|
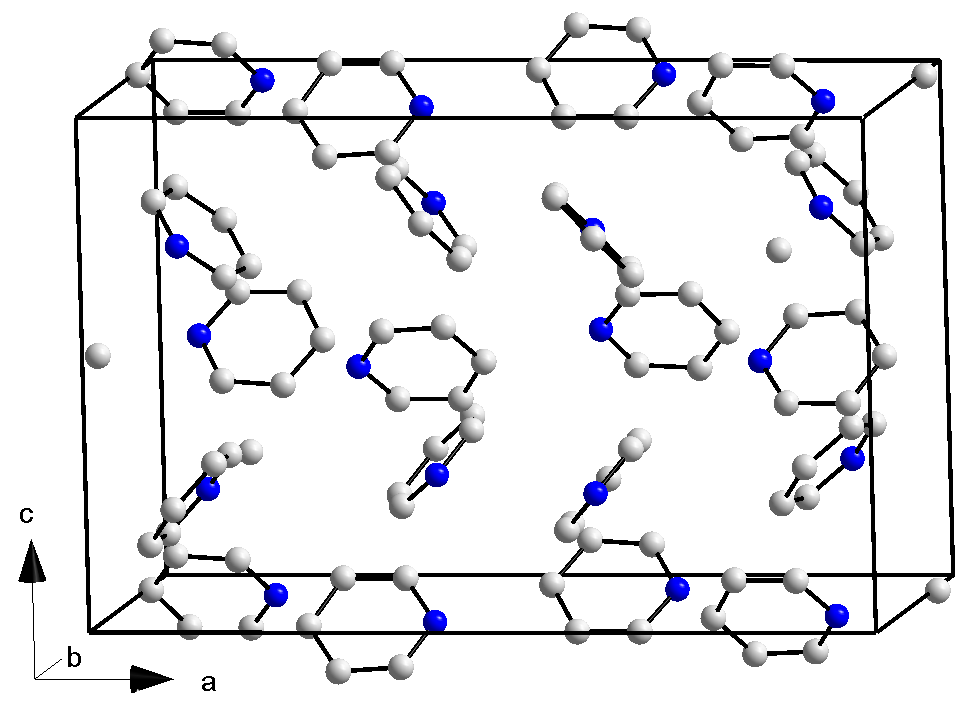
Lutidine
Lutidine is the trivial name used to describe the chemical compounds which are dimethyl derivatives of pyridine Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a .... Their chemical properties resemble those of pyridine, although the presence of the methyl groups may prohibit some of the more straightforward reactions. Lutidine comes in several isomers: * 2,3-Lutidine (2,3-dimethylpyridine) * 2,4-Lutidine (2,4-dimethylpyridine) * 2,5-Lutidine (2,5-dimethylpyridine) * 2,6-Lutidine (2,6-dimethylpyridine) * 3,4-Lutidine (3,4-dimethylpyridine) * 3,5-Lutidine (3,5-dimethylpyridine) All isomers share the molecular weight 107,16 g/mol and the chemical formula C7H9N. {{Authority control Pyridines Amines Amine solvents ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridines
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a distinctive, unpleasant fish-like smell. Pyridine is colorless, but older or impure samples can appear yellow, due to the formation of extended, unsaturated polymeric chains, which show significant electrical conductivity. The pyridine ring occurs in many important compounds, including agrochemicals, pharmaceuticals, and vitamins. Historically, pyridine was produced from coal tar. As of 2016, it is synthesized on the scale of about 20,000 tons per year worldwide. Properties Physical properties The molecular electric dipole moment is 2.2 debyes. Pyridine is diamagnetic and has a diamagnetic susceptibility of −48.7 × 10−6 cm3·mol−1. The standard enthalpy of formation is 100.2 kJ·mol−1 in the liquid phase ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridine
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a distinctive, unpleasant fish-like smell. Pyridine is colorless, but older or impure samples can appear yellow, due to the formation of extended, unsaturated polymeric chains, which show significant electrical conductivity. The pyridine ring occurs in many important compounds, including agrochemicals, pharmaceuticals, and vitamins. Historically, pyridine was produced from coal tar. As of 2016, it is synthesized on the scale of about 20,000 tons per year worldwide. Properties Physical properties The molecular electric dipole moment is 2.2 debyes. Pyridine is diamagnetic and has a diamagnetic susceptibility of −48.7 × 10−6 cm3·mol−1. The standard enthalpy of formation is 100.2 kJ·mol−1 in the liquid phase ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,4-Lutidine
2,4-Lutidine is a heterocyclic organic compound with the formula (CH3)2C5H3N. It is one of several dimethyl-substituted derivatives of pyridine, all of which are referred to as lutidines. It is a colorless liquid with mildly basic properties and a pungent, noxious odor. The compound has few uses. It is produced industrially by extraction from coal tars. Biodegradation The biodegradation of pyridines proceeds via multiple pathways. Although pyridine Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a d ... is an excellent source of carbon, nitrogen, and energy for certain microorganisms, methylation significantly retards degradation of the pyridine ring. Safety The is 200 mg/kg (oral, rats). See also * 2,6-Lutidine * 3,5-Lutidine References {{DEFAULTSORT:Lutidine, 2,4- Pyr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
3,5-Lutidine
3,5-Lutidine is a heterocyclic organic compound with the formula (CH3)2C5H3N. It is one of several dimethyl-substituted derivatives of pyridine, all of which are referred to as lutidines. It is a colorless liquid with mildly basic properties and a pungent odor. The compound is a precursor to the drug omeprazole. 3,5-Lutidine is produced industrially by condensation of acrolein, ammonia, and formaldehyde: :2CH2=CHCHO + CH2O + NH3 → (CH3)2C5H3N + 2 H2O Biodegradation The biodegradation of pyridines proceeds via multiple pathways. Although pyridine is an excellent source of carbon, nitrogen, and energy for certain microorganisms, methylation significantly retards degradation of the pyridine ring. Safety The is 200 mg/kg (oral, rats). See also * 2,6-Lutidine * 2,4-Lutidine 2,4-Lutidine is a heterocyclic organic compound with the formula (CH3)2C5H3N. It is one of several dimethyl-substituted derivatives of pyridine, all of which are referred to as lutidines. It is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trivial Name
In chemistry, a trivial name is a nonsystematic name for a chemical substance. That is, the name is not recognized according to the rules of any formal system of chemical nomenclature such as IUPAC nomenclature of inorganic chemistry, IUPAC inorganic or IUPAC nomenclature of organic chemistry, IUPAC organic nomenclature. A trivial name is not a chemical nomenclature, formal name and is usually a common name. Generally, trivial names are not useful in describing the essential properties of the thing being named. Properties such as the molecular structure of a chemical compound are not indicated. And, in some cases, trivial names can be ambiguous or will carry different meanings in different industries or in different geographic regions (for example, a trivial name such as ''white metal'' can mean various things). Trivial names are simpler. As a result, a limited number of trivial chemical names are retained names, an accepted part of the nomenclature. Trivial names often arise in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
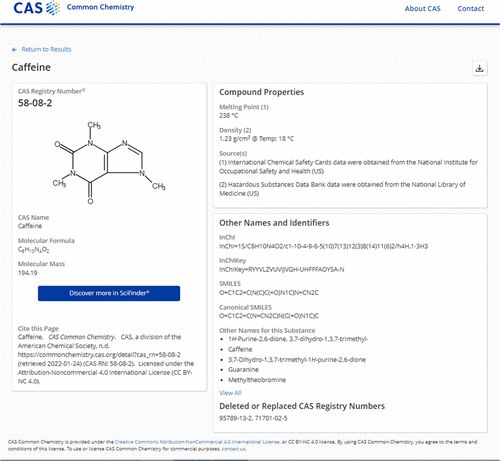
CAS Registry Number
A CAS Registry Number (also referred to as CAS RN or informally CAS Number) is a unique identification number assigned by the Chemical Abstracts Service (CAS), US to every chemical substance described in the open scientific literature. It includes all substances described from 1957 through the present, plus some substances from as far back as the early 1800s. It is a chemical database that includes organic and inorganic compounds, minerals, isotopes, alloys, mixtures, and nonstructurable materials (UVCBs, substances of unknown or variable composition, complex reaction products, or biological origin). CAS RNs are generally serial numbers (with a check digit), so they do not contain any information about the structures themselves the way SMILES and InChI strings do. The registry maintained by CAS is an authoritative collection of disclosed chemical substance information. It identifies more than 182 million unique organic and inorganic substances and 68 million protein and DNA seq ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
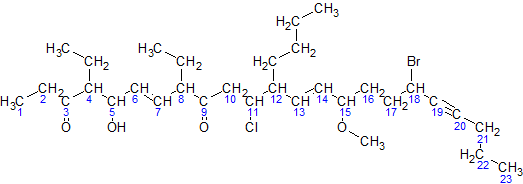
Structural Formula
The structural formula of a chemical compound is a graphic representation of the molecular structure (determined by structural chemistry methods), showing how the atoms are possibly arranged in the real three-dimensional space. The chemical bonding within the molecule is also shown, either explicitly or implicitly. Unlike other chemical formula types, which have a limited number of symbols and are capable of only limited descriptive power, structural formulas provide a more complete geometric representation of the molecular structure. For example, many chemical compounds exist in different isomeric forms, which have different enantiomeric structures but the same molecular formula. There are multiple types of ways to draw these structural formulas such as: Lewis Structures, condensed formulas, skeletal formulas, Newman projections, Cyclohexane conformations, Haworth projections, and Fischer projections. Several systematic chemical naming formats, as in chemical databases, are used ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
IUPAC Nomenclature Of Organic Chemistry
In chemical nomenclature, the IUPAC nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended by the International Union of Pure and Applied Chemistry (IUPAC). It is published in the ''Nomenclature of Organic Chemistry'' (informally called the Blue Book). Ideally, every possible organic compound should have a name from which an unambiguous structural formula can be created. There is also an IUPAC nomenclature of inorganic chemistry. To avoid long and tedious names in normal communication, the official IUPAC naming recommendations are not always followed in practice, except when it is necessary to give an unambiguous and absolute definition to a compound. IUPAC names can sometimes be simpler than older names, as with ethanol, instead of ethyl alcohol. For relatively simple molecules they can be more easily understood than non-systematic names, which must be learnt or looked over. However, the common or trivial name is often substantially ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. In this process, bonds between atoms may be broken and/or new bonds formed. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. Molecular compounds are held together by covalent bonds; ionic compounds are held together by ionic bonds; intermetallic compounds are held together by metallic bonds; coordination complexes are held together by coordinate covalent bonds. Non-stoichiometric compounds form a disputed marginal case. A chemical formula specifies the number of atoms of each element in a compound molecule, using the s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |