|
Mixed Metal Oxide
Mixed metal oxide (MMO) electrodes, also called Dimensionally Stable Anodes (DSA), are devices with high conductivity and corrosion resistance for use as anodes in electrolysis. They are made by coating a substrate, such as pure titanium plate or expanded mesh, with several kinds of metal oxides. One oxide is usually RuO2, IrO2, or PtO2, which conducts electricity and catalyzes the desired reaction such as the production of chlorine gas. The other metal oxide is typically titanium dioxide which does not conduct or catalyze the reaction, but is cheaper and prevents corrosion of the interior. The loading or amount of precious metal on the substrate (that is, other than the titanium) can be in the order of around 10 to 12 grams per square metre. Applications Applications include use as anodes in electrolytic cells for producing free chlorine from saltwater in swimming pools, in electrowinning of metals, in printed circuit board manufacture, electrotinning and zinc electro-galva ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MMO Anode
MMO may refer to: Entertainment * Massively multiplayer online game, a video game that can be played by many people simultaneously * Music Minus One, a record company in Westchester, New York Science and technology * Methane monooxygenase, an enzyme * Maximum operating Mach number * Mercury Magnetospheric Orbiter, a component of the Mercury mapper BepiColombo * Mini-Mag Orion, a proposed type of spacecraft propulsion * Mixed metal oxide, a class of materials often used in electrodes Other * Means, motive, and opportunity, the "indicators of suspicion" * Minimum municipal obligation, minimum contribution to a pension plan * Maio Airport, in Cape Verde, IATA code * Melton Mowbray railway station, England (National Rail station code MMO) * Marine Management Organisation, UK government body * Marine mammal observer, a professional in environmental consulting See also * Military Merit Order (other) {{disambiguation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit (e.g. a semiconductor, an electrolyte, a vacuum or air). Electrodes are essential parts of batteries that can consist of a variety of materials depending on the type of battery. The electrophore, invented by Johan Wilcke, was an early version of an electrode used to study static electricity. Anode and cathode in electrochemical cells Electrodes are an essential part of any battery. The first electrochemical battery made was devised by Alessandro Volta and was aptly named the Voltaic cell. This battery consisted of a stack of copper and zinc electrodes separated by brine-soaked paper disks. Due to fluctuation in the voltage provided by the voltaic cell it wasn't very practical. The first practical battery was invented in 1839 and named the Daniell cell after John Frederic Daniell. Still making use of the zinc–copper electrode combination. Since then many more batteries have be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anode
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic is ACID, for "anode current into device". The direction of conventional current (the flow of positive charges) in a circuit is opposite to the direction of electron flow, so (negatively charged) electrons flow out the anode of a galvanic cell, into an outside or external circuit connected to the cell. For example, the end of a household battery marked with a "-" (minus) is the anode. In both a galvanic cell and an electrolytic cell, the anode is the electrode at which the oxidation reaction occurs. In a galvanic cell the anode is the wire or plate having excess negative charge as a result of the oxidation reaction. In an electrolytic cell, the anode is the wire or plate upon which excess positive charge is imposed. As a result of this, anion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of elements from naturally occurring sources such as ores using an electrolytic cell. The voltage that is needed for electrolysis to occur is called the decomposition potential. The word "lysis" means to separate or break, so in terms, electrolysis would mean "breakdown via electricity". Etymology The word "electrolysis" was introduced by Michael Faraday in 1834, using the Greek words "amber", which since the 17th century was associated with electrical phenomena, and ' meaning "dissolution". Nevertheless, electrolysis, as a tool to study chemical reactions and obtain pure elements, precedes the coinage of the term and formal description by Faraday. History In the early nineteenth century, William Nicholson and Anthony Carlisle sought to further Volt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
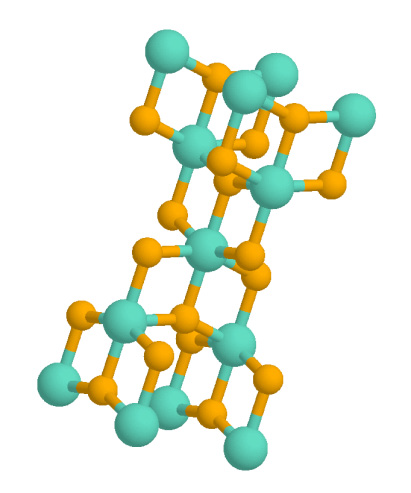
Ruthenium Dioxide
Ruthenium(IV) oxide is the inorganic compound with the formula Ru O2. This black solid is the most common oxide of ruthenium. It is widely used as an electrocatalyst for producing chlorine, chlorine oxides, and O2. Like many dioxides, RuO2 adopts the rutile structure. Preparation It is usually prepared by oxidation of ruthenium trichloride. Nearly stoichiometric single crystals of RuO2 can be obtained by chemical vapor transport, using O2 as the transport agent: :RuO2 + O2 RuO4 Films of RuO2 can be prepared by chemical vapor deposition (CVD) from volatile ruthenium compounds. RuO2 can also be prepared through electroplating from a solution of ruthenium trichloride. Electrostatically stabilized hydrosols of pristine ruthenium dioxide hydrate have been prepared by exploiting the autocatalytic reduction of ruthenium tetroxide in aqueous solution. The resulting particle populations may be controlled to comprise substantially monodisperse, uniform spheres with diameters in th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iridium Dioxide
Iridium(IV) oxide, IrO2, is the only well-characterised oxide of iridium. It is a blue-black solid. The compound adopts the TiO2 rutile structure, featuring six coordinate iridium and three coordinate oxygen. It is used with other rare oxides in the coating of anode-electrodes for industrial electrolysis and in microelectrodes for electrophysiology research. As described by its discoverers, it can be formed by treating the green form of iridium trichloride with oxygen at high temperatures: :2 IrCl3 + 2 O2 → 2 IrO2 + 3 Cl2 A hydrated form is also known. Application Iridium dioxide can be used as an anode electrode for industrial electrolysis and as a microelectrode for electrophysiological studies. Iridium dioxide can be used to make coated electrodes. References Iridium compounds Transition metal oxides {{inorganic-compound-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Platinum Oxide
Adams' catalyst, also known as platinum dioxide, is usually represented as platinum(IV) oxide hydrate, PtO2•H2O. It is a catalyst for hydrogenation and hydrogenolysis in organic synthesis. This dark brown powder is commercially available. The oxide itself is not an active catalyst, but it becomes active after exposure to hydrogen whereupon it converts to platinum black, which is responsible for reactions. Preparation Adams' catalyst is prepared from chloroplatinic acid H2PtCl6 or ammonium chloroplatinate, (NH4)2PtCl6, by fusion with sodium nitrate. The first published preparation was reported by V. Voorhees and Roger Adams. The procedure involves first preparing a platinum nitrate which is then heated to expel nitrogen oxides. :H2PtCl6 + 6 NaNO3 → Pt(NO3)4 + 6 NaCl (aq) + 2 HNO3 :Pt(NO3)4 → PtO2 + 4 NO2 + O2 The resulting brown cake is washed with water to free it from nitrates. The catalyst can either be used as is or dried and stored in a desiccator for later use. Platin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine Production
Chlorine gas can be produced by extracting from natural materials, including the electrolysis of a sodium chloride solution ( brine) and other ways. Gas extraction Chlorine can be manufactured by the electrolysis of a sodium chloride solution ( brine), which is known as the Chloralkali process. The production of chlorine results in the co-products caustic soda ( sodium hydroxide, NaOH) and hydrogen gas (H2). These two products, as well as chlorine itself, are highly reactive. Chlorine can also be produced by the electrolysis of a solution of potassium chloride, in which case the co-products are hydrogen and caustic potash (potassium hydroxide). There are three industrial methods for the extraction of chlorine by electrolysis of chloride solutions, all proceeding according to the following equations: :Cathode: 2 H+ (aq) + 2 e− → H2 (g) :Anode: 2 Cl− (aq) → Cl2 (g) + 2 e− Overall process: 2 NaCl (or KCl) + 2 H2O → Cl2 + H2 + 2 NaOH (or KOH) Mercury cell electrolysi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titanium Dioxide
Titanium dioxide, also known as titanium(IV) oxide or titania , is the inorganic compound with the chemical formula . When used as a pigment, it is called titanium white, Pigment White 6 (PW6), or CI 77891. It is a white solid that is insoluble to water, although mineral forms can appear black. As a pigment, it has a wide range of applications, including paint, sunscreen, and food coloring. When used as a food coloring, it has E number E171. World production in 2014 exceeded 9 million tonnes. It has been estimated that titanium dioxide is used in two-thirds of all pigments, and pigments based on the oxide have been valued at a price of $13.2 billion. Structure In all three of its main dioxides, titanium exhibits octahedral geometry, being bonded to six oxide anions. The oxides in turn are bonded to three Ti centers. The overall crystal structure of rutile is tetragonal in symmetry whereas anatase and brookite are orthorhombic. The oxygen substructures are all slight distort ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engineering is the field dedicated to controlling and preventing corrosion. In the most common use of the word, this means electrochemical oxidation of metal in reaction with an oxidant such as oxygen, hydrogen or hydroxide. Rusting, the formation of iron oxides, is a well-known example of electrochemical corrosion. This type of damage typically produces oxide(s) or salt(s) of the original metal and results in a distinctive orange colouration. Corrosion can also occur in materials other than metals, such as ceramics or polymers, although in this context, the term "degradation" is more common. Corrosion degrades the useful properties of materials and structures including strength, appearance and permeability to liquids and gases. Many structural ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine
Chlorine is a chemical element with the Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a yellow-green gas at room temperature. It is an extremely reactive element and a strong oxidising agent: among the elements, it has the highest electron affinity and the third-highest electronegativity on the revised Electronegativity#Pauling electronegativity, Pauling scale, behind only oxygen and fluorine. Chlorine played an important role in the experiments conducted by medieval Alchemy, alchemists, which commonly involved the heating of chloride Salt (chemistry), salts like ammonium chloride (sal ammoniac) and sodium chloride (common salt), producing various chemical substances containing chlorine such as hydrogen chloride, mercury(II) chloride (corrosive sublimate), and hydrochloric acid (in the form of ). However ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrowinning
Electrowinning, also called electroextraction, is the electrodeposition of metals from their ores that have been put in solution via a process commonly referred to as leaching. Electrorefining uses a similar process to remove impurities from a metal. Both processes use electroplating on a large scale and are important techniques for the economical and straightforward purification of non-ferrous metals. The resulting metals are said to be ''electrowon''. In electrowinning, an electrical current is passed from an inert anode (oxidation, made out of lead (Pb)) through a ''leach'' solution containing the dissolved metal ions so that the metal is recovered as it is deposited in an electroplating process onto the cathode (reduction, stainless steel, aluminium (Al), titanium (Ti)). In electrorefining, the anode consists of the impure metal (e.g., copper) to be refined. The impure metallic anode is oxidized and the metal dissolves into solution. The metal ions migrate through the acidic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |