transport into the stomach. The enzyme hydrogen potassium ATPase (H+/K+ ATPase) is unique to the parietal cells and transports the H+ against a concentration gradient of about 3 million to 1, which is the steepest ion gradient formed in the human body. Parietal cells are primarily regulated via histamine, acetylcholine and gastrin signalling from both central and local modulators. Structure Canaliculus A canaliculus is an adaptation found on gastric parietal cells. It is a deep infolding, or little channel, which serves to increase the surface area, e.g. for secretion. The parietal cell membrane is dynamic; the numbers of canalic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Parietal Cell
Parietal cells (also known as oxyntic cells) are epithelial cells in the stomach that secrete hydrochloric acid (HCl) and intrinsic factor. These cells are located in the gastric glands found in the lining of the fundus and body regions of the stomach. They contain an extensive secretory network of canaliculi from which the HCl is secreted by active transport into the stomach. The enzyme hydrogen potassium ATPase (H+/K+ ATPase) is unique to the parietal cells and transports the H+ against a concentration gradient of about 3 million to 1, which is the steepest ion gradient formed in the human body. Parietal cells are primarily regulated via histamine, acetylcholine and gastrin signalling from both central and local modulators. Structure Canaliculus A canaliculus is an adaptation found on gastric parietal cells. It is a deep infolding, or little channel, which serves to increase the surface area, e.g. for secretion. The parietal cell membrane is dynamic; the numbers of canalic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |