|
Linear Ubiquitin Chain Assembly Complex
Linear ubiquitin chain assembly complex (LUBAC) is a multi-protein complex and the only known E3 ubiquitin ligase able to conjugate ubiquitin in a head-to-tail manner to generate linear (M1-linked) polyubiquitin chains. The complex is currently known to be composed of three proteins: heme-oxidized IRP2 ubiquitin ligase 1 (HOIL-1), HOIL-1-interacting protein (HOIP), and Shank-associated RH domain-interacting protein (SHARPIN), ,. HOIL-1 and HOIP are both E3 ubiquitin ligases, however, the specific linear ubiquitin-ligating activity is enacted by HOIP. Mice deficient in HOIP are embryonically lethal. Two cases of mutated HOIP have been detected in humans. These patients presented with autoinflammation and immunodeficiency,. HOIL-1 is required for LUBAC assembly and stability as demonstrated by embryonic lethality in HOIL-1 deficient mice. Recently, it has been noted, that HOIL-1 is also able to catalyze formation of oxyester bonds between the C-terminus of ubiquitin and serine/threoni ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
E3 Ubiquitin Ligase
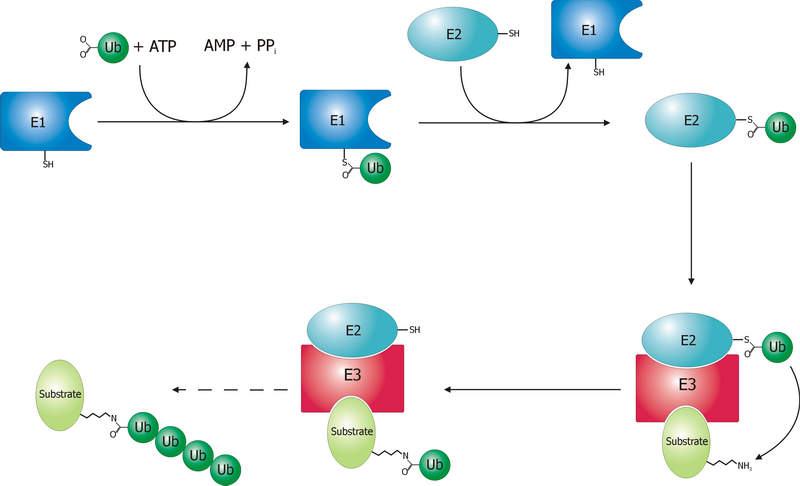
A ubiquitin ligase (also called an E3 ubiquitin ligase) is a protein that recruits an E2 ubiquitin-conjugating enzyme that has been loaded with ubiquitin, recognizes a protein substrate, and assists or directly catalyzes the transfer of ubiquitin from the E2 to the protein substrate. In simple and more general terms, the ligase enables movement of ubiquitin from a ubiquitin carrier to another thing (the substrate) by some mechanism. The ubiquitin, once it reaches its destination, ends up being attached by an isopeptide bond to a lysine residue, which is part of the target protein. E3 ligases interact with both the target protein and the E2 enzyme, and so impart substrate specificity to the E2. Commonly, E3s polyubiquitinate their substrate with Lys48-linked chains of ubiquitin, targeting the substrate for destruction by the proteasome. However, many other types of linkages are possible and alter a protein's activity, interactions, or localization. Ubiquitination by E3 ligases regu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyubiquitination
Ubiquitin is a small (8.6 kDa) regulatory protein found in most tissues of eukaryotic organisms, i.e., it is found ''ubiquitously''. It was discovered in 1975 by Gideon Goldstein and further characterized throughout the late 1970s and 1980s. Four genes in the human genome code for ubiquitin: UBB, UBC, UBA52 and RPS27A. The addition of ubiquitin to a substrate protein is called ubiquitylation (or, alternatively, ubiquitination or ubiquitinylation). Ubiquitylation affects proteins in many ways: it can mark them for degradation via the proteasome, alter their cellular location, affect their activity, and promote or prevent protein interactions. Ubiquitylation involves three main steps: activation, conjugation, and ligation, performed by ubiquitin-activating enzymes (E1s), ubiquitin-conjugating enzymes (E2s), and ubiquitin ligases (E3s), respectively. The result of this sequential cascade is to bind ubiquitin to lysine residues on the protein substrate via an isopeptide bond, cy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |