|
Isotopes Of Gallium
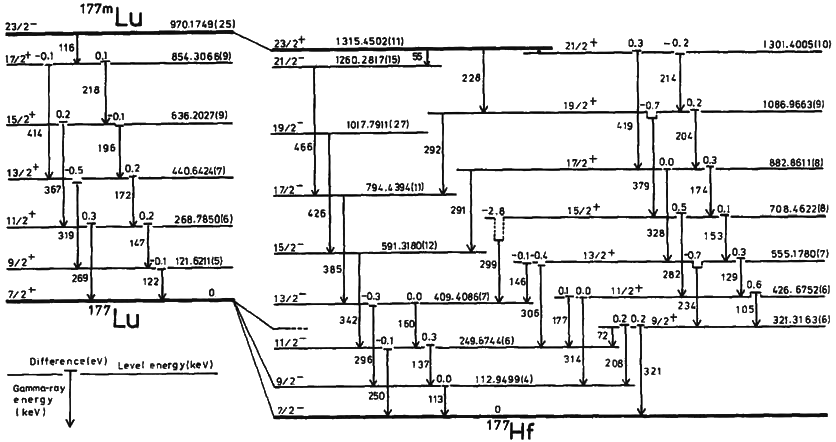
Natural gallium (31Ga) consists of a mixture of two stable isotopes: gallium-69 and gallium-71. The most commercially important radioisotopes are gallium-67 and gallium-68. Gallium-67 (half-life 3.3 days) is a gamma-emitting isotope (the gamma ray emitted immediately after electron capture) used in standard nuclear medical imaging, in procedures usually referred to as gallium scans. It is usually used as the free ion, Ga3+. It is the longest-lived radioisotope of gallium. The shorter-lived gallium-68 (half-life 68 minutes) is a positron-emitting isotope generated in very small quantities from germanium-68 in gallium-68 generators or in much greater quantities by proton bombardment of 68Zn in low-energy medical cyclotrons, for use in a small minority of diagnostic PET scans. For this use, it is usually attached as a tracer to a carrier molecule (for example the somatostatin analogue DOTATOC), which gives the resulting radiopharmaceutical a different tissue-uptake specifici ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gallium
Gallium is a chemical element with the symbol Ga and atomic number 31. Discovered by French chemist Paul-Émile Lecoq de Boisbaudran in 1875, Gallium is in group 13 of the periodic table and is similar to the other metals of the group (aluminium, indium, and thallium). Elemental gallium is a soft, silvery metal in standard temperature and pressure. In its liquid state, it becomes silvery white. If too much force is applied, the gallium may fracture conchoidally. Since its discovery in 1875, gallium has widely been used to make alloys with low melting points. It is also used in semiconductors, as a dopant in semiconductor substrates. The melting point of gallium is used as a temperature reference point. Gallium alloys are used in thermometers as a non-toxic and environmentally friendly alternative to mercury, and can withstand higher temperatures than mercury. An even lower melting point of , well below the freezing point of water, is claimed for the alloy galinstan (62–� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isomeric Transition
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy higher energy levels than in the ground state of the same nucleus. "Metastable" describes nuclei whose excited states have half-lives 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half life (ordinarily on the order of 10−12 seconds). The term "metastable" is usually restricted to isomers with half-lives of 10−9 seconds or longer. Some references recommend 5 × 10−9 seconds to distinguish the metastable half life from the normal "prompt" gamma-emission half-life. Occasionally the half-lives are far longer than this and can last minutes, hours, or years. For example, the nuclear isomer survives so long (at least 1015 years) that it has never been observed to decay spontaneously. The half-life of a nuclear isomer can even exceed that of the ground state of the same nuclide, as shown by as well as , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Gallium
Natural gallium (31Ga) consists of a mixture of two stable isotopes: gallium-69 and gallium-71. The most commercially important radioisotopes are gallium-67 and gallium-68. Gallium-67 (half-life 3.3 days) is a gamma-emitting isotope (the gamma ray emitted immediately after electron capture) used in standard nuclear medical imaging, in procedures usually referred to as gallium scans. It is usually used as the free ion, Ga3+. It is the longest-lived radioisotope of gallium. The shorter-lived gallium-68 (half-life 68 minutes) is a positron-emitting isotope generated in very small quantities from germanium-68 in gallium-68 generators or in much greater quantities by proton bombardment of 68Zn in low-energy medical cyclotrons, for use in a small minority of diagnostic PET scans. For this use, it is usually attached as a tracer to a carrier molecule (for example the somatostatin analogue DOTATOC), which gives the resulting radiopharmaceutical a different tissue-uptake specifici ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pancreatic Cancer
Pancreatic cancer arises when cell (biology), cells in the pancreas, a glandular organ behind the stomach, begin to multiply out of control and form a Neoplasm, mass. These cancerous cells have the malignant, ability to invade other parts of the body. A number of types of pancreatic cancer are known. The most common, pancreatic adenocarcinoma, accounts for about 90% of cases, and the term "pancreatic cancer" is sometimes used to refer only to that type. These adenocarcinomas start within the part of the pancreas that makes digestive enzymes. Several other types of cancer, which collectively represent the majority of the non-adenocarcinomas, can also arise from these cells. About 1–2% of cases of pancreatic cancer are neuroendocrine tumors, which arise from the hormone-producing neuroendocrine cell, cells of the pancreas. These are generally less aggressive than pancreatic adenocarcinoma. Signs and symptoms of the most-common form of pancreatic cancer may include jaundice, ye ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PET-CT
Positron emission tomography–computed tomography (better known as PET-CT or PET/CT) is a nuclear medicine technique which combines, in a single gantry, a positron emission tomography (PET) scanner and an x-ray computed tomography (CT) scanner, to acquire sequential images from both devices in the same session, which are combined into a single superposed ( co-registered) image. Thus, functional imaging obtained by PET, which depicts the spatial distribution of metabolic or biochemical activity in the body can be more precisely aligned or correlated with anatomic imaging obtained by CT scanning. Two- and three-dimensional image reconstruction may be rendered as a function of a common software and control system. PET-CT has revolutionized medical diagnosis in many fields, by adding precision of anatomic localization to functional imaging, which was previously lacking from pure PET imaging. For example, many diagnostic imaging procedures in oncology, surgical planning, radia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Indium-111
Indium-111 (111In) is a radioactive isotope of indium (In). It decays by electron capture to stable cadmium-111 with a half-life of 2.8 days. Indium-111 chloride (111InCl) solution is produced by proton irradiation of a cadmium target (112Cd(p,2n) or 111Cd(p,n)) in a cyclotron, as recommended by International Atomic Energy Agency (IAEA). The former method is more commonly used as it results in a high level of radionuclide purity. Indium-111 is commonly used in nuclear medicine diagnostic imaging by radiolabeling targeted molecules or cells. During its radioactive decay, it emits low energy gamma (γ) photons which can be imaged using planar or single-photon emission computed tomography (SPECT) gamma cameras (primary energies (ε) of 171.3 keV (91%) and 245.4 keV (94%)) Uses in nuclear medicine When formulated as an 111InCl solution, it can be used to bind antibodies, peptides, or other molecular targeted proteins or other molecules, typically using a chelate to bind ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neuroendocrine Tumors
Neuroendocrine tumors (NETs) are neoplasms that arise from cells of the endocrine (hormonal) and nervous systems. They most commonly occur in the intestine, where they are often called carcinoid tumors, but they are also found in the pancreas, lung, and the rest of the body. Although there are many kinds of NETs, they are treated as a group of tissue because the cells of these neoplasms share common features, such as looking similar, having special secretory granules, and often producing biogenic amines and polypeptide hormones. Classification WHO The World Health Organization (WHO) classification scheme places neuroendocrine tumors into three main categories, which emphasize the tumor grade rather than the anatomical origin: * well-differentiated neuroendocrine tumours, further subdivided into tumors with benign and those with uncertain behavior * well-differentiated (low grade) neuroendocrine carcinomas with low-grade malignant behavior * poorly differentiated (high grade) ne ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Somatostatin
Somatostatin, also known as growth hormone-inhibiting hormone (GHIH) or by several other names, is a peptide hormone that regulates the endocrine system and affects neurotransmission and cell proliferation via interaction with G protein-coupled somatostatin receptors and inhibition of the release of numerous secondary hormones. Somatostatin inhibits insulin and glucagon secretion. Somatostatin has two active forms produced by the alternative cleavage of a single preproprotein: one consisting of 14 amino acids (shown in infobox to right), the other consisting of 28 amino acids. Among the vertebrates, there exist six different somatostatin genes that have been named ''SS1, SS2, SS3, SS4, SS5'' and ''SS6''. Zebrafish have all six. The six different genes, along with the five different somatostatin receptors, allow somatostatin to possess a large range of functions. Humans have only one somatostatin gene, ''SST''. Nomenclature Synonyms of "somatostatin" include: * growth hormone� ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Positron Emission
Positron emission, beta plus decay, or β+ decay is a subtype of radioactive decay called beta decay, in which a proton inside a radionuclide nucleus is converted into a neutron while releasing a positron and an electron neutrino (). Positron emission is mediated by the weak force. The positron is a type of beta particle (β+), the other beta particle being the electron (β−) emitted from the β− decay of a nucleus. An example of positron emission (β+ decay) is shown with magnesium-23 decaying into sodium-23: : → + + Because positron emission decreases proton number relative to neutron number, positron decay happens typically in large "proton-rich" radionuclides. Positron decay results in nuclear transmutation, changing an atom of one chemical element into an atom of an element with an atomic number that is less by one unit. Positron emission occurs only very rarely naturally on earth, when induced by a cosmic ray or from one in a hundred thousand decays of pot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Excited State
In quantum mechanics, an excited state of a system (such as an atom, molecule or nucleus) is any quantum state of the system that has a higher energy than the ground state (that is, more energy than the absolute minimum). Excitation refers to an increase in energy level above a chosen starting point, usually the ground state, but sometimes an already excited state. The temperature of a group of particles is indicative of the level of excitation (with the notable exception of systems that exhibit negative temperature). The lifetime of a system in an excited state is usually short: spontaneous or induced emission of a quantum of energy (such as a photon or a phonon) usually occurs shortly after the system is promoted to the excited state, returning the system to a state with lower energy (a less excited state or the ground state). This return to a lower energy level is often loosely described as decay and is the inverse of excitation. Long-lived excited states are often called ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gamma Emission
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically shorter than those of X-rays. With frequencies above 30 exahertz (), it imparts the highest photon energy. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900 while studying radiation emitted by radium. In 1903, Ernest Rutherford named this radiation ''gamma rays'' based on their relatively strong penetration of matter; in 1900 he had already named two less penetrating types of decay radiation (discovered by Henri Becquerel) alpha rays and beta rays in ascending order of penetrating power. Gamma rays from radioactive decay are in the energy range from a few kiloelectronvolts ( keV) to approximately 8 megaelectronvolts ( MeV), corresponding to the typical energy levels in nuclei with reasonably long ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Capture
Electron capture (K-electron capture, also K-capture, or L-electron capture, L-capture) is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L electron shells. This process thereby changes a nuclear proton to a neutron and simultaneously causes the emission of an electron neutrino. : : or when written as a nuclear reaction equation, ^_e + ^_p -> ^_n + ^_ ν_e Since this single emitted neutrino carries the entire decay energy, it has this single characteristic energy. Similarly, the momentum of the neutrino emission causes the daughter atom to recoil with a single characteristic momentum. The resulting daughter nuclide, if it is in an excited state, then transitions to its ground state. Usually, a gamma ray is emitted during this transition, but nuclear de-excitation may also take place by internal conversion. Following capture of an inner electron from the atom, an outer electron ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |